Advanced Ruthenium-Catalyzed Synthesis of Quetiapine Intermediates for Commercial Scale-Up
Advanced Ruthenium-Catalyzed Synthesis of Quetiapine Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking more efficient and environmentally sustainable pathways for the production of critical antipsychotic medications. A significant breakthrough in this domain is detailed in patent CN112939892B, which discloses a novel preparation method for quetiapine and its key precursors using advanced ruthenium catalysis. This technology addresses long-standing challenges in the synthesis of 11-[4-[2-(2-hydroxyethoxy)ethyl-1-piperazinyl]]-dibenzo[b,f][1,4]thiazepine, a vital intermediate for the blockbuster drug quetiapine hemifumarate. By shifting away from traditional halogenated alkylation agents, this innovation offers a greener, higher-yielding alternative that aligns perfectly with modern green chemistry principles and stringent regulatory requirements for impurity control.
For R&D directors and process chemists, the implications of this patent are profound, offering a route that not only simplifies the synthetic workflow but also drastically reduces the environmental footprint of the manufacturing process. The method leverages the unique ability of ruthenium complexes to facilitate direct N-alkylation using diethylene glycol, a cheap and readily available feedstock. This stands in stark contrast to legacy methods that rely on hazardous chloro-derivatives, thereby presenting a compelling case for technology transfer and adoption in large-scale commercial production facilities aiming for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the quetiapine side-chain precursor has relied heavily on nucleophilic substitution reactions involving halogenated hydrocarbons. Specifically, the reaction between 11-piperazinyl dibenzo[b,f][1,4]thiazepine and 2-(2-chloroethoxy)ethanol has been the standard industrial approach. While chemically straightforward, this conventional pathway suffers from significant drawbacks that impact both economic viability and environmental compliance. The use of chloro-containing reagents inevitably generates stoichiometric amounts of inorganic salt waste, typically hydrochloric acid salts, which require neutralization and disposal, adding substantial cost to the waste treatment infrastructure.
Furthermore, the atom economy of the halogenated route is inherently poor, as the chlorine atom serves merely as a leaving group and contributes nothing to the final molecular structure. This inefficiency translates to higher raw material consumption per kilogram of product. Additionally, halogenated intermediates often pose safety risks due to their potential toxicity and carcinogenicity, necessitating rigorous containment measures and increasing the complexity of operator safety protocols. The presence of residual halogens in the final product can also complicate purification steps, as removing trace halogenated impurities to meet pharmacopeial standards often requires multiple recrystallizations or chromatographic separations, further eroding overall process yield.
The Novel Approach
The methodology described in patent CN112939892B represents a paradigm shift by employing a ruthenium-catalyzed direct alkylation strategy using diethylene glycol. This approach elegantly bypasses the need for pre-functionalized halogenated electrophiles, utilizing the hydroxyl group of the glycol directly in the presence of a specialized transition metal catalyst. The reaction proceeds under relatively mild thermal conditions, typically between 120°C and 160°C, and crucially, water is generated as the sole byproduct. This transformation effectively turns a waste-generating substitution reaction into a clean addition-elimination process, dramatically improving the atom utilization rate of the entire synthesis.
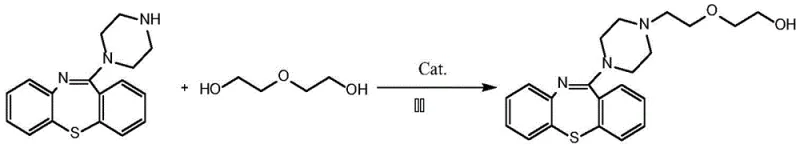
From a process engineering perspective, this novel route simplifies the downstream processing significantly. Since water is the only byproduct, it can be easily removed via azeotropic distillation or during the solvent evaporation step, eliminating the need for aqueous washes to remove inorganic salts. The patent data indicates that this method achieves impressive yields, with Example 1 demonstrating a yield of 93.0% and HPLC purity of 96.85%. Such high efficiency not only maximizes the output from expensive starting materials but also ensures a cleaner crude profile, reducing the burden on final purification units and enabling a more robust and reliable pharmaceutical intermediate supplier capability.
Mechanistic Insights into Ruthenium-Catalyzed N-Alkylation
The core of this technological advancement lies in the specific activation of the C-O bond in diethylene glycol by the ruthenium catalyst. Unlike traditional SN2 mechanisms that require a good leaving group like chloride, this transition metal-catalyzed process likely operates through a "borrowing hydrogen" or hydrogen autotransfer mechanism, although the patent broadly defines it as ruthenium catalysis. The ruthenium complex, such as bis(triphenylphosphine)carbonylacetonitrile ruthenium dichloride, coordinates with the alcohol substrate, facilitating the dehydrogenation of the hydroxyl group to form a reactive aldehyde intermediate in situ. This activated species then undergoes condensation with the secondary amine of the dibenzothiazepine core.
Following the condensation step, the ruthenium catalyst plays a second critical role by hydrogenating the resulting imine intermediate back to the stable amine product, effectively recycling the hydrogen atoms initially removed from the alcohol. This catalytic cycle ensures that no external reducing agents are required, and the overall stoichiometry remains clean. The choice of ligand on the ruthenium center is paramount; the patent highlights that phosphine ligands, specifically triphenylphosphine, provide the necessary electronic and steric environment to stabilize the active catalytic species at elevated temperatures without decomposing. This mechanistic elegance allows for the use of simple, non-toxic alcohols as alkylating agents, a feat that is difficult to achieve with base metal catalysts.
Impurity control in this system is inherently superior due to the specificity of the catalytic cycle. In conventional alkylation, over-alkylation or reaction at multiple sites on the piperazine ring can occur, leading to complex impurity profiles that are hard to separate. The ruthenium-catalyzed method, however, demonstrates high selectivity for the mono-alkylation of the piperazine nitrogen. The reaction conditions, particularly the use of anhydrous toluene or xylene, prevent hydrolysis of sensitive intermediates and minimize side reactions. The result is a product stream with high HPLC purity, often exceeding 96% even before final chromatographic purification, which is a critical metric for ensuring the quality of high-purity pharmaceutical intermediates entering the supply chain.
How to Synthesize Quetiapine Intermediate Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize the benefits of the ruthenium catalyst. The process begins with the careful selection of the catalyst loading and the molar ratio of the diol to the amine substrate. According to the patent examples, a molar ratio of catalyst to diethylene glycol to substrate of approximately 0.03:3:1 yields optimal results, balancing reaction rate with catalyst cost. The reaction is typically conducted in a pressure-resistant vessel to accommodate the elevated temperatures required to drive the equilibrium forward, with toluene serving as the preferred solvent due to its ability to form an azeotrope with water, thus driving the reaction to completion.
- Combine 11-piperazinyl dibenzo[b,f][1,4]thiazepine with diethylene glycol and a ruthenium catalyst (e.g., bis(triphenylphosphine)carbonylacetonitrile ruthenium dichloride) in an organic solvent like toluene.
- Heat the reaction mixture to a temperature range of 120°C to 160°C, preferably 140°C, and maintain stirring for 3 to 10 hours to ensure complete conversion.
- Cool the reaction to room temperature, remove the solvent under reduced pressure, and purify the crude product using chromatography to obtain the high-purity target intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed technology offers tangible strategic advantages beyond mere technical novelty. The primary benefit lies in the drastic simplification of the raw material portfolio. By replacing specialized, hazardous halogenated alkylating agents with commodity chemicals like diethylene glycol, manufacturers can insulate themselves from the price volatility and supply disruptions often associated with fine chemical halides. Diethylene glycol is produced on a massive global scale for the polyester industry, ensuring a stable, low-cost, and continuous supply stream that enhances the overall reliability of the quetiapine intermediate supply chain.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive waste treatment protocols. In the traditional halogenated route, the neutralization and disposal of acidic salt waste constitute a significant portion of the operational expenditure. By generating water as the only byproduct, this new method effectively removes these waste management costs entirely. Furthermore, the high atom economy means that less raw material is required to produce the same amount of product, directly lowering the cost of goods sold (COGS). The high yields reported, consistently above 90% in optimized examples, further contribute to substantial cost savings by minimizing material loss and maximizing throughput per batch.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes to a more predictable manufacturing schedule. Traditional methods often suffer from batch-to-batch variability due to the sensitivity of nucleophilic substitutions to moisture and impurities in the halogenated reagents. The ruthenium-catalyzed process, operating in anhydrous organic solvents with stable catalysts, demonstrates remarkable reproducibility. This consistency reduces the risk of failed batches and the need for re-processing, ensuring that delivery timelines to downstream API manufacturers are met with greater certainty. The use of common solvents like toluene and xylene also simplifies logistics, as these are standard items in any chemical inventory, reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on solvent emissions and hazardous waste, the green credentials of this synthesis route become a major asset. The absence of halogenated waste streams simplifies environmental permitting and reduces the liability associated with hazardous material handling. From a scalability perspective, the reaction conditions (120°C to 160°C) are well within the operating range of standard stainless steel reactors used in fine chemical plants, requiring no exotic high-pressure equipment. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates from pilot plant quantities to multi-ton annual production without significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within patent CN112939892B, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of the ruthenium-catalyzed method over traditional halogenated routes?
A: The ruthenium-catalyzed method utilizes diethylene glycol instead of toxic halogenated hydrocarbons like 2-(2-chloroethoxy)ethanol. This shift results in water being the only byproduct, significantly improving atom economy and eliminating the need for complex waste treatment associated with halogenated salts.
Q: Which ruthenium catalysts are most effective for this specific N-alkylation?
A: Patent CN112939892B identifies bis(triphenylphosphine)carbonylacetonitrile ruthenium dichloride as the preferred catalyst. Other viable options include bis(triphenylphosphine)carbonyl monohydrate ruthenium dichloride and tetrakis(triphenylphosphine)ruthenium dichloride, all operating effectively within the 120°C to 160°C range.
Q: How does solvent choice impact the reaction yield and purity?
A: The process requires anhydrous organic solvents such as toluene, xylene, or tert-amyl alcohol, with toluene being optimal. The use of aqueous solvents is strictly prohibited as moisture inhibits the catalytic cycle, while non-polar solvents facilitate the necessary thermal conditions and subsequent workup.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Intermediate Supplier
The transition to greener, more efficient synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle transition metal catalysis with the utmost precision, ensuring that stringent purity specifications are met for every batch. With rigorous QC labs and a commitment to continuous process improvement, we are uniquely positioned to deliver the high-purity pharmaceutical intermediates required by today's demanding global market.
We invite you to collaborate with us to leverage this advanced ruthenium-catalyzed technology for your quetiapine supply needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of this critical antipsychotic intermediate.
