Advanced Synthesis of Quetiapine Intermediates: Scalable Solutions for Global API Manufacturing
The pharmaceutical landscape for antipsychotic medications continues to evolve, with Quetiapine remaining a cornerstone therapy for schizophrenia and bipolar disorder. A pivotal advancement in the manufacturing of this critical active pharmaceutical ingredient is detailed in patent CN1239487C, which discloses a novel process for the preparation of 11-[4-[2-(2-hydroxyethoxy)ethyl]-1-piperazinyl]dibenzo[b,f]-1,4-thiazepine. This technical breakthrough addresses long-standing inefficiencies in the synthesis of Quetiapine intermediates, specifically targeting the instability of key precursors and the economic burdens of traditional purification methods. For global supply chain leaders and R&D directors, understanding this patented methodology is essential for optimizing production costs and ensuring the consistent availability of high-purity intermediates. The innovation lies not merely in a new chemical structure, but in a fundamental re-engineering of the synthetic pathway to favor industrial robustness over laboratory convenience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Quetiapine intermediates has been plagued by the reliance on unstable imino-halide derivatives, specifically the compound designated as Formula XI in the prior art. As illustrated in the structural representation below, this imino-halide contains a reactive carbon-halogen bond adjacent to the thiazepine nitrogen, rendering it highly susceptible to hydrolysis upon exposure to ambient moisture.
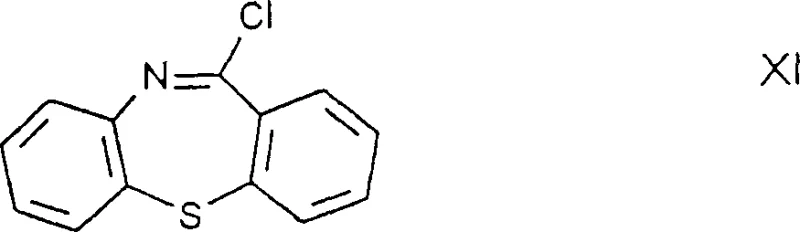
This inherent instability creates significant bottlenecks in commercial manufacturing, as it necessitates strictly anhydrous conditions and immediate processing to prevent degradation into hydrolysis byproducts that contaminate the final API. Furthermore, conventional routes often yield crude products that require silica gel column chromatography for purification, a technique that is notoriously difficult to scale, solvent-intensive, and economically prohibitive for multi-ton production. The cumulative effect of these limitations is a lower overall yield, typically hovering around 62.6% from urethane derivatives, and a supply chain vulnerable to batch-to-batch variability.
The Novel Approach
In stark contrast, the novel approach introduced in CN1239487C circumvents these pitfalls by utilizing a haloethylpiperazinylthiazepine derivative, known as Formula VIII, as the key reactive intermediate. This structural modification shifts the reactive halogen atom to the ethyl side chain of the piperazine ring, significantly enhancing the chemical stability of the molecule during storage and handling.
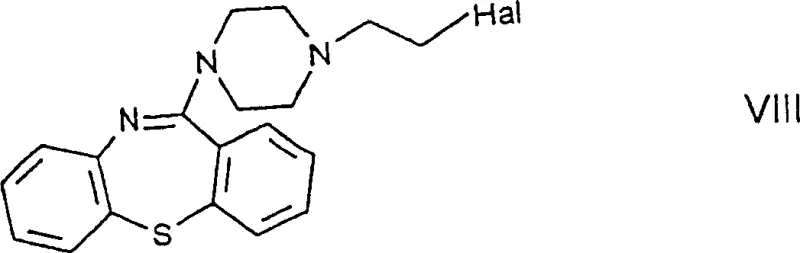
By employing this stable precursor, the new process eliminates the need for complex chromatographic purification, allowing for the isolation of high-purity products through straightforward crystallization techniques using solvents like diisopropyl ether. This shift from chromatography to crystallization is a critical enabler for commercial scale-up of complex psychotropic intermediates, as it drastically reduces solvent waste and processing time. The result is a more streamlined, cost-effective workflow that maintains high integrity of the thiazepine core while facilitating the final substitution reaction with ethylene glycol to form the target Quetiapine intermediate.
Mechanistic Insights into Williamson Ether Synthesis and Cyclization
The core chemical transformation in this novel pathway relies on a robust Williamson ether synthesis mechanism, where the haloethyl group of Formula VIII acts as the electrophile and the alkoxide of ethylene glycol serves as the nucleophile. In the preferred embodiment, sodium metal is utilized to convert ethylene glycol into its corresponding alcoholate in situ, creating a highly reactive nucleophilic species that attacks the terminal carbon of the chloroethyl side chain. This nucleophilic substitution is conducted at elevated temperatures, typically around 100°C, for a duration of approximately 9 hours to ensure complete conversion. The choice of sodium ethylene glycolate is strategic, as it provides a strong basic environment that drives the reaction forward without compromising the integrity of the dibenzothiazepine ring system, which can be sensitive to harsh acidic or overly aggressive basic conditions.
Impurity control is meticulously managed through the stability of the Formula VIII intermediate and the selectivity of the substitution reaction. Unlike the prior art imino-halides, which generate hydrolysis impurities that are difficult to separate, the byproducts in this new route are primarily inorganic salts and excess glycol, which are easily removed during the aqueous workup and subsequent crystallization steps. The patent data indicates that the final alkali base can be obtained with a remarkable yield of 98% before salt formation, demonstrating the high efficiency of this mechanistic pathway. Furthermore, the ability to convert the free base into a pharmaceutically acceptable salt, such as the half fumarate, through controlled crystallization ensures that the final impurity profile meets the stringent requirements of global regulatory bodies for high-purity pharmaceutical intermediates.
How to Synthesize Quetiapine Intermediate Efficiently
The operational execution of this synthesis involves a sequence of well-defined steps beginning with the preparation of the Formula VIII intermediate from urethane derivatives and hydroxyethyl piperazine, followed by halogenation and cyclization using phosphoryl chloride and vanadium pentoxide. Once the stable haloethyl precursor is secured, the critical etherification step is performed by reacting the precursor with sodium-treated ethylene glycol in a toluene medium. The reaction mixture is heated to reflux to drive the substitution to completion, after which the product is isolated through aqueous extraction and crystallization.
- Preparation of the key intermediate Formula VIII by reacting urethane derivatives with hydroxyethyl piperazine followed by halogenation and cyclization using phosphoryl chloride.
- Execution of the Williamson ether synthesis by reacting the Formula VIII haloethyl derivative with ethylene glycol in the presence of sodium metal at 100°C.
- Final purification and salt formation using fumaric acid to obtain the stable half fumarate salt of the Quetiapine intermediate with high crystallinity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic advantages that extend beyond simple chemical yield improvements. The primary value driver is the elimination of column chromatography, which translates directly into cost reduction in API manufacturing by removing the need for expensive silica gel, vast quantities of organic solvents, and the associated waste disposal costs. This process intensification allows for a more continuous and predictable production flow, reducing the operational expenditure per kilogram of the intermediate. Additionally, the enhanced stability of the Formula VIII intermediate mitigates the risk of raw material spoilage during storage and transit, ensuring that inventory value is preserved and supply continuity is maintained even in less-than-ideal warehouse conditions.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental shift in purification technology. By replacing silica gel chromatography with crystallization, manufacturers can significantly reduce solvent consumption and waste generation, leading to substantial cost savings in both raw materials and environmental compliance. The higher overall yield of 66-67% compared to the prior art's 62.6% means that less starting material is required to produce the same amount of final product, effectively lowering the cost of goods sold (COGS) and improving margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the chemical stability of the key intermediates. The conventional imino-halide required strict moisture control and rapid processing, creating fragile links in the supply chain. In contrast, the haloethylpiperazinylthiazepine derivative is robust, allowing for longer storage times and more flexible logistics planning. This stability reduces the risk of batch failures due to environmental exposure, ensuring a more reliable flow of materials to downstream API synthesis sites and reducing the need for safety stock buffers.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the new route is inherently greener and more scalable. Crystallization is a unit operation that scales linearly and predictably from the laboratory to the multi-ton reactor, whereas chromatography often faces non-linear scaling challenges. The reduction in solvent usage and the elimination of silica waste align with modern green chemistry principles, facilitating easier regulatory approval and reducing the environmental footprint of the manufacturing facility, which is increasingly a key criterion for supplier selection by major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims within CN1239487C, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these details is crucial for technical teams assessing the feasibility of technology transfer and for commercial teams negotiating supply agreements based on process efficiency.
Q: How does the novel process improve stability compared to conventional imino-halide methods?
A: The conventional method relies on Formula XI imino-halide, which is highly unstable and prone to hydrolysis in moist air, leading to yield loss and impurity generation. The novel process utilizes Formula VIII haloethylpiperazinylthiazepine, which offers superior chemical stability, allowing for easier handling and storage without the strict anhydrous conditions required by the prior art.
Q: What are the purification advantages of this new synthetic route?
A: Traditional methods often necessitate silica gel column chromatography to achieve acceptable purity, which is impractical for large-scale manufacturing due to solvent consumption and throughput limitations. The patented method enables purification through crystallization (e.g., using diisopropyl ether or hexane-ethyl acetate), significantly reducing operational costs and facilitating industrial scale-up.
Q: What is the overall yield efficiency of the new method versus the old method?
A: The prior art methods reported overall yields of approximately 55.3% to 62.6% calculated from urethane derivatives. The novel process described in CN1239487C achieves an overall yield of 66-67%, driven by high-yielding individual steps such as the 98% conversion in the final glycol substitution reaction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a patent is only realized through expert engineering and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by CN1239487C are fully actualized in a GMP environment. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of validating the impurity profiles of complex intermediates like Formula VIII and the final Quetiapine precursor, guaranteeing that every batch meets the exacting standards required by global regulatory agencies.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthesis for their supply chains. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this route for your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing you to make data-driven decisions that enhance your competitive edge in the antipsychotic market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
