Revolutionizing Quetiapine Intermediate Production: A Deep Dive into Ruthenium-Catalyzed Green Synthesis
Revolutionizing Quetiapine Intermediate Production: A Deep Dive into Ruthenium-Catalyzed Green Synthesis
The pharmaceutical industry is constantly seeking more efficient, sustainable, and cost-effective pathways for the synthesis of critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is detailed in patent CN112939892A, which discloses a novel preparation method for quetiapine, a widely prescribed atypical antipsychotic medication. This patent introduces a sophisticated ruthenium-catalyzed alkylation strategy that fundamentally shifts the synthetic paradigm away from traditional, waste-intensive halogenation methods. By leveraging the unique catalytic properties of ruthenium complexes, this technology enables the direct coupling of 11-piperazinyl dibenzo[b,f][1,4]thiazepine with diethylene glycol. The result is a streamlined process that not only delivers exceptional chemical yields but also aligns perfectly with modern green chemistry principles by generating water as the sole byproduct. For global procurement and R&D teams, this represents a pivotal opportunity to optimize supply chains for high-purity quetiapine intermediates while drastically reducing the environmental footprint of manufacturing operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of quetiapine and its key intermediates has relied heavily on nucleophilic substitution reactions involving halogenated alkylating agents. Specifically, the conventional route typically employs 2-(2-chloroethoxy)ethanol to introduce the necessary side chain onto the dibenzothiazepine core. While chemically feasible, this legacy approach suffers from severe drawbacks that impact both economic viability and environmental compliance. The use of chlorinated reagents inherently generates stoichiometric amounts of corrosive halide salts as waste, necessitating complex and costly downstream purification processes to meet stringent pharmaceutical purity standards. Furthermore, halogenated compounds are often subject to stricter regulatory controls due to their toxicity and potential carcinogenicity, complicating logistics and increasing the overall cost of goods sold (COGS). The atom economy of such substitution reactions is inherently poor, as the halogen leaving group is discarded rather than utilized, leading to significant material inefficiency that scales poorly in multi-ton production campaigns.
The Novel Approach
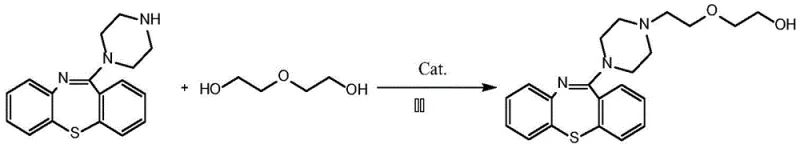
In stark contrast, the methodology outlined in patent CN112939892A offers a transformative solution by utilizing diethylene glycol, a benign and inexpensive commodity chemical, as the alkylating source. This innovative route bypasses the need for pre-functionalized halogenated intermediates entirely, relying instead on a catalytic activation mechanism mediated by specialized ruthenium complexes. The transition to this alcohol-based alkylation strategy effectively eliminates the generation of halide waste streams, replacing them with water as the only stoichiometric byproduct. This shift not only simplifies the workup procedure—often requiring merely phase separation and drying—but also dramatically improves the overall atom utilization of the process. For a reliable pharmaceutical intermediates supplier, adopting this technology means accessing a raw material supply chain that is more robust, less regulated, and significantly cheaper than the volatile market for specialized halogenated reagents, thereby securing a competitive advantage in cost reduction in API manufacturing.
Mechanistic Insights into Ruthenium-Catalyzed Alkylation
The core of this technological advancement lies in the sophisticated catalytic cycle facilitated by the ruthenium complex, likely operating through a "borrowing hydrogen" or hydrogen autotransfer mechanism. In this elegant sequence, the ruthenium catalyst first dehydrogenates the diethylene glycol to generate a reactive aldehyde intermediate in situ, simultaneously forming a ruthenium-hydride species. This transient aldehyde then undergoes condensation with the secondary amine of the 11-piperazinyl dibenzo[b,f][1,4]thiazepine precursor to form an imine or iminium ion. Subsequently, the ruthenium-hydride species generated in the initial step reduces this unsaturated intermediate back to the saturated amine product, regenerating the active catalyst and releasing a molecule of water. This closed-loop redox neutral process ensures that no external oxidants or reductants are required, minimizing reagent costs and safety hazards associated with handling reactive hydride sources. The choice of ligand environment on the ruthenium center, such as triphenylphosphine derivatives mentioned in the patent, is critical for tuning the electronic properties of the metal to facilitate these specific dehydrogenation and hydrogenation steps efficiently under mild thermal conditions.
Beyond the primary catalytic cycle, the control of impurity profiles is a paramount concern for R&D directors overseeing the scale-up of high-purity quetiapine intermediates. The selectivity of the ruthenium catalyst plays a crucial role in suppressing side reactions such as over-alkylation or ether formation, which are common pitfalls in alcohol aminations. The patent data indicates that by carefully optimizing the molar ratio of the catalyst to substrates and selecting appropriate organic solvents like toluene or xylene, the formation of undesired byproducts is minimized. The reaction conditions, specifically temperatures ranging from 120°C to 160°C, provide sufficient thermal energy to drive the equilibrium towards the product without promoting thermal degradation of the sensitive dibenzothiazepine scaffold. Furthermore, the use of anhydrous conditions and specific solvent systems helps to prevent hydrolysis of the intermediate imine species before reduction can occur, ensuring that the final isolated product meets the rigorous purity specifications of ≥95% required for downstream salt formation and final API processing.
How to Synthesize Quetiapine Intermediate Efficiently
The practical implementation of this synthesis involves a straightforward yet precise operational protocol that balances reaction kinetics with safety considerations. The process begins with the charging of a pressure-resistant reactor with the amine precursor, diethylene glycol, and the selected ruthenium catalyst in a dry organic solvent. The mixture is then heated to the optimal temperature window, typically around 140°C, and maintained under agitation for a duration of 3 to 10 hours to ensure complete conversion. Following the reaction, the workup procedure is remarkably simple compared to traditional methods; the solvent is removed under reduced pressure, and the crude residue is dissolved in ethyl acetate for aqueous washing to remove any polar impurities or catalyst residues.
- Charge a pressure tube with 11-piperazinyl dibenzo[b,f][1,4]thiazepine, diethylene glycol, and a ruthenium catalyst (e.g., bis(triphenylphosphine)carbonylacetonitrile ruthenium dichloride) in an organic solvent like toluene.
- Heat the reaction mixture to a temperature range of 120-160°C (optimally 140°C) and maintain stirring for 3 to 10 hours to facilitate the alkylation.
- Upon completion, cool the mixture, remove the solvent under reduced pressure, dissolve the residue in ethyl acetate, wash with water, dry, and purify via chromatography to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ruthenium-catalyzed process translates into tangible strategic benefits that extend far beyond the laboratory bench. The elimination of halogenated reagents removes a significant layer of regulatory complexity and hazard management from the supply chain, reducing the risk of shipment delays or storage compliance issues. Moreover, diethylene glycol is a globally available bulk chemical with a stable price point, insulating the manufacturing process from the volatility often seen in the market for specialized fine chemical building blocks. This stability allows for more accurate long-term cost forecasting and budget planning, essential for maintaining margins in the competitive generic pharmaceutical sector. The simplified downstream processing, characterized by the absence of salt waste and the generation of water as the only byproduct, also leads to substantial reductions in waste disposal costs and wastewater treatment loads, further enhancing the overall economic efficiency of the production facility.
- Cost Reduction in Manufacturing: The transition to a catalytic alkylation process using commodity alcohols fundamentally alters the cost structure of producing this key intermediate. By removing the need for expensive and hazardous halogenated alkylating agents, the direct material costs are significantly lowered. Additionally, the high atom economy of the borrowing hydrogen mechanism means that a greater proportion of the input mass is converted into valuable product rather than waste, maximizing the return on raw material investment. The simplified purification workflow, which avoids complex scavenging steps for heavy metals or halides, further reduces operational expenditures related to consumables and labor, driving down the total cost of ownership for the manufacturing process.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on diethylene glycol, a chemical produced on a massive industrial scale for diverse applications including antifreeze and polymers. This ubiquity ensures a continuous and reliable supply, mitigating the risk of shortages that can plague the supply of niche halogenated intermediates. The robustness of the reaction conditions also allows for flexibility in sourcing solvents, as the patent demonstrates efficacy in toluene, xylene, and other common organic media. This flexibility empowers supply chain managers to negotiate better terms with multiple vendors and adapt quickly to regional availability fluctuations, ensuring uninterrupted production schedules for reducing lead time for high-purity antipsychotic intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a major leap forward in sustainability. The generation of water as the sole byproduct aligns perfectly with increasingly stringent global environmental regulations regarding industrial effluent. The absence of halogenated waste streams simplifies the permitting process for new manufacturing lines and reduces the liability associated with hazardous waste handling. Furthermore, the mild reaction temperatures and the use of standard pressure equipment make the process highly amenable to commercial scale-up of complex psychiatric drug intermediates, allowing manufacturers to increase batch sizes safely without encountering the thermal runaway risks often associated with exothermic halogenation reactions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of the technology's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production workflows or for procurement specialists assessing the quality implications of the new supply source.
Q: What are the primary advantages of the ruthenium-catalyzed method over traditional halogenated routes?
A: The ruthenium-catalyzed method utilizes diethylene glycol instead of toxic halogenated reagents like 2-(2-chloroethoxy)ethanol. This results in water being the only byproduct, significantly reducing environmental waste and eliminating the need for expensive heavy metal scavenging steps associated with halogenation.
Q: What yields and purity levels can be expected from this synthesis protocol?
A: According to patent CN112939892A, this method consistently achieves product yields of ≥90% and HPLC purity levels exceeding 95%. Specific examples demonstrated yields up to 93.0% with purity reaching 96.85%, ensuring high efficiency for industrial scale-up.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process is designed for industrial production. It employs mild reaction conditions (120-160°C), uses commodity chemicals like diethylene glycol, and avoids hazardous halogenated solvents, making it safer and more cost-effective for commercial scale-up of complex psychiatric drug intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quetiapine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively analyzed the potential of the ruthenium-catalyzed route described in CN112939892A and is fully prepared to leverage this technology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of quetiapine intermediate we deliver meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to explore how this advanced synthesis platform can optimize your specific supply chain requirements. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a product, but to a comprehensive technical partnership focused on value creation. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume needs. We are ready to provide specific COA data and detailed route feasibility assessments to demonstrate how we can support your long-term strategic goals in the antipsychotic drug sector.
