Industrial Scale-Up of Pramipexole: Advanced Reduction Technology for Global Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing pathways for critical neurotherapeutic agents, particularly for the treatment of Parkinson's disease where supply consistency is paramount. Patent CN102442972B introduces a transformative industrial preparation method for pramipexole and its dihydrochloride monohydrate, addressing long-standing safety and efficiency bottlenecks in the synthesis of this dopamine agonist. This technical insight report analyzes the strategic value of this novel reduction protocol, which utilizes a sodium borohydride and iodine system to replace hazardous traditional reagents. By shifting from pyrophoric borane gas to a controlled in-situ generation mechanism, the process significantly enhances operational safety while maintaining exceptional stereochemical integrity. For R&D directors and supply chain leaders, understanding this technological pivot is essential for securing a reliable pramipexole intermediate supplier capable of meeting global regulatory and volume demands without compromising on cost or quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pramipexole has relied heavily on reduction strategies that introduce substantial operational risks and logistical complexities to the manufacturing environment. Early methodologies, such as those utilizing borane-THF complexes or gaseous diborane, require stringent safety protocols due to the spontaneous flammability and explosive nature of the reagents involved. Furthermore, alternative approaches employing boron trifluoride etherate as a catalyst often suffer from severe equipment corrosion issues and sensitivity to moisture, leading to inconsistent batch quality and increased maintenance downtime. The use of lithium aluminum hydride, while effective in laboratory settings, presents prohibitive costs and extreme sensitivity to air and water that make it unsuitable for large-scale industrial application. These conventional routes often necessitate specialized explosion-proof facilities and complex waste treatment systems for hazardous byproducts, thereby inflating the overall cost of goods sold. Consequently, manufacturers relying on these legacy technologies face significant supply chain vulnerabilities and regulatory scrutiny regarding process safety and environmental compliance.
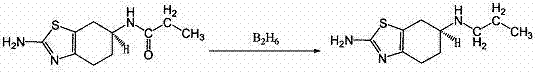
The Novel Approach
The innovative process disclosed in the patent data fundamentally reengineers the reduction step by employing a sodium borohydride and iodine system within a tetrahydrofuran solvent matrix. This approach allows for the in-situ generation of the active reducing species under mild conditions, effectively bypassing the need to handle dangerous borane gas directly. The reaction proceeds at temperatures ranging from -30°C to 50°C, which are easily manageable in standard stainless steel reactors without the need for extreme cryogenic infrastructure. By carefully controlling the molar ratios of sodium borohydride to iodine, the method ensures complete conversion of the propionamido precursor to the desired propylamino structure with high selectivity. This technological shift not only mitigates the safety hazards associated with traditional reducing agents but also simplifies the post-reaction workup and solvent recovery processes. The result is a streamlined production workflow that offers superior scalability and reliability for commercial manufacturing of high-purity pharmaceutical intermediates.
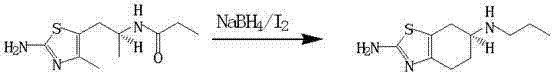
Mechanistic Insights into NaBH4/I2-Catalyzed Reduction
At the core of this synthesis lies a sophisticated chemical mechanism where iodine reacts with sodium borohydride to generate borane equivalents directly within the reaction medium. This in-situ generation ensures that the concentration of the active reducing agent is maintained at an optimal level throughout the reaction cycle, preventing side reactions that could lead to impurity formation. The mild thermal profile, specifically the controlled warming to 35-40°C after the initial addition at low temperatures, facilitates a smooth reduction of the amide functionality to the amine without affecting the sensitive tetrahydrobenzothiazole ring system. The stereochemical integrity of the S-configuration is rigorously preserved during this transformation, which is critical for the biological activity of the final API. Furthermore, the quenching process involving hydrochloric acid effectively decomposes any residual boron species, simplifying the purification steps and ensuring that heavy metal or boron residues remain well below regulatory thresholds. This mechanistic precision allows for the consistent production of pramipexole base with molar yields reaching up to 98%, demonstrating the robustness of the chemical pathway.
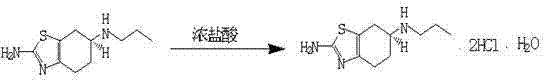
Following the reduction, the conversion to the dihydrochloride monohydrate form is executed through a controlled salification process that maximizes crystal purity and stability. The free base is dissolved in an alcohol solvent and treated with concentrated hydrochloric acid at low temperatures to precipitate the salt form. The use of activated carbon during this stage plays a pivotal role in adsorbing colored impurities and trace organic byproducts, contributing to the final product purity of greater than 99.9%. The specific molar ratio of hydrochloric acid to the free base is optimized to prevent excessive solubility losses while ensuring complete salt formation. This careful balance results in a decomposition point of 290-297°C, indicating a highly stable crystalline structure suitable for long-term storage and downstream tablet formulation. The entire sequence from reduction to salt formation is designed to minimize solvent usage and maximize recovery, aligning with green chemistry principles and cost-efficiency goals.
How to Synthesize Pramipexole Efficiently
The operational execution of this synthesis route requires precise adherence to the specified addition sequences and temperature controls to ensure optimal yield and safety. The process begins with the dissolution of the starting material in THF followed by the addition of sodium borohydride, after which the iodine solution is introduced dropwise under strict temperature monitoring. Detailed standardized synthesis steps see the guide below for specific parameters regarding stirring rates, addition times, and workup procedures.
- Dissolve (-)-2-amino-6-propionamido-4,5,6,7-tetrahydrobenzothiazole in THF and add sodium borohydride under stirring at ambient temperature.
- Cool the mixture to -30 to 10°C and slowly drip iodine THF solution to generate borane in situ for the reduction reaction.
- Warm to 35-40°C for reaction completion, quench with acid, basify to precipitate the free base, and convert to dihydrochloride monohydrate using concentrated HCl.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this manufacturing technology offers substantial advantages by eliminating the dependency on hazardous and expensive reagents that typically drive up operational costs. The shift away from pyrophoric borane and corrosive boron trifluoride reduces the need for specialized containment infrastructure, allowing for production in standard chemical facilities with lower capital expenditure. This simplification of the process equipment requirements directly translates to reduced maintenance costs and higher asset utilization rates over the lifecycle of the production line. Additionally, the use of readily available and stable reagents like sodium borohydride and iodine ensures a resilient supply chain that is less susceptible to market volatility or logistics disruptions associated with dangerous goods transportation. For supply chain heads, this means a more predictable lead time for high-purity pharmaceutical intermediates and a lower risk profile for business continuity planning.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reducing agents such as lithium aluminum hydride or gaseous borane significantly lowers the raw material cost per kilogram of the final product. By avoiding the need for specialized explosion-proof reactors and complex gas handling systems, the capital investment required for setting up production lines is drastically reduced. The mild reaction conditions also result in lower energy consumption for heating and cooling, contributing to overall operational expense savings. Furthermore, the high molar yield minimizes raw material waste, ensuring that a greater proportion of input materials are converted into saleable product, thereby optimizing the cost structure for cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The reliance on stable, non-hazardous reagents simplifies logistics and storage requirements, removing the regulatory burdens associated with transporting pyrophoric or toxic substances. This ease of handling ensures that raw material inventory can be maintained at optimal levels without the risk of degradation or safety incidents, leading to consistent production schedules. The robustness of the process against minor variations in conditions means that batch-to-batch consistency is high, reducing the likelihood of production delays due to failed quality control tests. Consequently, partners can rely on a steady flow of commercial scale-up of complex pharmaceutical intermediates without the interruptions common in more volatile synthetic routes.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing common solvents and standard reaction vessels that can be easily expanded from pilot scale to multi-ton commercial production. The absence of heavy metal catalysts and the use of benign reagents simplify waste treatment protocols, ensuring compliance with increasingly stringent environmental regulations. The efficient solvent recovery systems integrated into the workflow further reduce the environmental footprint by minimizing volatile organic compound emissions. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing operation, making it an attractive option for environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the industrial production of pramipexole using this advanced reduction methodology. These answers are derived directly from the patent specifications and practical manufacturing considerations to provide clarity for potential partners.
Q: Why is the NaBH4/I2 system preferred over traditional borane reduction for pramipexole?
A: Traditional methods using gaseous borane or borane-THF complexes pose significant safety risks due to pyrophoricity and explosion hazards. The NaBH4/I2 system generates borane in situ under mild conditions, eliminating the need for high-pressure gas handling and reducing equipment corrosion risks associated with BF3 catalysts.
Q: What purity levels can be achieved with this industrial preparation method?
A: The patented process consistently yields pramipexole dihydrochloride monohydrate with a purity exceeding 99.9%. The specific control of reaction temperatures and the use of activated carbon for decolorization ensure that impurity profiles meet stringent pharmaceutical standards for API intermediates.
Q: Is this synthesis route scalable for commercial metric ton production?
A: Yes, the method utilizes common solvents like THF and stable reagents like sodium borohydride and iodine. The reaction conditions are mild (20-50°C) and do not require extreme cryogenic temperatures or specialized high-pressure vessels, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pramipexole Dihydrochloride Monohydrate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced synthetic pathways like the one described in CN102442972B to deliver superior value to global pharmaceutical partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pramipexole intermediate meets the highest international standards for safety and efficacy. Our commitment to technological excellence allows us to offer a reliable pramipexole intermediate supplier partnership that is built on trust, quality, and consistent performance.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain needs. By collaborating with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to our optimized manufacturing process. Let us help you secure a stable supply of high-purity pharmaceutical intermediates while driving down costs and enhancing your overall production efficiency.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
