Advanced Catalytic Hydrogenation of Biomass Phenolics for High-Purity Cyclohexanol Manufacturing
The global chemical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by the urgent need to mitigate the environmental impact of fossil fuel dependency. In this context, patent CN107089898B presents a groundbreaking methodology for the catalytic hydrogenation of biomass-derived phenolic compounds into valuable cyclohexanol derivatives. This technology leverages a heterogeneous palladium-on-titanium-dioxide (Pd/TiO2) catalyst system to achieve high-efficiency conversion under relatively mild conditions. For R&D directors and process engineers, this represents a critical advancement in green chemistry, offering a viable pathway to transform renewable lignin degradation products into high-value pharmaceutical intermediates and fuel additives. The significance of this patent lies not only in its chemical efficacy but also in its alignment with global sustainability goals, providing a robust framework for the commercial scale-up of complex bio-based intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing cyclohexanol compounds often rely on homogeneous catalysts or harsh reaction environments that pose significant challenges for industrial application. Conventional processes frequently utilize volatile organic compounds (VOCs) as solvents, which necessitate complex recovery systems and generate substantial hazardous waste streams, thereby inflating operational expenditures and complicating regulatory compliance. Furthermore, many existing catalytic systems suffer from poor selectivity, leading to the formation of unwanted by-products such as cyclohexanones or over-hydrogenated species, which require energy-intensive purification steps to meet the stringent purity specifications demanded by the pharmaceutical sector. The reliance on non-renewable feedstocks in these legacy processes also exposes supply chains to the volatility of petrochemical markets, creating long-term strategic vulnerabilities for manufacturers seeking stability and cost predictability in their raw material sourcing.
The Novel Approach
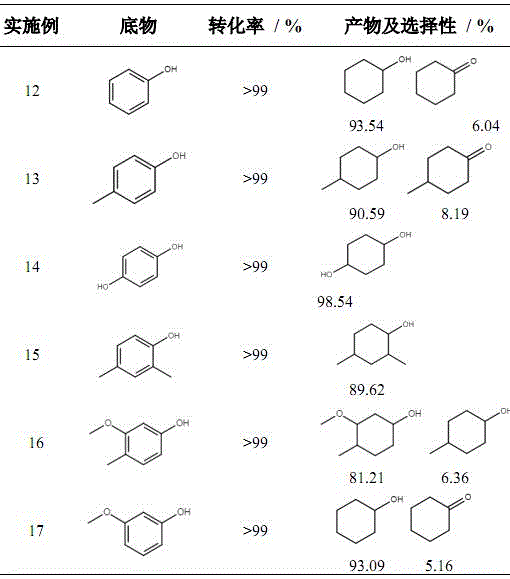
In stark contrast, the novel approach detailed in the patent utilizes a heterogeneous Pd/TiO2 catalyst that enables the reaction to proceed efficiently in water, a benign and inexpensive solvent. This shift to an aqueous medium eliminates the need for toxic organic solvents, drastically simplifying the downstream processing and waste treatment protocols. The catalyst exhibits exceptional activity and selectivity, capable of converting a wide range of phenolic substrates, including guaiacol, phenol, and p-cresol, into their corresponding cyclohexanols with conversion rates often exceeding 99%. This versatility allows for the processing of diverse biomass streams, enhancing the economic viability of biorefineries. By integrating renewable feedstocks with a robust catalytic system, this method offers a sustainable solution for cost reduction in fine chemical manufacturing, ensuring a stable supply of critical intermediates while minimizing the environmental footprint.
Mechanistic Insights into Pd/TiO2-Catalyzed Hydrogenation
The core of this technological breakthrough lies in the synergistic interaction between the palladium active sites and the titanium dioxide support. The Pd nanoparticles dispersed on the TiO2 surface facilitate the dissociative adsorption of molecular hydrogen, generating active hydrogen species that are crucial for the reduction process. Simultaneously, the aromatic ring of the phenolic substrate adsorbs onto the catalyst surface, likely through pi-interactions, positioning it optimally for hydrogen attack. The unique electronic properties of the TiO2 support may also play a role in stabilizing the metal particles and modifying their electronic state, thereby enhancing catalytic performance and resistance to deactivation. This mechanistic understanding is vital for R&D teams aiming to optimize reaction parameters, as it suggests that controlling the dispersion of palladium and the surface properties of the support can further tune the selectivity towards the desired alcohol product over the ketone by-product.
Furthermore, the use of water as a solvent is not merely an environmental choice but a mechanistic advantage in this specific system. Water can participate in hydrogen bonding networks that stabilize transition states or intermediates, potentially lowering the activation energy for the hydrogenation of the aromatic ring. The high polarity of water also aids in the solubility of polar phenolic substrates, ensuring efficient mass transfer to the catalyst surface. From an impurity control perspective, the high selectivity observed (e.g., 93.54% for cyclohexanol from phenol) indicates that the catalyst effectively suppresses side reactions such as demethoxylation or ring opening, which are common pitfalls in phenolic hydrogenation. This level of control is essential for producing high-purity pharmaceutical intermediates, where even trace impurities can compromise the safety and efficacy of the final drug product.
How to Synthesize Cyclohexanol Compounds Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a pilot or production setting. The process begins with the meticulous preparation of the catalyst, involving the impregnation of titanium dioxide with a palladium precursor, followed by controlled drying, calcination, and reduction steps to activate the metal sites. Once the catalyst is prepared, the hydrogenation reaction is conducted in a pressurized reactor where the phenolic substrate is mixed with water and the catalyst under a hydrogen atmosphere. The reaction conditions, including temperature and pressure, are adjustable to accommodate different substrates, allowing for flexibility in processing various biomass-derived feedstocks. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the Pd/TiO2 catalyst by dispersing titanium dioxide in water, adding a palladium precursor, stirring, drying, calcining at 400°C, and reducing under hydrogen.
- Load the reactor with the prepared catalyst, water as the solvent, and the specific phenolic substrate (e.g., guaiacol or phenol).
- Conduct the hydrogenation reaction at temperatures between 40-200°C and pressures of 0.1-5 MPa, followed by filtration and analysis.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this catalytic hydrogenation technology offers compelling strategic advantages that extend beyond simple chemical conversion. The primary benefit stems from the utilization of water as the reaction medium, which is universally available, non-flammable, and virtually free compared to specialized organic solvents. This substitution leads to a significant reduction in raw material costs and eliminates the logistical complexities associated with the storage and handling of hazardous chemicals. Moreover, the high conversion efficiency and selectivity of the Pd/TiO2 catalyst minimize the generation of waste by-products, thereby reducing the burden on waste treatment facilities and lowering the overall cost of goods sold (COGS). These factors collectively contribute to a more resilient and cost-effective supply chain, enabling manufacturers to offer competitive pricing without compromising on quality or sustainability standards.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and the reduction in energy consumption for solvent recovery directly translate to lower operational expenditures. Additionally, the high selectivity of the catalyst reduces the need for complex purification steps, such as extensive distillation or chromatography, which are often the most costly parts of fine chemical production. By streamlining the process flow, manufacturers can achieve substantial cost savings, making the final cyclohexanol products more price-competitive in the global market.
- Enhanced Supply Chain Reliability: Relying on biomass-derived phenolics as feedstocks diversifies the raw material base away from fluctuating petrochemical markets. Since biomass is a renewable resource with a more stable long-term availability, this shift mitigates the risk of supply disruptions caused by geopolitical tensions or oil price volatility. Furthermore, the robustness of the heterogeneous catalyst ensures consistent performance over extended periods, reducing the frequency of catalyst replacement and maintenance downtime, which is critical for maintaining continuous production schedules.
- Scalability and Environmental Compliance: The simplicity of the catalyst preparation and the use of water make this process inherently scalable from laboratory to industrial levels without significant engineering hurdles. The green nature of the process aligns perfectly with increasingly stringent environmental regulations, reducing the risk of fines and facilitating easier permitting for new production facilities. This environmental compliance not only protects the company's reputation but also opens up opportunities in markets that prioritize sustainably sourced ingredients, adding a premium value proposition to the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What is the optimal solvent system for this hydrogenation process?
A: According to patent CN107089898B, water is identified as the optimal solvent, providing the highest conversion rates compared to organic solvents like methanol or ethyl acetate, while also offering significant environmental and cost benefits.
Q: Can this method be applied to substrates other than guaiacol?
A: Yes, the technology demonstrates broad substrate scope, successfully converting phenol, p-cresol, hydroquinone, and various substituted phenols into their corresponding cyclohexanol derivatives with high selectivity.
Q: What are the typical reaction conditions required?
A: The process typically operates at moderate temperatures ranging from 40°C to 200°C and pressures between 0.1 MPa and 5 MPa, depending on the specific substrate reactivity and desired throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclohexanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in patent CN107089898B for the production of high-value cyclohexanol intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch, meeting the exacting demands of the global pharmaceutical and fine chemical industries.
We invite you to collaborate with us to leverage this green hydrogenation technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-purity cyclohexanol compounds for your business.
