Scalable Production of Venlafaxine Intermediates via Mild Borohydride Reduction Technology
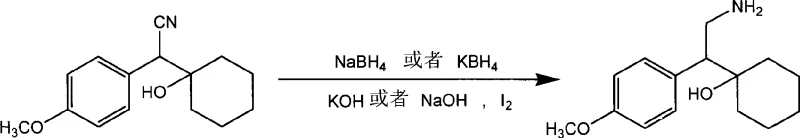
The pharmaceutical industry continuously seeks robust and economically viable synthetic routes for key antidepressant intermediates, and Patent CN101503365A presents a significant technological breakthrough in the preparation of Venlafaxine intermediates. This patent discloses a highly efficient method for synthesizing 1-[2-amino-1-(4-methoxyphenyl)ethyl]cyclohexanol, a critical precursor in the manufacturing of Venlafaxine hydrochloride, by utilizing a novel reduction system involving sodium borohydride or potassium borohydride catalyzed by elemental iodine. Unlike traditional approaches that rely on hazardous or expensive reagents, this invention offers a pathway characterized by exceptionally mild reaction conditions, simplified post-treatment procedures, and superior product purity. For global procurement teams and R&D directors seeking a reliable pharmaceutical intermediates supplier, this technology represents a paradigm shift towards safer and more cost-effective manufacturing protocols that align with modern green chemistry principles and stringent quality standards required for active pharmaceutical ingredient production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the reduction of the nitrile group in Venlafaxine precursors has been plagued by significant technical and economic challenges that hinder efficient commercial scale-up of complex pharmaceutical intermediates. Conventional methods frequently employ dangerous and pyrophoric catalysts such as Raney Nickel, which poses severe safety risks regarding storage, handling, and potential fire hazards within a manufacturing facility. Alternative routes utilize expensive reducing agents like red aluminum or precious metal catalysts including palladium and platinum compounds, which drastically inflate the raw material costs and introduce supply chain vulnerabilities associated with fluctuating precious metal markets. Furthermore, methods employing lithium aluminum hydride, while chemically effective, generate substantial quantities of aluminum hydroxide flocculent during the hydrolysis step, creating a gelatinous mass that makes filtration and phase separation incredibly difficult, slow, and labor-intensive, often resulting in poor recovery rates and yields that stagnate around 60% to 70%.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach detailed in the patent utilizes an in-situ generated active reducing species derived from the interaction of alkali metal borohydrides and elemental iodine under basic conditions. This system operates effectively at mild temperatures ranging from -10°C to 25°C, eliminating the need for cryogenic conditions or high-pressure hydrogenation equipment. The reaction proceeds with remarkable efficiency, converting the nitrile substrate directly to the primary amine with minimal side reactions, thereby ensuring high product purity without the need for complex chromatographic purification. By avoiding the use of transition metals and difficult-to-handle hydrides, this method streamlines the entire production workflow, offering a clear pathway for cost reduction in API manufacturing while simultaneously enhancing the safety profile of the chemical process for operators and the environment alike.
Mechanistic Insights into Iodine-Activated Borohydride Reduction
The core innovation of this synthesis lies in the mechanistic activation of the borohydride anion by elemental iodine, which generates a highly reactive borane-like species capable of selectively reducing the nitrile functionality to a primary amine. In the presence of a strong base such as sodium hydroxide or potassium hydroxide, the iodine reacts with the borohydride to form intermediate species that possess higher electrophilicity than standard borohydride ions, facilitating the nucleophilic attack on the cyano carbon. This activation allows the reduction to proceed rapidly even at ambient or slightly sub-ambient temperatures, preventing the thermal degradation of sensitive functional groups that might occur under harsher reducing conditions. The reaction mechanism ensures that the cyclohexanol moiety remains intact while the nitrile group is fully reduced, demonstrating excellent chemoselectivity that is crucial for maintaining the structural integrity of the complex molecular scaffold required for downstream Venlafaxine synthesis.
Furthermore, the impurity control mechanism inherent in this process is superior to metal-catalyzed hydrogenations which often suffer from over-reduction or dehalogenation issues. The homogeneous nature of the initial reaction mixture, followed by the controlled generation of the active reducing agent, minimizes the formation of oligomeric by-products or incomplete reduction intermediates that typically contaminate the crude product. The subsequent workup involves a straightforward acid-base extraction sequence where the amine product is easily separated from boron and iodine by-products, which remain in the aqueous phase or are removed during solvent evaporation. This results in a crude product with high HPLC purity, often exceeding 94%, which significantly reduces the burden on downstream purification units and ensures that the final intermediate meets the rigorous specifications demanded by regulatory bodies for high-purity pharmaceutical intermediates intended for human consumption.
How to Synthesize 1-[2-amino-1-(4-methoxyphenyl)ethyl]cyclohexanol Efficiently
The synthesis protocol outlined in the patent provides a detailed and reproducible framework for laboratory and pilot-scale production, emphasizing precise temperature control and reagent addition rates to maximize yield and safety. The process begins with the preparation of the activated reducing system in an anhydrous solvent like tetrahydrofuran, followed by the careful addition of the nitrile substrate to maintain reaction exotherms within safe limits. Operators must adhere to strict stoichiometric ratios of base, borohydride, and iodine to ensure complete conversion while minimizing reagent waste. The detailed standardized synthetic steps below provide a comprehensive guide for implementing this technology in a GMP-compliant environment, ensuring consistent batch-to-batch quality and operational reliability.
- Prepare the activated reducing system by mixing sodium hydroxide or potassium hydroxide with sodium borohydride or potassium borohydride in an anhydrous solvent such as tetrahydrofuran, followed by the controlled dropwise addition of an iodine solution at low temperatures between -10°C and 0°C.
- Introduce the substrate 1-cyano-[(4-methoxyphenyl)methyl]cyclohexanol into the reaction system while maintaining the temperature between -10°C and 25°C, allowing the reduction to proceed for 1 to 10 hours followed by a reflux period of 0.5 to 2 hours to ensure complete conversion.
- Perform the post-reaction workup by removing the solvent under reduced pressure, quenching with hydrochloric acid, adjusting the pH to 8-9 with sodium carbonate, and extracting the target amine product using ethyl acetate to obtain high-purity material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this borohydride-iodine reduction technology translates into tangible strategic advantages that extend far beyond simple chemical yield improvements. By shifting away from precious metal catalysts and hazardous pyrophoric materials, manufacturers can significantly de-risk their supply chains against volatile commodity pricing and regulatory restrictions on heavy metal residues. The simplified workup procedure eliminates the need for specialized filtration equipment required to handle aluminum sludge, thereby reducing capital expenditure on processing hardware and lowering the overall operational overhead associated with waste management and disposal. This process optimization leads to substantial cost savings and enhances the overall agility of the manufacturing operation, allowing for faster response times to market demands.
- Cost Reduction in Manufacturing: The elimination of expensive palladium, platinum, or red aluminum reagents results in a drastic reduction in direct material costs, while the high molar yield exceeding 90% minimizes the loss of valuable starting materials. Furthermore, the avoidance of complex filtration steps and the use of common solvents like THF or glyme reduce utility consumption and labor hours, contributing to a leaner and more economically efficient production model that maximizes profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sodium borohydride, potassium hydroxide, and iodine are commodity chemicals with stable and abundant global supply chains, unlike specialty catalysts that may face sourcing bottlenecks or long lead times. This reliance on readily available raw materials ensures continuous production capability and reduces the risk of shutdowns due to material shortages, providing a robust foundation for long-term supply agreements and reducing lead time for high-purity pharmaceutical intermediates required by downstream API manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of heavy metals make this process inherently easier to scale from kilogram to multi-ton production without requiring extensive re-engineering of safety systems. The reduced generation of hazardous solid waste, particularly aluminum sludge, simplifies environmental compliance and wastewater treatment, aligning the manufacturing process with increasingly stringent global environmental regulations and sustainability goals pursued by major pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route, derived directly from the comparative data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational benefits and technical feasibility for stakeholders evaluating this technology for integration into their existing manufacturing portfolios. Understanding these nuances is critical for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of this borohydride-iodine method over traditional lithium aluminum hydride reductions?
A: The borohydride-iodine method eliminates the formation of gelatinous aluminum hydroxide sludge, which makes filtration and separation extremely difficult in lithium aluminum hydride processes. Additionally, it operates under much milder conditions and utilizes safer, more cost-effective reagents compared to the hazardous and moisture-sensitive nature of lithium aluminum hydride.
Q: What yield improvements can be expected with this novel synthetic route?
A: While conventional methods often struggle with yields ranging between 60% and 70%, this patented process consistently achieves molar yields exceeding 90%, significantly reducing raw material waste and improving overall process efficiency for commercial manufacturing.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial suitability, featuring mild reaction temperatures ranging from -10°C to 25°C and simple post-treatment steps that avoid complex purification hurdles, making it highly scalable for ton-level production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Venlafaxine Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our facilities are equipped with state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Venlafaxine intermediate we produce meets the highest international standards for safety and efficacy required by top-tier pharmaceutical partners.
We invite you to collaborate with us to leverage this innovative borohydride-iodine reduction technology for your supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this process can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
