Advanced Fe3O4-Catalyzed Decarboxylative Cross-Ketonization for High-Purity Photoinitiator Intermediates
The global demand for high-performance photoinitiators, particularly alpha-hydroxy aryl ketones like 1173 and 184, continues to surge across the UV curing, adhesive, and coating industries. At the heart of this supply chain lies the critical intermediate: aryl alkyl ketone. Traditionally, the production of these key building blocks has been plagued by severe environmental constraints and complex purification hurdles. However, a groundbreaking technological shift is emerging, anchored by the innovative methodology detailed in Chinese Patent CN107778155B. This patent introduces a highly efficient, environmentally benign preparation method for (substituted) phenyl alkyl ketones that fundamentally redefines the manufacturing landscape. By leveraging a catalytic decarboxylation strategy using nano-sized ferroferric oxide (Fe3O4), this process eliminates the need for corrosive Lewis acids and hazardous halogenated reagents. For R&D directors and procurement strategists alike, this represents a pivotal opportunity to secure a reliable photoinitiator intermediate supplier capable of delivering superior quality while adhering to increasingly strict global environmental regulations. The technology not only simplifies the synthetic route but also integrates an in-situ product separation mechanism that dramatically enhances throughput and reduces energy consumption per unit of output.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
For decades, the industrial standard for synthesizing aryl alkyl ketones has relied heavily on the Friedel-Crafts acylation reaction. While historically significant for its low raw material costs, this legacy technology imposes substantial hidden burdens on modern chemical manufacturing. The process typically necessitates the use of more than equivalent amounts of dangerous chemicals such as aluminum trichloride (AlCl3) and chlorine gas. These reagents are not only hazardous to handle, requiring specialized containment and safety protocols, but they also generate prodigious quantities of waste acid and waste alkali during the quenching and workup phases. The disposal of this hazardous waste stream creates immense environmental pressure and escalating compliance costs for manufacturers. Furthermore, the reaction selectivity in Friedel-Crafts processes can be problematic, often leading to poly-acylation byproducts or isomeric mixtures that complicate downstream purification. The resulting impurity profiles often require extensive washing and neutralization steps, which prolong the production cycle and increase the overall carbon footprint of the facility. Consequently, finding a sustainable alternative that maintains economic viability while eliminating these toxic liabilities has become a primary objective for forward-thinking chemical enterprises.
The Novel Approach
In stark contrast to the corrosive nature of traditional acylation, the novel approach described in CN107778155B utilizes a catalytic decarboxylative cross-ketonization between aromatic and aliphatic carboxylic acid salts. This method transforms simple, readily available sodium salts into valuable ketone intermediates with remarkable efficiency. The core innovation lies in the use of Fe3O4 nanoparticles as a robust, magnetically separable catalyst, coupled with a high-boiling solvent system such as Dowtherm or minus oil. 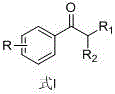 As illustrated in the general structure, the versatility of this method allows for various substituents (R, R1, R2) to be incorporated, making it applicable to a wide range of photoinitiator precursors. The reaction proceeds cleanly, with water and carbon dioxide serving as the primary byproducts, thereby virtually eliminating the generation of hazardous saline waste. Moreover, the process incorporates a dynamic separation technique where the product is distilled out of the reaction system as it forms. This continuous removal drives the equilibrium forward, significantly boosting conversion rates and simplifying the isolation of the final product. This paradigm shift offers a clear pathway for cost reduction in fine chemical manufacturing by removing the expensive waste treatment infrastructure associated with legacy Friedel-Crafts processes.
As illustrated in the general structure, the versatility of this method allows for various substituents (R, R1, R2) to be incorporated, making it applicable to a wide range of photoinitiator precursors. The reaction proceeds cleanly, with water and carbon dioxide serving as the primary byproducts, thereby virtually eliminating the generation of hazardous saline waste. Moreover, the process incorporates a dynamic separation technique where the product is distilled out of the reaction system as it forms. This continuous removal drives the equilibrium forward, significantly boosting conversion rates and simplifying the isolation of the final product. This paradigm shift offers a clear pathway for cost reduction in fine chemical manufacturing by removing the expensive waste treatment infrastructure associated with legacy Friedel-Crafts processes.
Mechanistic Insights into Fe3O4-Catalyzed Decarboxylative Cross-Ketonization
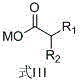
The efficacy of this synthesis hinges on the unique properties of the ferroferric oxide (Fe3O4) catalyst, specifically engineered with a particle size of less than or equal to 100 nm. At this nanoscale dimension, the catalyst exhibits a vastly increased surface area-to-volume ratio, ensuring maximum contact with the reactant carboxylate salts. The reaction mechanism involves the activation of the carboxylate groups on both the aromatic ring (Formula II) and the aliphatic chain (Formula III). 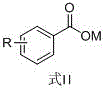
 Under the elevated thermal conditions of 200-300°C, the catalyst facilitates the decarboxylation of these salts, generating reactive acyl and alkyl radical species or organometallic intermediates on the catalyst surface. These species then undergo coupling to form the desired carbon-carbon bond of the ketone. The use of alkali metal salts (where M is an alkali metal like sodium) ensures that the reactants are stable and easy to handle prior to the reaction, avoiding the moisture sensitivity associated with acid chlorides used in Grignard-type approaches. The high boiling point of the solvent (200-410°C) is critical, as it provides the necessary thermal energy to overcome the activation barrier for decarboxylation while remaining liquid to facilitate mass transfer. This mechanistic pathway avoids the formation of stable complexes that often trap catalysts in other systems, allowing the Fe3O4 to remain active over multiple cycles.
Under the elevated thermal conditions of 200-300°C, the catalyst facilitates the decarboxylation of these salts, generating reactive acyl and alkyl radical species or organometallic intermediates on the catalyst surface. These species then undergo coupling to form the desired carbon-carbon bond of the ketone. The use of alkali metal salts (where M is an alkali metal like sodium) ensures that the reactants are stable and easy to handle prior to the reaction, avoiding the moisture sensitivity associated with acid chlorides used in Grignard-type approaches. The high boiling point of the solvent (200-410°C) is critical, as it provides the necessary thermal energy to overcome the activation barrier for decarboxylation while remaining liquid to facilitate mass transfer. This mechanistic pathway avoids the formation of stable complexes that often trap catalysts in other systems, allowing the Fe3O4 to remain active over multiple cycles.
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic aromatic substitution. Because the reaction relies on the coupling of two distinct carboxylate fragments rather than the attack of an aromatic ring by a strong electrophile, the regioselectivity is inherently governed by the structure of the starting salts. There is no risk of poly-substitution on the aromatic ring, a common issue in Friedel-Crafts chemistry that leads to difficult-to-remove tars and heavy ends. The primary byproducts are gaseous CO2 and water, which are easily vented or separated, leaving the organic phase relatively clean. Experimental data from the patent indicates that the crude distillate often contains over 90% of the target ketone by GC analysis (e.g., 93.2% for isobutyrophenone and 95.7% for n-butyrylbenzene). This high initial purity significantly reduces the load on the final purification step, which typically involves a straightforward reduced pressure distillation to achieve >99% purity. For R&D teams, this translates to a robust process window where minor fluctuations in reaction time or temperature do not catastrophically impact the impurity profile, ensuring consistent batch-to-batch quality essential for pharmaceutical and electronic grade applications.
How to Synthesize Aryl Alkyl Ketone Efficiently
The operational simplicity of this patented method makes it highly attractive for rapid technology transfer and scale-up. The process begins with the uniform mixing of the aromatic carboxylate (Formula II) and the aliphatic carboxylate (Formula III) in a molar ratio ranging from 1.5:1 to 3.0:1, alongside the nano-Fe3O4 catalyst and the high-boiling solvent. This mixture is heated under a nitrogen atmosphere to prevent oxidative degradation of the organic components. A key feature of the protocol is the transition from a reflux setup to a distillation setup after an initial reaction period of 2 to 5 hours. This allows the system to first reach conversion and then continuously strip the product from the reaction matrix. Detailed standardized operating procedures, including specific heating ramps and vacuum levels for different derivatives, are critical for maximizing yield.
- Uniformly mix sodium benzoate (Formula II) and sodium aliphatic carboxylate (Formula III) with Fe3O4 catalyst (particle size ≤100nm) and high-boiling solvent under nitrogen.
- Heat the mixture to 200-300°C and maintain reaction for 2-5 hours to initiate decarboxylative cross-ketonization.
- Switch apparatus to distillation mode to continuously separate the product from the reaction system while maintaining temperature, allowing for catalyst and solvent recycling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Fe3O4-catalyzed route offers transformative benefits that extend far beyond simple yield improvements. The elimination of hazardous reagents like aluminum trichloride and chlorine gas fundamentally alters the risk profile of the manufacturing site, reducing insurance premiums and regulatory scrutiny. Furthermore, the ability to recycle both the catalyst and the solvent creates a closed-loop system that minimizes raw material waste and lowers the variable cost of production significantly. The simplified workup, which avoids extensive aqueous washing and neutralization steps, reduces the consumption of process water and the volume of wastewater requiring treatment. These factors combine to create a leaner, more agile manufacturing process that is resilient to supply chain disruptions affecting hazardous chemical logistics.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling when analyzed through the lens of total cost of ownership. By replacing stoichiometric amounts of expensive and hazardous Lewis acids with a catalytic amount of inexpensive iron oxide, the direct material cost is drastically reduced. More importantly, the downstream savings are substantial; the absence of corrosive waste streams eliminates the need for costly neutralization agents and hazardous waste disposal fees. The process also features high atom economy relative to traditional methods, as the only lost mass corresponds to the carboxyl groups leaving as CO2. Additionally, the catalyst and solvent recycling capability means that these high-value inputs do not need to be replenished for every batch, leading to cumulative savings that compound over the lifecycle of the production campaign. This structural efficiency ensures that the final product remains cost-competitive even in volatile raw material markets.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complex logistics of handling dangerous goods. Traditional methods relying on acid chlorides or chlorine gas are susceptible to transportation restrictions and storage limitations. In contrast, the carboxylate salts used in this novel method are stable, non-hazardous solids that are easy to store and transport in bulk. This stability simplifies inventory management and reduces the risk of supply interruptions due to regulatory changes in hazardous material transport. Furthermore, the robustness of the reaction conditions (200-300°C) allows for flexible scheduling and easier scale-up without the need for exotic cryogenic cooling or ultra-low temperature equipment. This operational flexibility enables manufacturers to respond more rapidly to spikes in demand, ensuring that downstream customers in the UV curing and coatings sectors receive their materials on time.
- Scalability and Environmental Compliance: As global environmental regulations tighten, the scalability of a chemical process is inextricably linked to its environmental footprint. This decarboxylation method is inherently green, producing only water and carbon dioxide as byproducts, which aligns perfectly with modern sustainability goals and ESG (Environmental, Social, and Governance) mandates. The lack of saline wastewater simplifies the permitting process for new production lines and reduces the burden on onsite effluent treatment plants. From a scale-up perspective, the use of high-boiling solvents and distillation-based product separation is a unit operation well-understood in large-scale chemical engineering. The ability to run the reaction in a semi-continuous mode, where product is distilled off and fresh reactants are added to the residual catalyst/solvent pot, allows for high throughput in existing reactor infrastructure without the need for massive capital investment in new containment systems.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new synthesis route is essential for stakeholders evaluating its potential for integration into existing supply chains. The following questions address common inquiries regarding catalyst performance, product quality, and process safety, derived directly from the technical disclosures in the patent literature. These insights are intended to provide clarity on how this method compares to incumbent technologies and what operational adjustments might be required for successful implementation.
Q: How does the Fe3O4 catalyst improve the environmental profile compared to traditional Friedel-Crafts acylation?
A: Unlike traditional Friedel-Crafts reactions that utilize stoichiometric amounts of hazardous aluminum trichloride and generate massive quantities of waste acid and alkali, this patented method employs recyclable magnetic Fe3O4 nanoparticles. The only byproducts are water and carbon dioxide, drastically reducing the environmental burden and waste treatment costs associated with hazardous chemical disposal.
Q: What is the expected purity and yield for the synthesized aryl alkyl ketones?
A: According to the experimental data in patent CN107778155B, the process achieves high efficiency with GC content often exceeding 90% in the crude distillate (e.g., 93.2% for isobutyrophenone, 95.7% for n-butyrylbenzene). Subsequent purification via reduced pressure distillation yields products with purity greater than 99%, meeting stringent specifications for photoinitiator applications.
Q: Is the catalyst and solvent system suitable for continuous industrial recycling?
A: Yes, the process is specifically designed for industrial continuity. The high-boiling solvent (such as Dowtherm or minus oil) and the solid Fe3O4 catalyst remain in the reaction kettle after the product is distilled off. They can be directly reused for the next batch by simply adding fresh carboxylate salts, significantly lowering raw material consumption and operational downtime.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Alkyl Ketone Supplier
The transition to greener, more efficient synthetic routes requires a partner with deep technical expertise and a proven track record in process development. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and distillation requirements of the Fe3O4-catalyzed decarboxylation process, ensuring that the high purity specifications (>99%) demonstrated in the lab are consistently met at the commercial scale. With our rigorous QC labs and commitment to continuous improvement, we guarantee that every batch of aryl alkyl ketone meets the stringent demands of the photoinitiator and fine chemical industries.
We invite you to explore how this advanced technology can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic sourcing goals. Let us partner with you to build a more sustainable and efficient future for high-performance chemical intermediates.
