Advanced Iron-Catalyzed Oxidation Technology for Scalable Ketone Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and cost-effective pathways for synthesizing critical structural motifs, particularly ketones, which serve as pivotal intermediates in the production of active pharmaceutical ingredients (APIs) and agrochemicals. A significant technological breakthrough in this domain is detailed in patent CN106748690B, which discloses a novel method for synthesizing ketones through the iron-catalyzed oxidation of alkenes. This innovation represents a paradigm shift from traditional precious metal-dependent processes to a more economical and environmentally benign approach utilizing earth-abundant iron catalysts. By leveraging air or molecular oxygen as the terminal oxidant in the presence of a hydrosilane promoter, this technology achieves exceptional conversion rates and selectivity under mild conditions. The method addresses long-standing challenges in organic synthesis, offering a robust platform for the preparation of high-purity ketone intermediates that are essential for downstream drug development and material science applications.
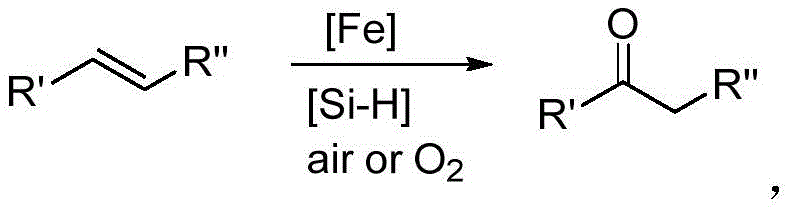
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial standard for converting alkenes to ketones has been the Wacker oxidation process, which relies heavily on palladium catalysts and copper salts as co-oxidants. While effective, this traditional methodology presents severe economic and environmental drawbacks that hinder its sustainability in modern green chemistry initiatives. The reliance on palladium, a precious metal with volatile market pricing and limited global supply, introduces significant cost instability and supply chain risks for large-scale manufacturers. Furthermore, the necessity of copper co-catalysts generates substantial amounts of heavy metal waste, necessitating complex and expensive purification steps to meet stringent regulatory limits on residual metals in pharmaceutical products. Alternative methods, such as those utilizing benzoquinone as an oxidant, often suffer from poor atom economy and generate stoichiometric amounts of organic waste, complicating waste management and increasing the overall carbon footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy technologies, the method described in patent CN106748690B introduces a highly efficient iron-catalyzed system that fundamentally alters the economic and operational landscape of ketone synthesis. By substituting expensive palladium with inexpensive and non-toxic iron salts, this approach drastically reduces raw material costs while eliminating the burden of heavy metal contamination. The utilization of air or oxygen as the sole oxidant ensures that the only byproduct is water or silanol derivatives, aligning perfectly with green chemistry principles. The inclusion of hydrosilanes as promoters activates the iron catalyst in situ, enabling the reaction to proceed smoothly at temperatures ranging from 20°C to 180°C. This versatility allows for the processing of a vast array of substrates, from simple styrenes to complex bioactive molecules, achieving isolated yields as high as 98% without the need for harsh reaction conditions or exotic reagents.
Mechanistic Insights into Fe-Catalyzed Alkene Oxidation
The success of this transformation lies in the unique synergistic interaction between the iron catalyst and the hydrosilane promoter, which facilitates the activation of molecular oxygen under mild conditions. Mechanistically, the hydrosilane serves not merely as a reducing agent but as a crucial activator that generates highly reactive iron-hydride or iron-silyl species in situ. These active catalytic species are capable of coordinating with the alkene substrate and activating the oxygen molecule, thereby lowering the energy barrier for the oxidation process. This mechanism allows the reaction to bypass the high-energy intermediates typically associated with uncatalyzed auto-oxidation, resulting in superior regioselectivity and chemoselectivity. The ability to tune the electronic and steric properties of the iron catalyst by selecting specific ligands or salt forms, such as ferrous chloride, iron acetylacetonate, or iron trifluoromethanesulfonate, provides chemists with precise control over the reaction outcome, ensuring consistent quality across different batches.
Furthermore, the robustness of this catalytic system is evidenced by its remarkable tolerance towards diverse functional groups, a critical factor for the synthesis of complex pharmaceutical intermediates. The reaction conditions are sufficiently mild to preserve sensitive moieties such as halides, esters, nitro groups, and free hydroxyls, which might otherwise decompose under the harsh acidic or basic conditions of traditional oxidation methods. Experimental data from the patent demonstrates successful conversion of substrates containing bromine, chlorine, and iodine atoms, as well as heterocyclic rings like furans, thiophenes, and pyridines, without compromising the integrity of these functional handles. This broad substrate scope extends to biologically relevant molecules, including steroid derivatives and carbohydrate-based alkenes, proving that the method is applicable to the late-stage functionalization of advanced intermediates. Such versatility minimizes the need for protecting group strategies, thereby streamlining synthetic routes and reducing the total number of steps required to reach the final target molecule.
How to Synthesize Ketone Intermediates Efficiently
Implementing this iron-catalyzed oxidation protocol in a laboratory or pilot plant setting involves a straightforward procedure that balances safety with high efficiency. The process typically begins with the dissolution of the alkene substrate in a compatible organic solvent, such as ethanol, acetonitrile, or tert-butanol, followed by the addition of the chosen iron catalyst and hydrosilane promoter. The reaction mixture is then subjected to an atmosphere of air or oxygen, often at slightly elevated pressures to enhance reaction kinetics, and heated to the optimal temperature determined by the specific substrate's reactivity. After the reaction reaches completion, indicated by TLC or HPLC analysis, a simple workup involving neutralization with ammonia water and extraction with organic solvents yields the crude ketone, which can be further purified by column chromatography or crystallization. For detailed operational parameters and specific reagent ratios tailored to your specific substrate, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining the alkene substrate, an iron catalyst (such as ferrous chloride or iron acetylacetonate), and a hydrosilane promoter in a suitable organic solvent or aqueous solution.
- Introduce air or oxygen into the reaction vessel, maintaining a pressure between 1 to 10 atmospheres depending on the specific substrate requirements and desired reaction rate.
- Heat the mixture to a temperature ranging from 20°C to 180°C and stir for 0.25 to 60 hours, followed by workup with ammonia water and extraction to isolate the target ketone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, adopting this iron-catalyzed technology offers transformative benefits that extend far beyond simple reaction yield improvements. The shift from precious metal catalysts to base metal iron fundamentally reshapes the cost structure of ketone manufacturing, removing exposure to the volatile pricing of palladium and rhodium markets. Additionally, the use of air or oxygen as a reagent eliminates the need for purchasing and storing hazardous stoichiometric oxidants, simplifying logistics and reducing inventory costs. The simplified purification process, driven by the absence of heavy metal residues, translates directly into reduced processing time and lower consumption of purification media, such as scavenger resins or specialized filtration equipment. These factors collectively contribute to a more resilient and cost-efficient supply chain capable of responding rapidly to market demands.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with iron salts results in a drastic reduction in raw material expenses, as iron is orders of magnitude cheaper and more abundant globally. Moreover, the elimination of copper co-catalysts and benzoquinone oxidants removes significant waste disposal costs associated with heavy metal treatment. The simplified downstream processing, which avoids complex heavy metal scavenging steps, further lowers operational expenditures by reducing solvent usage and labor hours. This comprehensive cost optimization makes the production of high-value ketone intermediates economically viable even for price-sensitive generic drug markets.
- Enhanced Supply Chain Reliability: Relying on earth-abundant iron and commodity hydrosilanes mitigates the risk of supply disruptions often associated with precious metals sourced from geopolitically unstable regions. The use of air or oxygen as the oxidant ensures that the primary reactant is virtually unlimited and available on-site, removing dependency on external suppliers for critical oxidizing agents. This self-sufficiency enhances production continuity and allows manufacturers to maintain consistent output schedules regardless of external market fluctuations. The robustness of the catalyst system also means that variations in raw material quality have minimal impact on the final product, ensuring reliable batch-to-batch consistency.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic heavy metals make this process inherently safer and easier to scale from gram to multi-ton quantities without requiring specialized containment infrastructure. The generation of minimal waste, primarily consisting of benign silanol byproducts, simplifies compliance with increasingly stringent environmental regulations regarding effluent discharge. This eco-friendly profile not only reduces the regulatory burden but also enhances the corporate sustainability image, which is becoming a key differentiator in B2B partnerships with major pharmaceutical companies committed to green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this iron-catalyzed oxidation technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement officers assessing long-term supply viability. The answers reflect the consensus of current chemical literature and the specific findings of the patented invention.
Q: What are the primary advantages of this iron-catalyzed method over traditional Wacker oxidation?
A: Unlike traditional Wacker oxidation which relies on expensive palladium catalysts and copper co-oxidants, this method utilizes abundant and low-cost iron catalysts. It eliminates the need for toxic heavy metals, significantly reducing purification costs and environmental waste while maintaining high selectivity and yields up to 98%.
Q: Can this methodology tolerate complex functional groups found in pharmaceutical intermediates?
A: Yes, the process demonstrates excellent functional group compatibility. It successfully oxidizes alkenes containing sensitive groups such as halogens, esters, nitro groups, hydroxyls, and even complex structures like steroids and sugars without requiring extensive protecting group strategies.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The use of air or oxygen as the terminal oxidant and commercially available hydrosilanes makes the process economically viable for scale-up. The mild reaction conditions and simple workup procedures facilitate safe and efficient manufacturing from kilogram to multi-ton scales.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketone Intermediates Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has extensively evaluated the iron-catalyzed oxidation process described in CN106748690B and confirmed its potential for delivering high-purity ketone intermediates with superior cost-efficiency. As a leading CDMO partner, we possess the infrastructure and expertise to translate this laboratory-scale innovation into commercial reality. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our rigorous QC labs and stringent purity specifications guarantee that every batch of ketone intermediate meets the highest industry standards, free from the heavy metal contaminants that plague traditional synthesis routes.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this iron-catalyzed route for your specific molecule. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and preliminary quotations. Let us help you optimize your supply chain and reduce manufacturing costs while contributing to a more sustainable future for the chemical industry.
