Advanced Metal-Free Catalytic Oxidation for High-Purity Pharmaceutical Intermediates
Advanced Metal-Free Catalytic Oxidation for High-Purity Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic methodologies that balance efficiency with regulatory compliance. Patent CN107011133B introduces a groundbreaking approach for the direct oxidation of benzylic C-H bonds into ketones, utilizing a synergistic catalytic system composed of radical initiators and nitrite esters. This technology represents a significant paradigm shift from traditional stoichiometric oxidants and transition metal catalysts to a greener, non-metallic protocol driven by molecular oxygen. For R&D directors and process chemists, this method offers a compelling solution to the persistent challenge of metal residue removal in drug substance manufacturing. By leveraging a radical-mediated mechanism under mild thermal conditions, typically between 80°C and 90°C, this process achieves high atom economy while maintaining excellent functional group tolerance across a diverse range of aromatic substrates.
From a supply chain perspective, the adoption of this technology aligns perfectly with the industry's move towards sustainable manufacturing practices. The use of oxygen as the terminal oxidant not only reduces the environmental footprint by eliminating heavy metal waste streams but also simplifies the purification workflow. The reaction mixture can be directly concentrated and purified via standard column chromatography using common eluent systems like petroleum ether and ethyl acetate. This operational simplicity translates to reduced processing time and lower solvent consumption, making it an attractive candidate for the commercial scale-up of complex pharmaceutical intermediates. As a reliable pharmaceutical intermediate supplier, understanding such technological advancements allows us to offer clients more cost-effective and compliant synthetic routes for key building blocks like acetophenones and benzophenones.
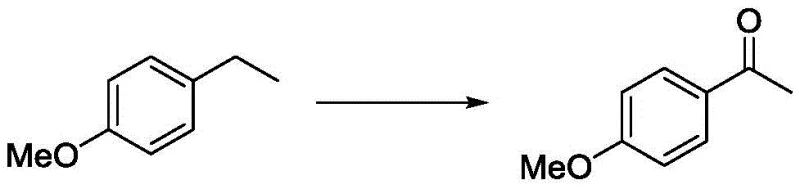
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the oxidation of aryl side chains to ketones has relied heavily on harsh stoichiometric oxidants such as potassium permanganate or potassium dichromate. These classical methods suffer from severe drawbacks, including poor selectivity, especially when multiple oxidizable sites are present on the aromatic ring, leading to complex mixtures of over-oxidized byproducts that are difficult to separate. Furthermore, the environmental and safety liabilities associated with chromium and manganese waste disposal have made these processes increasingly untenable for modern GMP manufacturing. While transition metal catalysts like Rhodium, Copper, and Iron complexes were developed to improve selectivity, they introduce a new set of challenges regarding cost and purity. The high price of precious metal ligands combined with the stringent regulatory limits on residual metals in APIs necessitates expensive purification steps, such as scavenging or recrystallization, which erode profit margins and extend lead times.
The Novel Approach
The methodology described in CN107011133B overcomes these hurdles by employing a metal-free catalytic system that utilizes N-hydroxyphthalimide (NHPI) or similar radical initiators in conjunction with alkyl nitrites. This dual-catalyst system effectively activates molecular oxygen to generate radical species capable of abstracting benzylic hydrogen atoms with high precision. Unlike peroxide-based oxidations which can be hazardous and uncontrollable, this oxygen-driven process operates safely at atmospheric pressure or with simple oxygen balloons. The ability to tune the reaction outcome by adjusting catalyst loading is particularly noteworthy; for instance, electron-rich substrates require milder conditions compared to electron-deficient ones, allowing for a customizable approach to synthesis. This flexibility ensures that even sensitive functional groups remain intact, providing a cleaner reaction profile and higher isolated yields compared to traditional metal-catalyzed counterparts.
Mechanistic Insights into NHPI-Nitrite Synergistic Catalysis
The core of this innovation lies in the synergistic interaction between the radical initiator and the nitrite ester under an oxygen atmosphere. The mechanism initiates with the generation of phthalimide-N-oxyl (PINO) radicals from NHPI, which are highly effective at abstracting hydrogen atoms from the benzylic position due to their favorable bond dissociation energy dynamics. The presence of the nitrite ester facilitates the regeneration of the active radical species and helps propagate the chain reaction by interacting with the oxygen molecules. This creates a continuous catalytic cycle where molecular oxygen serves as the hydrogen acceptor, ultimately forming water as the benign byproduct. For R&D teams, understanding this radical pathway is crucial for optimizing reaction parameters such as temperature and solvent choice. The patent specifies solvents like acetonitrile or ethyl acetate, which stabilize the radical intermediates without interfering with the oxidation process, ensuring consistent reproducibility across different batches.
Furthermore, the system exhibits remarkable chemoselectivity, particularly in substrates containing multiple benzylic positions. The kinetic control exerted by the catalyst concentration allows for the selective formation of mono-ketones versus di-ketones. For example, in the oxidation of diethylbenzene derivatives, limiting the catalyst loading favors the mono-oxidized product, while excess catalyst drives the reaction to the di-ketone. This level of control is rarely achieved with non-selective oxidants like permanganate. The absence of metal ions also means there is no risk of metal-catalyzed side reactions such as halogenation or ring oxidation, which preserves the integrity of the aromatic core. This mechanistic clarity provides a solid foundation for scaling the process, as the reaction kinetics are well-defined and manageable within standard reactor configurations.
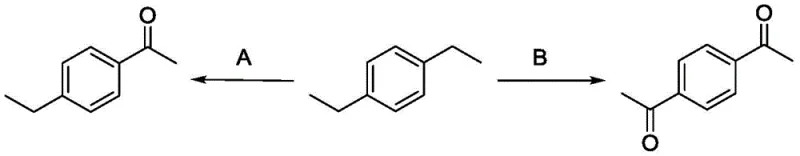
How to Synthesize Benzylic Ketones Efficiently
Implementing this oxidation strategy in a laboratory or pilot plant setting requires adherence to specific operational protocols to maximize yield and safety. The general procedure involves charging a reaction vessel with the radical initiator, such as NHPI, and subjecting it to vacuum drying to eliminate moisture which could quench the radical species. Subsequently, the solvent, nitrite co-catalyst, and the benzylic substrate are introduced under an inert or oxygen atmosphere depending on the specific optimization stage. The mixture is then heated to the prescribed temperature range, typically 80°C to 90°C, and maintained for a duration of 24 to 36 hours to ensure complete conversion. Following the reaction, the workup is straightforward, involving solvent removal and purification, which underscores the practical utility of this method for rapid process development.
- Prepare the reaction vessel by adding the radical initiator (e.g., NHPI) and drying under vacuum to remove moisture.
- Introduce the solvent (e.g., acetonitrile), nitrite ester co-catalyst, and the benzylic substrate under an oxygen atmosphere.
- Heat the mixture to 80-90°C for 24-36 hours, then purify the resulting ketone via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this metal-free oxidation technology offers substantial strategic benefits beyond mere technical performance. The elimination of expensive transition metal catalysts directly impacts the bill of materials, removing the volatility associated with precious metal pricing. Moreover, the simplified downstream processing reduces the demand for specialized metal scavengers and extensive filtration equipment, leading to significant cost reduction in API manufacturing. The use of oxygen as a reagent is perhaps the most impactful factor; it is inexpensive, readily available, and generates no toxic waste, thereby lowering the costs associated with environmental compliance and waste disposal. These factors collectively enhance the overall economic viability of producing high-volume ketone intermediates.
- Cost Reduction in Manufacturing: By replacing stoichiometric metal oxidants and precious metal catalysts with inexpensive organic initiators and oxygen, the direct material costs are drastically lowered. The process avoids the need for costly metal removal steps, which often require specialized resins or multiple recrystallizations, thereby streamlining the production workflow. This efficiency gain allows for more competitive pricing structures for bulk chemical supplies without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reagents required for this synthesis, such as NHPI and alkyl nitrites, are commercially available and stable, reducing the risk of supply disruptions common with specialized metal complexes. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality, ensuring consistent output. This reliability is critical for maintaining continuous production schedules and meeting tight delivery deadlines for downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The use of molecular oxygen and the absence of heavy metals make this process inherently scalable and environmentally friendly. It aligns with green chemistry principles by minimizing hazardous waste generation, which simplifies regulatory approvals and reduces the burden on waste treatment facilities. The ability to scale from gram to kilogram quantities without significant re-optimization ensures that the supply chain can respond flexibly to market demands while maintaining a strong sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this catalytic oxidation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. Understanding these nuances helps stakeholders make informed decisions about adopting this methodology for their specific synthetic needs. We encourage technical teams to review the full experimental section for detailed substrate scope and optimization data.
Q: How does this method prevent metal contamination in drug substances?
A: Unlike traditional methods using Chromium, Manganese, or Transition Metals like Rhodium and Copper, this patent utilizes a purely organic catalytic system comprising N-hydroxyphthalimide (NHPI) and alkyl nitrites. This eliminates the risk of heavy metal residues, which is critical for meeting stringent ICH Q3D guidelines for elemental impurities in active pharmaceutical ingredients.
Q: Can this process selectively produce mono-ketones from di-alkyl benzenes?
A: Yes, the patent demonstrates exceptional selectivity control. By adjusting the molar ratio of the catalyst and oxidant, specifically using 0.5 equivalents of initiator for mono-oxidation and 1.0 equivalent for di-oxidation, manufacturers can precisely target either mono-ketones or di-ketones from substrates like diethylbenzene, reducing downstream separation costs.
Q: What are the safety advantages of using oxygen over peroxides?
A: Conventional non-metal methods often rely on unstable organic peroxides like TBHP, which pose significant thermal hazards and storage risks. This technology employs molecular oxygen or air as the terminal oxidant, which is inherently safer, generates water as the only byproduct, and offers superior atom economy for large-scale industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ketone Intermediates Supplier
At NINGBO INNO PHARMCHEM, we are committed to integrating cutting-edge synthetic technologies like the metal-free benzylic oxidation described in CN107011133B into our manufacturing portfolio. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ketone intermediates meets the highest quality standards required by the global pharmaceutical industry. Our infrastructure is designed to handle complex chemistries safely and efficiently, providing our partners with a secure source of critical building blocks.
We invite you to collaborate with us to leverage these advanced synthetic routes for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how this metal-free technology can optimize your supply chain. Contact us today to discuss how we can support your development goals with high-quality, cost-effective pharmaceutical intermediates.
