Advanced Visible-Light Catalyzed Production of Alpha-Hydroxy Ketones for Commercial Scale-Up
Introduction to Green Oxidative Hydroxylation Technology
The synthesis of alpha-hydroxy ketones, also known as acyloins, represents a critical transformation in the construction of complex organic molecules, serving as indispensable building blocks for numerous biologically active compounds and pharmaceutical agents. As detailed in the groundbreaking patent CN108083999B, a novel green preparation method has been developed that fundamentally shifts the paradigm from hazardous stoichiometric oxidations to a sustainable, catalytic approach driven by visible light. This technology leverages the abundant and inexpensive power of compact fluorescent lamps to activate molecular oxygen from the air, thereby eliminating the need for toxic heavy metal oxidants or dangerous peroxide reagents that have historically plagued this chemical transformation. For R&D directors and process chemists, this innovation offers a pathway to cleaner impurity profiles and simplified downstream processing, while supply chain managers will appreciate the reliance on commodity chemicals like iodine and methanol which ensure robust availability and pricing stability. The method specifically targets the direct hydroxylation of alpha-methylene ketones, a challenging substrate class that often resists conventional oxidation protocols, thus filling a significant gap in synthetic methodology for high-purity pharmaceutical intermediates.
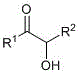
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of alpha-hydroxy ketones has relied heavily on methodologies that are increasingly incompatible with modern green chemistry standards and industrial safety regulations. Traditional routes often employ stoichiometric amounts of hypervalent iodine reagents, which generate substantial quantities of hazardous iodine-containing waste streams that require costly and complex disposal procedures. Alternatively, the use of organic peroxides or tert-butyl hydroperoxide as oxidants introduces significant safety risks due to their thermal instability and potential for explosive decomposition, necessitating specialized equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, many existing catalytic systems depend on expensive transition metals such as palladium or ruthenium, which not only inflate raw material costs but also leave trace metal residues in the final product, creating severe challenges for meeting stringent regulatory limits in pharmaceutical manufacturing. These legacy methods often suffer from limited substrate scope, particularly struggling with alpha-methylene ketones, and typically require harsh reaction conditions that compromise energy efficiency and operational simplicity.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN108083999B introduces a remarkably mild and efficient catalytic system that utilizes visible light irradiation to drive the oxidative hydroxylation reaction under ambient conditions. By employing a catalytic amount of elemental iodine combined with DABCO (1,4-diazabicyclo[2.2.2]octane) in methanol, this process activates molecular oxygen directly from the air, transforming it into a potent yet safe oxidant without generating stoichiometric waste. The reaction proceeds smoothly at room temperature, eliminating the energy costs associated with heating or cooling, and utilizes a 23 W compact fluorescent lamp as a sustainable energy source that is easily scalable in photoreactors. This approach not only achieves high yields across a diverse range of substrates, including those with sensitive functional groups, but also simplifies the workup procedure to a standard silica gel column chromatography, significantly reducing solvent consumption and processing time. The ability to convert simple alpha-methylene ketones directly into valuable secondary alpha-hydroxy ketones with such high atom economy represents a transformative advancement for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Visible-Light Iodine Catalysis
The mechanistic underpinning of this transformation involves a sophisticated interplay between photo-excited iodine species and molecular oxygen, facilitating a radical pathway that selectively functionalizes the alpha-position of the ketone. Upon irradiation with visible light, the iodine catalyst undergoes homolytic cleavage or excitation to generate reactive iodine radicals or excited states capable of abstracting a hydrogen atom from the alpha-methylene position of the ketone substrate. This initial hydrogen atom transfer generates a carbon-centered radical intermediate, which is subsequently trapped by molecular oxygen present in the air atmosphere to form a peroxy radical species. The presence of DABCO plays a crucial dual role in this cycle, acting both as a base to facilitate enolization or proton transfer steps and as a co-catalyst that assists in the regeneration of the active iodine species while decomposing peroxide intermediates into the desired hydroxyl group. This catalytic cycle ensures that only trace amounts of iodine are required to turnover large quantities of substrate, minimizing halogenated impurities and simplifying the purification process. The specificity for alpha-methylene ketones over alpha-methine ketones suggests a kinetic preference driven by the bond dissociation energy of the C-H bonds and the stability of the resulting radical intermediates, providing chemists with a predictable tool for regioselective functionalization.
From an impurity control perspective, this mechanism offers distinct advantages over traditional oxidation methods by avoiding the formation of over-oxidized byproducts such as 1,2-diketones or cleavage products that often arise from harsh chemical oxidants. The mild nature of the visible-light promoted radical process ensures that sensitive functional groups on the aromatic ring, such as halogens, trifluoromethyl groups, or ethers, remain intact throughout the reaction, preserving the structural integrity required for downstream coupling reactions. Furthermore, the absence of transition metals eliminates the risk of metal-catalyzed side reactions or the formation of difficult-to-remove metal complexes, resulting in a cleaner crude reaction mixture. The use of methanol as a solvent not only solubilizes the polar intermediates but also participates in the stabilization of radical species, further enhancing the selectivity towards the desired alpha-hydroxy ketone product. This high level of chemoselectivity is paramount for R&D teams aiming to streamline the synthesis of complex drug candidates where protecting group strategies can add significant cost and time to the overall development timeline.
How to Synthesize Alpha-Hydroxy Ketones Efficiently
The practical implementation of this green synthesis route is designed for ease of operation, requiring standard laboratory glassware and commercially available reagents that are accessible from any major chemical supplier. The protocol involves a straightforward one-pot procedure where the ketone substrate, catalytic iodine, and DABCO are dissolved in methanol, followed by exposure to visible light under an open air atmosphere, removing the need for inert gas lines or high-pressure reactors. Reaction monitoring can be easily performed via TLC or HPLC, with typical completion times ranging from 14 to 30 hours depending on the electronic nature of the substrate substituents. Following the reaction, the mixture is concentrated and purified via silica gel chromatography using a standard petroleum ether and ethyl acetate gradient, yielding the pure alpha-hydroxy ketone as a colorless liquid or solid with high purity. Detailed standardized synthesis steps for specific substrates are provided in the guide below to assist process engineers in replicating these results.
- Sequentially add ketone substrate, catalytic iodine (20 mol%), DABCO (50 mol%), and methanol solvent into a glass reaction vessel.
- Irradiate the mixture with a 23 W compact fluorescent lamp (CFL) while stirring at room temperature in an air atmosphere for 14 to 30 hours.
- Separate the resulting reaction mixture using silica gel column chromatography with a PE/EtOAc mobile phase to isolate pure alpha-hydroxy ketone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this visible-light catalyzed protocol presents a compelling value proposition centered on cost efficiency, safety, and supply continuity. By replacing expensive stoichiometric oxidants and precious metal catalysts with commodity chemicals like iodine and DABCO, the raw material cost profile is drastically improved, allowing for significant margin expansion in high-volume production scenarios. The elimination of hazardous peroxides reduces the regulatory burden associated with the storage and transport of dangerous goods, lowering insurance premiums and facility compliance costs while enhancing overall site safety. Moreover, the reliance on atmospheric oxygen as the oxidant removes the logistical complexity of sourcing and handling specialized gaseous reagents, ensuring that production is never halted due to supply chain disruptions of critical oxidizing agents. The mild reaction conditions also extend the lifespan of reactor equipment by reducing corrosion and thermal stress, contributing to lower capital maintenance expenditures over the long term.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost reagents with low-cost alternatives and the reduction of waste disposal fees. Since the method avoids the use of stoichiometric hypervalent iodine or transition metals, the cost of goods sold (COGS) is significantly lowered, and the expense associated with removing trace metal impurities to meet pharmaceutical standards is completely eradicated. The high atom economy of using air as the oxidant means that fewer raw materials are purchased per kilogram of product, directly impacting the bottom line. Additionally, the energy consumption is minimal due to the room temperature operation and the use of low-power CFL bulbs, further contributing to operational expenditure savings without compromising reaction efficiency or yield.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly strengthened by the use of universally available reagents that are not subject to the geopolitical volatility often seen with rare earth metals or specialized fine chemicals. Iodine and DABCO are produced at massive scales for various industries, ensuring a stable and continuous supply even during global market fluctuations. The simplicity of the reaction setup, which does not require specialized high-pressure or cryogenic equipment, allows for flexible manufacturing across multiple sites or contract manufacturing organizations (CMOs) without extensive retooling. This flexibility reduces lead time for high-purity pharmaceutical intermediates by enabling rapid scale-up and decentralized production strategies, ensuring that critical drug supply chains remain robust and responsive to market demand.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this technology aligns perfectly with modern sustainability goals and regulatory frameworks such as REACH and TSCA. The generation of minimal waste and the absence of toxic heavy metals simplify the wastewater treatment process, reducing the environmental footprint of the manufacturing facility. The photochemical nature of the reaction is inherently scalable using flow chemistry or multi-lamp batch reactors, allowing for seamless transition from gram-scale R&D to ton-scale commercial production. This ease of scale-up ensures that the commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly, meeting tight project timelines while maintaining strict adherence to environmental, health, and safety (EHS) standards required by global regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational parameters and strategic benefits for stakeholders evaluating this method for their own manufacturing pipelines. Understanding these details is crucial for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of this visible-light method over traditional oxidation?
A: Unlike traditional methods using stoichiometric hypervalent iodine or toxic peroxides, this patent utilizes molecular oxygen from air as the terminal oxidant and visible light as a clean energy source, significantly reducing hazardous waste and operational costs.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates at room temperature with simple reagents (iodine, DABCO, methanol) and avoids expensive transition metal catalysts, making it highly scalable and economically viable for commercial manufacturing of pharmaceutical intermediates.
Q: What is the substrate scope for this alpha-hydroxylation reaction?
A: The method demonstrates broad applicability across various alpha-methylene ketones, including those with electron-donating or withdrawing groups on the aromatic ring, as well as heteroaromatic and aliphatic substrates, yielding secondary alpha-hydroxy ketones efficiently.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this visible-light catalyzed synthesis for producing high-value pharmaceutical intermediates with superior purity and sustainability. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market supply is seamless and efficient. Our state-of-the-art facilities are equipped with advanced photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alpha-hydroxy ketone delivered meets the highest industry standards. We are committed to leveraging this green technology to provide our clients with a competitive edge through cost-effective and environmentally responsible manufacturing solutions.
We invite you to engage with our technical procurement team to discuss how this innovative route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in delivering high-quality chemical solutions for the global pharmaceutical market.
