Advanced Metal-Free Synthesis of Polysubstituted Pyrroles for Pharmaceutical and Agrochemical Applications
Advanced Metal-Free Synthesis of Polysubstituted Pyrroles for Pharmaceutical and Agrochemical Applications
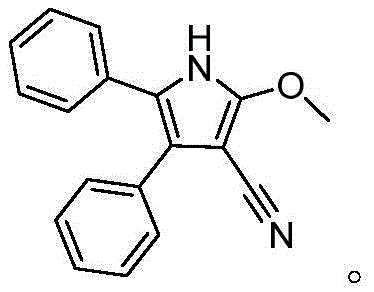
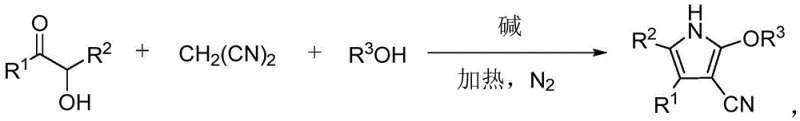
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient, sustainable, and cost-effective synthetic routes for high-value intermediates. A significant breakthrough in this domain is documented in Chinese Patent CN110452148B, which discloses a novel method for synthesizing polysubstituted pyrrole compounds. This technology leverages a three-component tandem reaction involving α-hydroxy ketones, malononitrile, and alcohols under basic promotion. Unlike traditional methodologies that often rely on toxic reagents or expensive catalytic systems, this approach offers a streamlined pathway to construct the pyrrole core, a structural motif ubiquitous in bioactive molecules ranging from anticancer agents like sunitinib to advanced agrochemical fungicides. For R&D directors and procurement strategists in the pharmaceutical and fine chemical sectors, understanding the nuances of this metal-free protocol is critical for optimizing supply chains and reducing the cost of goods sold (COGS) for complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrrole ring has relied on classical named reactions such as the Paal-Knorr synthesis or the Hantzsch pyrrole synthesis. While these methods are foundational, they frequently suffer from significant drawbacks when applied to modern, complex molecule synthesis. The Paal-Knorr reaction, for instance, typically requires 1,4-diketones which can be difficult to source or synthesize with high regioselectivity. Furthermore, many contemporary methods for generating polysubstituted pyrroles involve the use of isonitriles, which are notorious for their toxicity and unpleasant odor, posing severe safety and environmental challenges in a manufacturing setting. Additionally, a substantial number of modern catalytic approaches depend on precious transition metals like palladium or rhodium. While effective, these metals introduce a critical bottleneck: the stringent requirement for removing trace metal residues to meet pharmaceutical purity standards, a process that drastically increases processing time and waste generation.
The Novel Approach
The methodology outlined in patent CN110452148B represents a paradigm shift by utilizing a multicomponent reaction strategy that bypasses these traditional hurdles. By employing α-hydroxy ketones, malononitrile, and alcohols as the primary building blocks, the reaction achieves high atom economy and structural diversity in a single pot. Crucially, the alcohol serves a dual role as both a reactant and the solvent, which simplifies the reaction matrix and reduces the volume of auxiliary solvents required. The use of simple bases such as sodium hydroxide or sodium tert-butoxide eliminates the dependency on transition metals entirely. This not only lowers the raw material costs but also simplifies the downstream purification process, as there is no need for specialized scavengers to remove heavy metals. The result is a robust, green chemistry protocol that is inherently safer and more economically viable for the production of high-purity pharmaceutical intermediates.
Mechanistic Insights into Base-Promoted Multicomponent Cyclization
From a mechanistic perspective, this transformation is a fascinating example of base-promoted cascade cyclization. The reaction initiates with the deprotonation of the active methylene group in malononitrile by the base, generating a nucleophilic carbanion. This species attacks the carbonyl carbon of the α-hydroxy ketone, leading to an intermediate that undergoes dehydration and subsequent cyclization. The alcohol molecule then participates in the reaction, likely through a nucleophilic attack or transesterification-like process, to install the alkoxy group at the 2-position of the pyrrole ring. This intricate dance of bond formations occurs seamlessly under thermal conditions, typically between 80°C and 110°C, demonstrating the high reactivity of the chosen substrates. The tolerance for various functional groups on the α-hydroxy ketone, including electron-withdrawing halogens and electron-donating methoxy groups, suggests a mechanism that is resilient to electronic perturbations, ensuring consistent performance across a wide range of substrates.
Impurity control is another critical aspect where this mechanism excels. In transition-metal catalyzed reactions, side products often arise from homocoupling or incomplete catalytic cycles, leading to complex impurity profiles that are difficult to separate. In this base-mediated system, the reaction pathway is more direct, and the byproducts are generally inorganic salts or unreacted starting materials that are easily removed during the aqueous workup. The patent data indicates that the reaction proceeds cleanly to form the desired polysubstituted pyrrole core with minimal formation of oligomeric side products. This cleanliness is vital for regulatory compliance, as it ensures that the final API intermediate meets the rigorous impurity specifications required by global health authorities. The ability to achieve high purity without extensive chromatographic purification at scale is a significant advantage for process chemists aiming to streamline manufacturing workflows.
How to Synthesize Polysubstituted Pyrroles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The patent provides a detailed framework for executing this transformation, emphasizing the importance of an inert atmosphere and precise temperature control. The procedure begins with the preparation of a mixed solution containing the α-hydroxy ketone, malononitrile, alcohol, and the chosen base. It is imperative to purge the reaction vessel with nitrogen to prevent oxidation of sensitive intermediates, which could otherwise lead to reduced yields or the formation of colored impurities. The reaction mixture is then heated and stirred for a duration ranging from 6 to 24 hours, depending on the specific reactivity of the substrates involved. Following the reaction, a standard workup involving water washing and ethyl acetate extraction effectively isolates the crude product, which can be further purified via column chromatography if necessary. For a comprehensive, step-by-step operational guide tailored to your specific facility capabilities, please refer to the standardized synthesis protocol below.
- Charge a reaction vessel with alpha-hydroxy ketone, malononitrile, alcohol (acting as both reactant and solvent), and a base promoter such as sodium tert-butoxide.
- Evacuate the vessel and backfill with nitrogen gas to create an inert atmosphere, then heat the mixture to 80-110°C with stirring for 6 to 24 hours.
- Cool the reaction to room temperature, perform aqueous workup followed by ethyl acetate extraction, dry the organic layer, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic methodology offers tangible strategic benefits that extend beyond mere chemical efficiency. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By avoiding the use of scarce or geopolitically sensitive transition metals, manufacturers can mitigate the risk of supply disruptions and price volatility associated with precious metal catalysts. Furthermore, the use of commodity chemicals like alcohols and simple inorganic bases ensures a stable and cost-effective supply of reagents. This stability translates directly into more predictable production schedules and reduced lead times for the delivery of critical intermediates to downstream API manufacturers. The elimination of heavy metal catalysts also means that the environmental footprint of the manufacturing process is significantly reduced, aligning with increasingly stringent global environmental regulations and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free protocol is substantial. By removing the need for expensive palladium or rhodium catalysts, the direct material cost of the reaction is lowered considerably. Moreover, the downstream processing costs are significantly reduced because the complex and costly steps required to scavenge trace metals from the final product are rendered unnecessary. This simplification of the purification train allows for faster batch turnover and lower utility consumption. Additionally, since the alcohol acts as the solvent, there is a reduction in the volume of organic solvents that need to be purchased, recovered, or disposed of, further driving down the operational expenditure associated with solvent management and waste treatment facilities.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a robust pharmaceutical supply chain. This synthesis method enhances reliability by utilizing broadly available starting materials that are produced by multiple suppliers globally, reducing the risk of single-source dependency. The robustness of the reaction conditions, which tolerate a wide range of functional groups and moisture levels better than sensitive organometallic processes, ensures consistent batch-to-batch quality. This consistency minimizes the risk of batch failures and the need for re-processing, which can cause significant delays in the supply chain. Consequently, partners can rely on a more steady flow of high-quality intermediates, ensuring that their own production timelines for finished drug products remain uninterrupted and efficient.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden challenges, but this method is designed with scalability in mind. The exothermic nature of the reaction is manageable, and the use of common solvents like alcohols facilitates heat transfer and mixing in large reactors. From an environmental compliance standpoint, the absence of toxic isonitriles and heavy metals simplifies the permitting process and reduces the liability associated with hazardous waste disposal. The high atom economy of the multicomponent reaction means that a greater proportion of the raw materials end up in the final product, minimizing waste generation. This alignment with green chemistry principles not only reduces disposal costs but also enhances the corporate social responsibility profile of the manufacturing entity, making it a more attractive partner for environmentally conscious multinational corporations.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical and commercial viability of this synthesis method, we have compiled a set of frequently asked questions based on the patent data and industry expertise. These questions cover critical aspects such as catalyst residues, substrate flexibility, and process safety, providing clarity for technical decision-makers evaluating this technology for integration into their existing portfolios. Understanding these details is essential for assessing the feasibility of adopting this route for specific target molecules and for planning the necessary infrastructure adjustments.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method utilizes inexpensive inorganic or organic bases (e.g., NaOH, t-BuONa) as promoters, completely eliminating the need for costly transition metal catalysts and the subsequent heavy metal removal steps.
Q: What is the substrate scope for the alcohol component in this reaction?
A: The method demonstrates excellent adaptability, accommodating various alcohols including methanol, ethanol, n-butanol, and even diols like 1,4-butanediol, allowing for diverse functionalization of the pyrrole ring.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process features mild reaction conditions, readily available raw materials, and simple workup procedures, making it highly scalable and safe for commercial manufacturing of fine chemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrrole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the one described in patent CN110452148B. As a leading CDMO and supplier of fine chemical intermediates, we possess the technical expertise to translate such innovative laboratory protocols into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this metal-free synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of polysubstituted pyrrole intermediate meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you require custom synthesis of specific pyrrole derivatives or a comprehensive Customized Cost-Saving Analysis for your current supply chain, our technical procurement team is ready to assist. Contact us today to request specific COA data, discuss route feasibility assessments, and discover how our commitment to innovation and quality can drive value for your organization. Let us be your trusted partner in navigating the complexities of modern chemical manufacturing.
