Advanced Catalytic Coupling for High-Purity Alpha-Hydroxy Ketones: A Commercial Scale-Up Perspective
The landscape of fine chemical synthesis is constantly evolving, driven by the need for more efficient, sustainable, and cost-effective pathways to critical building blocks. A pivotal advancement in this domain is documented in Chinese Patent CN102177125B, which discloses a novel process for producing alpha-hydroxy ketone compounds. These versatile intermediates are indispensable in the construction of complex pharmacophores, serving as key precursors for a myriad of active pharmaceutical ingredients (APIs). The patent introduces a groundbreaking methodology that utilizes specific alkoxyimidazolidine compounds, represented by Formula (1), as highly effective organocatalysts. Unlike traditional approaches that rely on unstable carbene precursors generated in situ, this invention leverages pre-formed, stable catalysts that exhibit remarkable tolerance to reaction conditions. For R&D directors and procurement managers alike, this technology represents a significant leap forward, promising enhanced purity profiles and streamlined manufacturing workflows. By shifting the paradigm from sensitive carboxylate systems to robust alkoxyimidazolidines, the industry can achieve greater consistency in the production of high-purity pharmaceutical intermediates, ultimately securing the supply chain for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-hydroxy ketones via aldehyde coupling has been fraught with technical challenges that hinder large-scale adoption. Prior art, such as that described in U.S. Patent Application 2008/0051608, relies heavily on the use of 1,3-disubstituted imidazolinium-2-carboxylates. These catalytic species are typically generated in situ by reacting imidazolinium salts with carbon dioxide in the presence of a base. This requirement introduces significant operational complexity, as the handling of carbon dioxide gas necessitates specialized pressure equipment and strict control over stoichiometry. Furthermore, the resulting carboxylate intermediates are often sensitive to moisture and atmospheric oxygen, leading to potential catalyst deactivation and inconsistent reaction outcomes. From a commercial perspective, these sensitivities translate into higher operational expenditures due to the need for rigorous exclusion of air and water, as well as the additional costs associated with gas handling infrastructure. The variability inherent in generating the active catalyst species in situ can also result in fluctuating yields and impurity profiles, posing a substantial risk to the quality control standards required for reliable API intermediate supplier operations.
The Novel Approach
In stark contrast to these legacy methods, the process outlined in CN102177125B employs pre-synthesized alkoxyimidazolidine compounds that function as stable, ready-to-use catalysts. This fundamental shift eliminates the need for in situ carboxylation, thereby removing the dependency on carbon dioxide feedstocks and the associated engineering controls. The novel approach allows for the direct coupling of aldehyde compounds, such as those represented by Formula (2) and Formula (4), under remarkably mild conditions. As illustrated in the reaction scheme below, the cross-coupling of distinct aldehydes leads to the formation of valuable alpha-hydroxy ketone structures like Formula (5).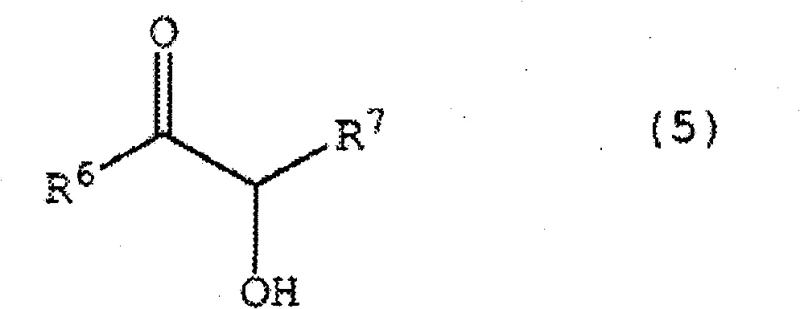 This methodology not only simplifies the reactor setup but also enhances the reproducibility of the synthesis. By utilizing a stable catalyst that can be weighed and added directly to the reaction mixture, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through reduced cycle times and lower utility consumption. The ability to operate effectively across a broad temperature range, from -20°C to 200°C, provides process engineers with the flexibility to optimize kinetics without compromising safety or equipment integrity, making this a superior choice for the commercial scale-up of complex polymer additives and fine chemicals.
This methodology not only simplifies the reactor setup but also enhances the reproducibility of the synthesis. By utilizing a stable catalyst that can be weighed and added directly to the reaction mixture, manufacturers can achieve cost reduction in pharmaceutical intermediate manufacturing through reduced cycle times and lower utility consumption. The ability to operate effectively across a broad temperature range, from -20°C to 200°C, provides process engineers with the flexibility to optimize kinetics without compromising safety or equipment integrity, making this a superior choice for the commercial scale-up of complex polymer additives and fine chemicals.
Mechanistic Insights into Alkoxyimidazolidine-Catalyzed Coupling
The efficacy of this process lies in the unique structural features of the alkoxyimidazolidine catalyst, depicted in Formula (1). The core of the catalyst is an imidazolidine ring substituted with bulky groups at the nitrogen atoms (R3 and R4) and an alkoxy group at the C2 position (R5).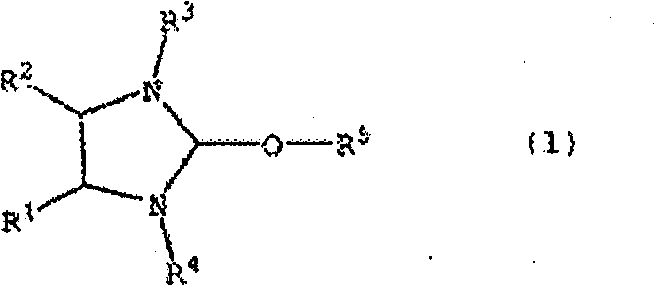 These bulky substituents, such as 2,6-diisopropylphenyl or 2,4,6-trimethylphenyl groups, play a critical role in stabilizing the catalyst against decomposition while simultaneously creating a steric environment that favors the desired coupling pathway. Mechanistically, the alkoxyimidazolidine acts as a precursor to an N-heterocyclic carbene (NHC) or functions directly as a nucleophilic catalyst that activates the aldehyde substrate. The electron-donating nature of the alkoxy group modulates the electrophilicity of the C2 carbon, facilitating the formation of the key Breslow intermediate or its equivalent. This activation lowers the energy barrier for the nucleophilic attack of one aldehyde onto another, driving the formation of the carbon-carbon bond that characterizes the alpha-hydroxy ketone motif. The robustness of this catalytic cycle ensures that the reaction proceeds with high turnover numbers, minimizing the amount of catalyst required and reducing the burden on downstream purification processes.
These bulky substituents, such as 2,6-diisopropylphenyl or 2,4,6-trimethylphenyl groups, play a critical role in stabilizing the catalyst against decomposition while simultaneously creating a steric environment that favors the desired coupling pathway. Mechanistically, the alkoxyimidazolidine acts as a precursor to an N-heterocyclic carbene (NHC) or functions directly as a nucleophilic catalyst that activates the aldehyde substrate. The electron-donating nature of the alkoxy group modulates the electrophilicity of the C2 carbon, facilitating the formation of the key Breslow intermediate or its equivalent. This activation lowers the energy barrier for the nucleophilic attack of one aldehyde onto another, driving the formation of the carbon-carbon bond that characterizes the alpha-hydroxy ketone motif. The robustness of this catalytic cycle ensures that the reaction proceeds with high turnover numbers, minimizing the amount of catalyst required and reducing the burden on downstream purification processes.
Furthermore, the specific substitution pattern of the catalyst exerts profound control over the impurity profile of the final product. In conventional base-catalyzed aldol condensations, side reactions such as self-condensation or polymerization of the aldehyde substrates are common, leading to complex mixtures that are difficult to separate. However, the steric bulk of the R3 and R4 groups in the alkoxyimidazolidine catalyst effectively shields the active site, preventing uncontrolled oligomerization. This steric shielding ensures that the coupling occurs selectively between the intended aldehyde partners, significantly reducing the formation of high-molecular-weight byproducts. For quality assurance teams, this means a cleaner crude reaction mixture, which translates to higher recovery rates during isolation and a final product that meets stringent purity specifications with less intensive chromatographic purification. The ability to suppress these parasitic side reactions is a key factor in achieving the high yields reported in the patent examples, such as the 75% yield observed in the synthesis of 4-(methylthio)-2-oxo-1-butanol.
How to Synthesize Alpha-Hydroxy Ketones Efficiently
Implementing this technology in a laboratory or pilot plant setting requires adherence to specific procedural guidelines to maximize efficiency and safety. The synthesis begins with the preparation or procurement of the specific alkoxyimidazolidine catalyst, ensuring that the bulky aryl or alkyl groups are correctly installed to provide the necessary steric protection. Once the catalyst is secured, the process involves the strategic mixing of the aldehyde substrates in an appropriate solvent system. The patent highlights the versatility of solvents, ranging from non-polar hydrocarbons like toluene and hexane to polar aprotic solvents like DMF and THF, allowing for optimization based on substrate solubility. The detailed standardized synthesis steps see the guide below for a comprehensive walkthrough of the reaction parameters, addition rates, and workup procedures necessary to replicate the high performance described in the intellectual property.
- Preparation of the alkoxyimidazolidine catalyst (Formula 1) by reacting imidazolinium salts with alkoxides in alcohol solvents.
- Mixing the aldehyde substrates (Formula 2 and Formula 4) in a suitable solvent such as toluene or THF under inert atmosphere.
- Adding the catalyst solution to the aldehyde mixture at controlled temperatures ranging from -20°C to 200°C, followed by purification via distillation or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this alkoxyimidazolidine-catalyzed process offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of carbon dioxide handling and the use of stable, shelf-stable catalysts drastically simplify the raw material logistics, reducing the risk of supply disruptions associated with specialty gases or sensitive reagents. This simplification directly contributes to a more resilient supply chain, ensuring that production schedules can be maintained even in the face of external logistical challenges. Moreover, the mild reaction conditions reduce the energy intensity of the manufacturing process, aligning with global sustainability goals and potentially lowering utility costs. The robustness of the catalyst also implies longer equipment life and reduced maintenance requirements, as the corrosive effects of strong bases or acidic workups often associated with alternative methods are mitigated.
- Cost Reduction in Manufacturing: The transition to this novel catalytic system eliminates the need for expensive and complex gas handling infrastructure required for carbon dioxide-based methods. By using a stable solid or liquid catalyst that can be dosed directly, facilities can reduce capital expenditure on reactor modifications and lower operational costs related to gas procurement and storage. Additionally, the high selectivity of the catalyst minimizes waste generation, leading to significant savings in raw material consumption and waste disposal fees. The simplified workup procedure, often requiring only concentration and filtration rather than extensive chromatography, further drives down the cost per kilogram of the final API intermediate, enhancing overall profit margins.
- Enhanced Supply Chain Reliability: The catalysts described in the patent, such as 2-methoxy-1,3-bis[(2,6-diisopropyl)phenyl]imidazolidine, are chemically stable and can be stored for extended periods without degradation. This stability allows manufacturers to maintain strategic stockpiles of the catalyst, buffering against market volatility and ensuring continuous production capability. Furthermore, the starting materials—various aldehydes—are commodity chemicals available from multiple global suppliers, reducing dependency on single-source vendors. This diversification of the supply base is critical for maintaining reducing lead time for high-purity pharmaceutical intermediates, as it prevents bottlenecks that could arise from shortages of specialized reagents.
- Scalability and Environmental Compliance: The process operates effectively in common industrial solvents like toluene and methanol, which are easily recovered and recycled, supporting a circular economy approach to chemical manufacturing. The absence of heavy metal catalysts removes the need for costly and environmentally burdensome metal scavenging steps, simplifying regulatory compliance and reducing the environmental footprint of the facility. The mild temperature requirements also enhance safety profiles, lowering the risk of thermal runaways and making the process easier to scale from kilogram to multi-ton production volumes. This scalability ensures that the technology can meet growing market demand without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-hydroxy ketone synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN102177125B, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing portfolios and for procurement specialists assessing the long-term viability of the supply chain.
Q: What are the advantages of using alkoxyimidazolidine catalysts over traditional imidazolinium carboxylates?
A: Alkoxyimidazolidine catalysts offer superior stability and do not require the in-situ generation from carbon dioxide and base, simplifying the process and reducing sensitivity to moisture and air compared to traditional methods.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process operates under mild conditions (e.g., 40°C) and uses commercially available solvents like toluene, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What types of aldehydes are compatible with this coupling reaction?
A: The method supports a wide range of aliphatic, aromatic, and heteroaromatic aldehydes, including those with bulky substituents, allowing for the synthesis of diverse alpha-hydroxy ketone structures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxy Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the alkoxyimidazolidine-catalyzed coupling process for the next generation of pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific solvent systems and temperature profiles required by this technology, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the highest industry standards. We are committed to delivering high-purity alpha-hydroxy ketones that serve as the foundation for your most critical drug development programs.
We invite you to collaborate with us to unlock the full commercial potential of this innovative synthesis route. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions with confidence. Together, we can drive efficiency and innovation in the global supply of fine chemical intermediates.
