Advanced Manufacturing of 2-Chloropyrimidine: A Cost-Effective Route for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, scalable pathways for constructing nitrogen-containing heterocycles, which serve as the backbone for countless active pharmaceutical ingredients (APIs). Patent CN102079725A introduces a transformative methodology for the preparation of 2-chloropyrimidine, a critical building block in medicinal chemistry used extensively in the synthesis of antiviral and anticancer agents. Unlike traditional routes that rely on scarce or hazardous reagents, this innovation leverages a cascade of reactions starting from dicyandiamide and ammonium chloride, two widely available commodity chemicals. The technical breakthrough lies not only in the economic viability of the raw materials but also in the operational simplicity of the three-step sequence, which avoids the need for complex purification between intermediate stages. By integrating the formation of guanidine hydrochloride directly into the cyclization step, the process minimizes material loss and handling time, offering a streamlined solution for commercial scale-up of complex heterocyclic intermediates. This report analyzes the mechanistic depth and commercial implications of this patented technology for global supply chain optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-chloropyrimidine has been plagued by significant economic and environmental hurdles that hinder large-scale adoption. Prior art methods frequently utilized diethyl malonate and phosphorus oxychloride (POCl3) as key reagents; however, POCl3 is notoriously toxic, corrosive, and generates substantial amounts of phosphorous-containing wastewater, creating severe disposal challenges for modern manufacturing facilities. Furthermore, alternative routes described in literature often necessitate the direct purchase of 2-aminopyrimidine or 2-mercaptopyrimidine as starting materials, both of which are specialized fine chemicals with volatile market prices and limited availability from reliable pharmaceutical intermediate suppliers. Another critical bottleneck in conventional protocols is the reliance on hot toluene for extraction and purification, a solvent with a high boiling point that requires significant energy input for removal and poses occupational health risks due to its toxicity. These factors collectively result in a high cost of goods sold (COGS) and a fragile supply chain susceptible to raw material shortages, making traditional methods unsuitable for the high-volume demands of the generic pharmaceutical sector.
The Novel Approach
The methodology disclosed in CN102079725A fundamentally restructures the synthetic logic by adopting a "bottom-up" approach, constructing the pyrimidine ring from simple acyclic precursors rather than modifying expensive existing rings. The process initiates with the pyrolysis of dicyandiamide and ammonium chloride to generate guanidine hydrochloride in situ, effectively bypassing the need to purchase this intermediate separately. This crude guanidine salt is then subjected to a condensation reaction with 1,1,3,3-tetramethoxypropane in an industrial hydrochloric acid medium, facilitating a smooth cyclization to form 2-aminopyrimidine without the need for isolation. 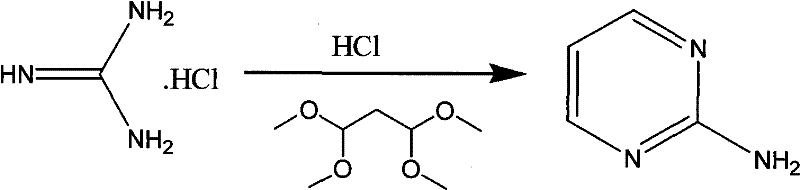 The final transformation involves a diazotization-chlorination sequence using sodium nitrite and zinc chloride, which proceeds under mild acidic conditions to yield the target 2-chloropyrimidine. This route eliminates the use of phosphorus oxychloride entirely and replaces hot toluene extraction with dichloromethane, a solvent that is easier to recover and recycle. The result is a process that offers cost reduction in pharmaceutical intermediate manufacturing through both raw material savings and simplified downstream processing, ensuring a more stable and continuous production capability.
The final transformation involves a diazotization-chlorination sequence using sodium nitrite and zinc chloride, which proceeds under mild acidic conditions to yield the target 2-chloropyrimidine. This route eliminates the use of phosphorus oxychloride entirely and replaces hot toluene extraction with dichloromethane, a solvent that is easier to recover and recycle. The result is a process that offers cost reduction in pharmaceutical intermediate manufacturing through both raw material savings and simplified downstream processing, ensuring a more stable and continuous production capability.
Mechanistic Insights into Zinc Chloride Catalyzed Diazotization
The core chemical innovation of this patent resides in the efficient conversion of the amino group to a chloro substituent via a modified Sandmeyer-type reaction mechanism. In the final step, 2-aminopyrimidine is treated with sodium nitrite in the presence of excess hydrochloric acid to form the unstable diazonium salt intermediate. Crucially, the addition of zinc chloride (ZnCl2) acts as a Lewis acid catalyst that stabilizes the transition state and facilitates the nucleophilic attack of the chloride ion on the diazonium species. The reaction is conducted at strictly controlled low temperatures, typically between -20°C and 0°C, to prevent the decomposition of the diazonium salt into phenolic byproducts or tars, which would severely compromise the purity profile. The stoichiometry is carefully balanced, with a preferred molar ratio of 2-aminopyrimidine to hydrochloric acid to zinc chloride to sodium nitrite of approximately 1:3:3:3, ensuring complete conversion while minimizing the formation of side products. This catalytic system is superior to non-catalyzed variants, providing higher yields and cleaner reaction profiles essential for high-purity pyrimidine derivatives required in GMP environments.
Impurity control is another vital aspect of this mechanistic design, particularly regarding the avoidance of oxygenated impurities. In traditional acidic hydrolysis conditions, there is a risk of the diazonium salt reacting with water to form 2-hydroxypyrimidine, a difficult-to-remove impurity that can persist through subsequent coupling reactions. The presence of high concentrations of chloride ions from the industrial hydrochloric acid and zinc chloride creates a kinetic preference for chlorination over hydrolysis, effectively suppressing the formation of hydroxy-species. Furthermore, the use of dichloromethane for extraction allows for the selective partitioning of the lipophilic 2-chloropyrimidine away from inorganic salts and polar byproducts remaining in the aqueous phase. The final product is obtained as a faint yellow solid with a melting point of 64-65°C and an HPLC purity exceeding 98%, demonstrating the robustness of the purification strategy. This level of quality control is paramount for customers seeking to minimize purification burdens in their own API synthesis workflows.
How to Synthesize 2-Chloropyrimidine Efficiently
The execution of this synthesis requires precise thermal management and sequential addition of reagents to maximize yield and safety. The process begins with the solid-state pyrolysis of dicyandiamide and ammonium chloride, followed by a liquid-phase cyclization and a final low-temperature diazotization. Operators must adhere strictly to the temperature gradients specified in the patent, particularly during the exothermic addition of sodium nitrite, to maintain process safety and product integrity. The detailed standardized operating procedures, including specific addition rates, stirring speeds, and quenching protocols, are outlined below to ensure reproducibility at the pilot and plant scales.
- Pyrolyze dicyandiamide and ammonium chloride at 150-230°C to generate crude guanidine hydrochloride.
- React guanidine hydrochloride with 1,1,3,3-tetramethoxypropane in industrial hydrochloric acid at 70-100°C to form 2-aminopyrimidine.
- Perform diazotization using sodium nitrite and zinc chloride in hydrochloric acid at -20 to 0°C to yield 2-chloropyrimidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits beyond simple unit price reductions. The shift from specialized, low-volume starting materials to bulk commodity chemicals drastically de-risks the supply chain, ensuring that production schedules are not held hostage by the availability of niche reagents like diethyl malonate or pure aminopyrimidines. Additionally, the elimination of phosphorus oxychloride removes a highly regulated substance from the facility's inventory, reducing regulatory compliance costs and insurance liabilities associated with handling toxic corrosives. The simplified workup procedure, which avoids energy-intensive distillation of high-boiling solvents like toluene, translates directly into lower utility costs and faster batch cycle times, enhancing overall manufacturing throughput.
- Cost Reduction in Manufacturing: The primary driver for cost efficiency in this process is the substitution of expensive precursors with dicyandiamide and ammonium chloride, which are produced on a massive industrial scale and benefit from stable pricing dynamics. By generating the guanidine intermediate in situ, the process eliminates the markup associated with purchasing isolated guanidine salts, while the use of industrial grade hydrochloric acid further minimizes solvent expenses. The removal of the hot toluene extraction step also reduces solvent consumption and recovery costs, as dichloromethane is easier to distill and recycle. These cumulative efficiencies result in a significantly lower cost base per kilogram of finished product, allowing for more competitive pricing in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: Relying on commodity feedstocks ensures a consistent and uninterrupted flow of materials, as dicyandiamide and ammonium chloride are produced by numerous manufacturers worldwide, preventing single-source bottlenecks. The robustness of the reaction conditions, which tolerate slight variations in temperature and stoichiometry without catastrophic yield loss, adds a layer of operational resilience to the manufacturing process. This reliability is crucial for maintaining long-term supply agreements with pharmaceutical clients who require guaranteed delivery timelines for their own production planning. Furthermore, the stability of the intermediates allows for flexible scheduling, enabling manufacturers to stockpile key precursors during periods of low demand to buffer against market fluctuations.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard reactor configurations and avoiding exotic catalysts or extreme pressure conditions that complicate engineering design. From an environmental perspective, the absence of phosphorus waste streams simplifies wastewater treatment, reducing the load on effluent treatment plants and lowering disposal fees. The switch to dichloromethane, while still requiring careful management, allows for more efficient closed-loop solvent recovery systems compared to toluene, aligning with modern green chemistry principles. These factors make the technology highly attractive for facilities aiming to expand capacity while meeting increasingly stringent environmental, social, and governance (ESG) targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 2-chloropyrimidine synthesis route. These answers are derived directly from the experimental data and beneficial effects reported in patent CN102079725A, providing clarity on yield expectations, purity profiles, and operational requirements. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing portfolios.
Q: What are the primary cost advantages of this synthesis route?
A: This method replaces expensive precursors like diethyl malonate or pure 2-aminopyrimidine with commodity chemicals such as dicyandiamide and ammonium chloride, significantly lowering raw material costs.
Q: How does this process improve environmental compliance?
A: By eliminating the use of highly toxic phosphorus oxychloride and avoiding large volumes of hot toluene for extraction, the process reduces hazardous waste generation and simplifies solvent recovery.
Q: What purity levels can be achieved with this method?
A: The optimized protocol, utilizing dichloromethane extraction and precise temperature control during diazotization, consistently yields product with HPLC purity greater than 98%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Chloropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven scaling capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are faithfully reproduced in our manufacturing suites. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of 2-chloropyrimidine meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your downstream synthesis processes.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized version of this patented route, we can help you achieve significant reductions in your overall material costs while securing a stable supply of this critical intermediate. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than theoretical projections. Let us collaborate to optimize your supply chain and accelerate your drug development timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
