Advanced Maleate Salt Formation Technology for High-Purity Silodosin Intermediate Manufacturing
The pharmaceutical landscape for treating benign prostatic hyperplasia (BPH) relies heavily on the efficient production of high-quality active ingredients, with Silodosin standing out as a critical therapeutic agent. Patent CN102382029A introduces a transformative approach to synthesizing a key silodosin intermediate, specifically focusing on the preparation of its maleate salt form. This innovation addresses longstanding challenges in purification efficiency and yield stability that have plagued previous manufacturing protocols. By shifting from traditional oxalate salt formation to a novel maleate crystallization strategy, the process achieves a remarkable enhancement in impurity removal, particularly targeting structurally similar by-products that often compromise final drug safety. For a reliable pharmaceutical intermediates supplier, adopting such advanced crystallization technologies is essential to meet the stringent purity specifications demanded by global regulatory bodies. This report analyzes the technical merits of this maleate formation method, highlighting its potential to redefine cost reduction in pharmaceutical intermediates manufacturing through simplified operations and superior product consistency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of this specific indoline-based intermediate relied heavily on oxalate salt formation, a method fraught with significant operational inefficiencies and yield limitations. The conventional oxalate crystallization process typically suffers from low recovery rates, necessitating the addition of specific seed crystals to initiate precipitation, which adds complexity and variability to the batch process. Furthermore, this traditional approach often requires extended standing periods to allow for complete crystal growth, drastically increasing the production cycle time and tying up valuable reactor capacity. In large-scale operations, the oxalate method demonstrates poor suitability for industrial amplification, as the purification effect on specific by-products, such as the bis-adduct impurity, remains suboptimal even after multiple recrystallization attempts. These inherent drawbacks result in higher production costs and inconsistent quality profiles, creating bottlenecks for supply chain managers seeking reliable sources of high-purity silodosin intermediate.
The Novel Approach
In stark contrast, the novel method described in the patent utilizes maleic acid to form a stable salt with the intermediate compound in a carefully optimized mixed solvent system. This approach eliminates the need for seed crystals and prolonged standing times, allowing for rapid and reproducible crystallization directly upon cooling. The use of a good solvent like tetrahydrofuran (THF) combined with a poor solvent such as isopropyl ether creates an ideal thermodynamic environment for the selective precipitation of the target maleate salt. This strategic solvent pairing ensures that the desired product crystallizes efficiently while leaving the majority of impurities dissolved in the mother liquor, thereby achieving high purity in a single step. The operational simplicity of this new route not only enhances yield but also significantly reduces the labor and energy inputs required for purification, marking a substantial advancement over the cumbersome oxalate-based legacy processes.
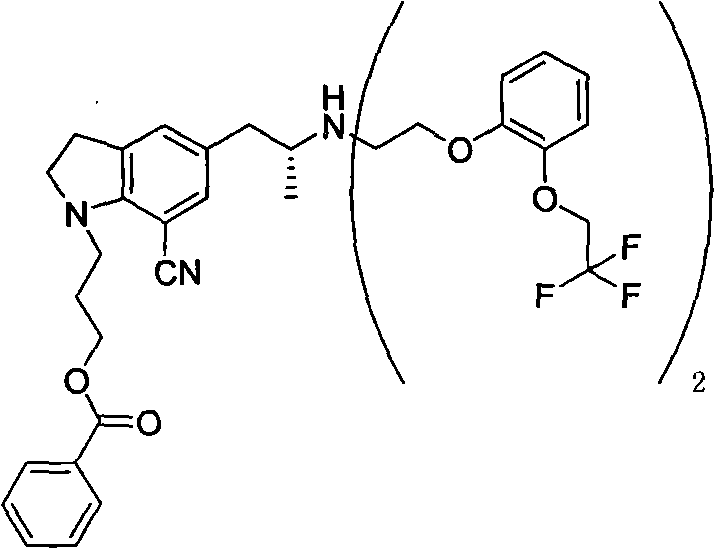
Mechanistic Insights into Maleate Salt Crystallization and Impurity Control
The core mechanism driving the success of this purification strategy lies in the differential solubility properties of the maleate salt compared to its impurities within the selected solvent matrix. When the crude intermediate reacts with maleic acid, the resulting salt forms a crystal lattice that is thermodynamically stable and less soluble in the THF and isopropyl ether mixture than the free base or the oxalate analog. This precise control over solubility allows for the effective exclusion of the bis-adduct impurity, a structurally related by-product that typically co-precipitates in less selective systems. The patent data indicates that through this maleate salification process, the content of the problematic bis-adduct impurity can be reduced to below 1%, a level of purity that is difficult to achieve with conventional methods. This high degree of selectivity is crucial for downstream processing, as it minimizes the burden on subsequent purification steps and ensures the final API meets rigorous quality standards.
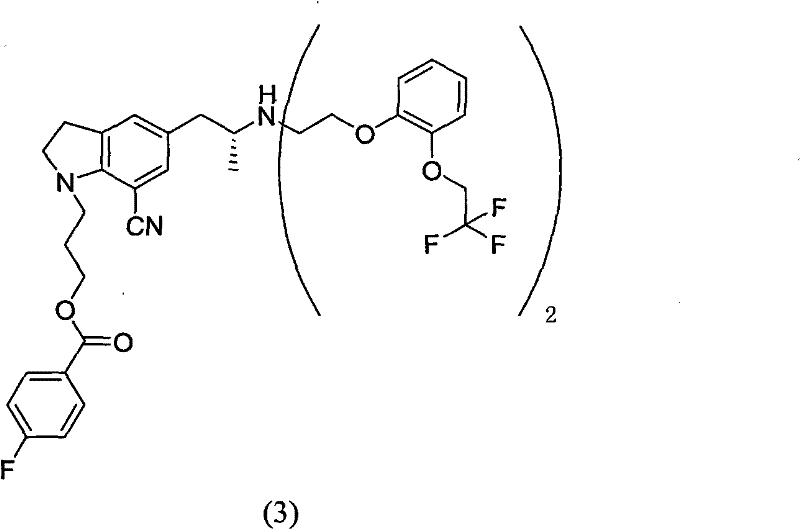
Furthermore, the mechanistic pathway facilitates a seamless transition to the final active pharmaceutical ingredient through a straightforward hydrolysis step. Once the high-purity maleate salt is isolated, it can be directly subjected to alkaline hydrolysis to regenerate the free base intermediate or proceed to the final silodosin synthesis without additional purification burdens. The stability of the maleate salt ensures that the intermediate can be stored or transported with minimal risk of degradation, providing flexibility in manufacturing scheduling. This robustness is particularly valuable when scaling up complex pharmaceutical intermediates, as it reduces the risk of batch failure due to instability or unexpected impurity formation. The ability to consistently produce material with impurity levels significantly lower than industry averages underscores the technical superiority of this maleate-based crystallization protocol.
How to Synthesize Silodosin Intermediate Efficiently
The synthesis of this critical silodosin intermediate via the maleate salt route involves a streamlined sequence of dissolution, reaction, and crystallization steps designed for maximum efficiency. The process begins by dissolving the crude compound and maleic acid in a heated mixture of THF and isopropyl ether, followed by controlled cooling to induce precipitation of the pure salt. Detailed standard operating procedures regarding specific temperature ramps, stirring rates, and filtration techniques are essential for replicating the high yields reported in the patent data. For R&D teams looking to implement this technology, understanding the precise stoichiometry and solvent ratios is key to optimizing the process for their specific equipment constraints. The detailed standardized synthesis steps are outlined in the guide below to ensure consistent replication of these results.
- Dissolve the crude silodosin intermediate compound and maleic acid in a heated mixture of THF and isopropyl ether.
- Cool the solution to 0-15°C to induce crystallization of the maleate salt while impurities remain in solution.
- Filter the solid maleate salt and optionally wash with poor solvent to remove residual impurities before hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this maleate salt formation technology offers profound advantages for procurement managers and supply chain heads focused on efficiency and cost control. The elimination of seed crystals and the reduction in processing time directly translate to increased throughput and lower operational expenditures per kilogram of produced intermediate. By simplifying the purification workflow, manufacturers can reduce the consumption of solvents and energy, contributing to both cost reduction in pharmaceutical intermediates manufacturing and improved environmental sustainability profiles. The robustness of the process also enhances supply chain reliability, as the risk of batch rejection due to purity failures is significantly mitigated. This stability allows for more accurate forecasting and inventory management, ensuring a continuous flow of materials to downstream API production facilities without unexpected interruptions.
- Cost Reduction in Manufacturing: The novel maleate salt method drastically simplifies the operational workflow by removing the need for expensive seed crystals and extended reaction times, leading to substantial cost savings in labor and utility consumption. The higher yield achieved through this method means that less raw material is wasted, optimizing the overall material balance and reducing the cost of goods sold. Additionally, the use of common, recoverable solvents like THF and isopropyl ether facilitates efficient solvent recycling programs, further driving down variable production costs. These cumulative efficiencies make the maleate route a financially superior choice for large-scale commercial production compared to legacy oxalate methods.
- Enhanced Supply Chain Reliability: The stability and reproducibility of the maleate crystallization process ensure a consistent supply of high-quality intermediate, reducing the volatility often associated with complex chemical manufacturing. By minimizing the occurrence of off-spec batches, suppliers can maintain tighter delivery schedules and fulfill contractual obligations with greater confidence. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing pharmaceutical companies to accelerate their own development and production timelines. A stable supply of this key building block supports the uninterrupted manufacturing of Silodosin, safeguarding against market shortages and ensuring patient access to essential medications.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates, utilizing standard unit operations that are easily adaptable to multi-ton production scales. The efficient removal of impurities reduces the load on waste treatment systems, aligning with increasingly strict environmental regulations and corporate sustainability goals. The ability to achieve high purity in fewer steps minimizes the generation of chemical waste, supporting a greener manufacturing footprint. This scalability ensures that the technology can grow with market demand, providing a future-proof solution for the long-term supply of silodosin intermediates.
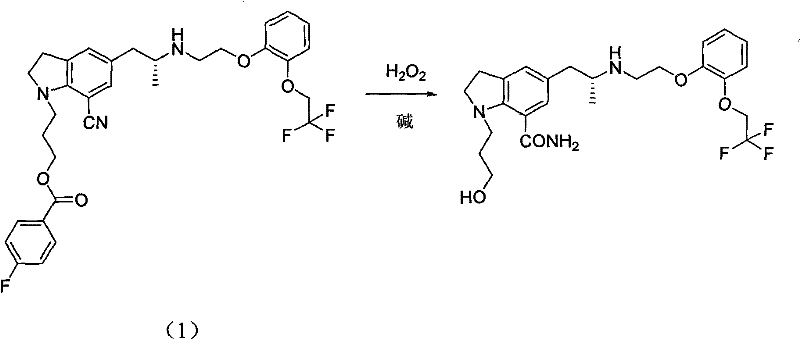
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this maleate salt formation technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and quality implications for potential partners. Understanding these details is vital for making informed decisions about integrating this process into existing supply chains. The answers provided reflect the consensus on the technical feasibility and commercial viability of this advanced purification method.
Q: Why is the maleate salt preferred over the oxalate salt for this intermediate?
A: The maleate salt offers superior stability and higher yields without the need for seed crystals or extended standing periods required by the oxalate method, significantly simplifying the operational workflow.
Q: How does this method handle the bis-adduct impurity?
A: The specific solvent system utilizing THF and isopropyl ether creates a solubility differential that keeps the bis-adduct impurity in the mother liquor, reducing its content to below 1% effectively.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method eliminates complex seeding steps and uses common industrial solvents, making it highly scalable and robust for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Silodosin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of employing advanced purification technologies like the maleate salt formation method to deliver superior pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to full-scale manufacturing. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We understand that the consistency of the silodosin intermediate is paramount for the efficacy and safety of the final drug product, and our facilities are equipped to handle the specific solvent systems and crystallization requirements of this novel process.
We invite you to collaborate with us to leverage these technical advancements for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. By partnering with NINGBO INNO PHARMCHEM, you secure a reliable source of high-purity materials backed by deep technical expertise and a commitment to operational excellence.
