Advanced Levocetirizine Synthesis: Technical Insights for Global Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for second-generation antihistamines, and the technical disclosure within patent CN101657437A offers a significant advancement in the preparation of Levocetirizine and its pharmaceutically acceptable salts. This specific intellectual property outlines a novel methodology utilizing diglycollic acid or its derivatives to construct the critical ether-acid side chain, bypassing several inefficiencies inherent in legacy processes. For R&D directors and technical decision-makers, understanding the nuances of this pathway is essential, as it directly impacts the impurity profile and the overall feasibility of GMP manufacturing. The innovation lies not just in the final coupling but in the strategic selection of intermediates that preserve chirality while streamlining the reaction sequence. By leveraging this specific chemical architecture, manufacturers can achieve higher throughput and reduced operational complexity, which are paramount metrics in modern fine chemical production.
Furthermore, the implications of this synthesis extend beyond the laboratory bench to the broader supply chain ecosystem, offering a reliable Levocetirizine intermediate supplier with a distinct competitive edge. The ability to produce high-purity Levocetirizine without resorting to cumbersome resolution techniques translates into tangible value for procurement teams looking for cost reduction in antihistamine manufacturing. As global demand for allergy medications remains resilient, securing a supply chain that is both chemically elegant and commercially viable is a strategic imperative. This report delves deep into the mechanistic and operational advantages of this patented route, providing a comprehensive analysis for stakeholders aiming to optimize their API sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Levocetirizine has been plagued by inefficient resolution steps and multi-stage protection strategies that erode overall yield and increase production costs. Prior art, such as the methods disclosed in GB 2,225,321, often relies on the hydrolysis of optically pure nitriles derived from racemic precursors, a process where the initial splitting yield can be as low as 12.7%. This severe loss of material at the early stages necessitates larger reactor volumes and increased raw material consumption to achieve the same final output, creating a bottleneck for commercial scale-up of complex pharmaceutical intermediates. Additionally, conventional routes frequently require the protection and subsequent deprotection of amine functionalities, introducing extra unit operations that generate additional waste streams and prolong the manufacturing cycle time. These inefficiencies compound the risk of impurity carryover, challenging quality control teams to maintain stringent purity specifications required for regulatory approval.
The Novel Approach
In stark contrast, the methodology presented in CN101657437A introduces a streamlined pathway that capitalizes on the direct reactivity of diglycollic acid derivatives with chiral piperazine intermediates. By employing optically pure starting materials like R-(-)-4-chlorobenzhydrylamine, the process inherently avoids the need for late-stage resolution, thereby preserving the stereochemical integrity of the molecule throughout the synthesis. This approach significantly simplifies the reaction scheme, eliminating the need for protecting groups on the nitrogen atoms which were previously a source of yield loss and operational delay. The use of specific amidation conditions followed by selective reduction allows for a more direct construction of the target molecule, enhancing the overall atom economy of the process. This technological shift represents a pivotal improvement for reducing lead time for high-purity API intermediates, enabling manufacturers to respond more agilely to market demands.
Mechanistic Insights into Diglycollic Acid Mediated Amidation
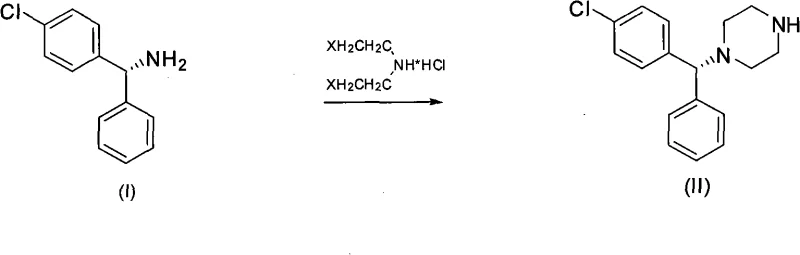
The core of this novel synthesis revolves around the precise interaction between the chiral piperazine intermediate and activated diglycollic acid species, a reaction that demands careful control of stoichiometry and solvent environment to maximize efficiency. The patent details the use of diglycollic anhydride or halogenated derivatives which react with the secondary amine of the piperazine ring to form a stable amide linkage, a critical structural motif in the Levocetirizine backbone. This amidation step is typically conducted in aprotic polar solvents such as acetonitrile or dimethylformamide, often in the presence of phase transfer catalysts like tetrabutylammonium bromide to facilitate the reaction kinetics in heterogeneous mixtures. The mechanistic advantage here is the high regioselectivity of the acylation, which minimizes the formation of bis-acylated byproducts that are difficult to separate downstream. Furthermore, the reaction conditions are mild enough to prevent racemization at the chiral center, ensuring that the optical purity established in the starting material is faithfully transmitted to the final active pharmaceutical ingredient.
Following the formation of the amide intermediate, the process employs a sophisticated selective reduction strategy to convert the carbonyl functionality into the required methylene ether linkage without affecting other sensitive groups. Reagents such as borane-tetrahydrofuran complexes or sodium borohydride in the presence of carboxylic acids are utilized to achieve this transformation with high chemoselectivity. This step is crucial because it avoids the harsh conditions associated with traditional hydride reductions that might compromise the integrity of the chlorophenyl moiety or the piperazine ring. The subsequent hydrolysis of any remaining ester or nitrile groups is performed under controlled pH conditions, typically between 4.2 and 4.8 during extraction, to ensure optimal recovery of the free acid before salt formation. This meticulous control over reaction parameters underscores the robustness of the method, making it highly suitable for industrial application where reproducibility is key.
How to Synthesize Levocetirizine Efficiently
The practical implementation of this synthesis route involves a series of well-defined unit operations that begin with the preparation of the chiral piperazine precursor and culminate in the crystallization of the final dihydrochloride salt. Operators must adhere to strict temperature profiles, particularly during the amidation phase where temperatures range from 60°C to the boiling point of the solvent to ensure complete conversion. The workup procedure involves precise pH adjustments during extraction phases to separate the product from inorganic salts and unreacted starting materials, a step that is critical for achieving the high HPLC purity levels reported in the patent examples. Detailed standardized synthesis steps are essential for maintaining batch-to-batch consistency and ensuring that the final product meets all pharmacopeial standards for impurity limits.
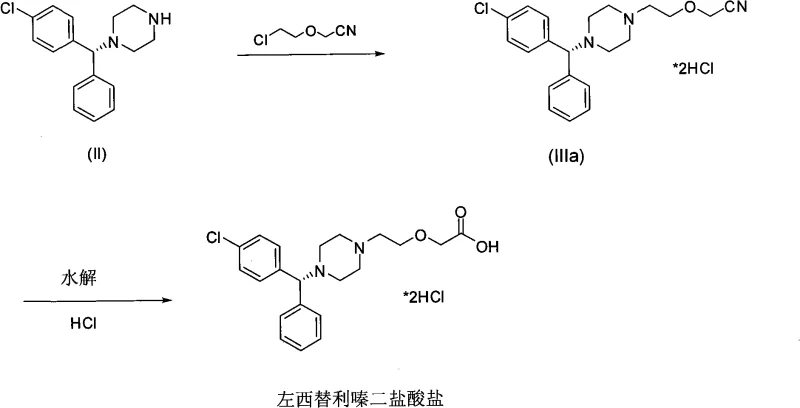
- Prepare the chiral intermediate (-)-1-[(4-chlorophenyl)phenylmethyl]piperazine by reacting R-(-)-4-chlorobenzhydrylamine with bis(2-haloethyl)amine hydrochloride in the presence of organic bases.
- React the chiral piperazine intermediate with diglycolic anhydride or its derivatives in aprotic solvents like acetonitrile to form the amide intermediate.
- Perform selective reduction of the amide intermediate using borane complexes or sodium borohydride systems, followed by hydrolysis and salt formation to obtain Levocetirizine dihydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers substantial benefits that resonate deeply with procurement managers and supply chain heads focused on efficiency and reliability. The elimination of low-yield resolution steps and protection-deprotection sequences translates directly into a more streamlined manufacturing process, which inherently lowers the cost of goods sold by reducing raw material waste and energy consumption. This efficiency gain is particularly significant when scaling from pilot batches to multi-ton commercial production, where even marginal improvements in yield can result in significant financial savings. Moreover, the use of common industrial solvents like acetonitrile and toluene ensures that the supply chain for raw materials is robust and less susceptible to geopolitical disruptions or niche vendor bottlenecks.
- Cost Reduction in Manufacturing: The process design inherently reduces manufacturing costs by removing the need for expensive chiral resolving agents and the associated loss of material during resolution. By starting with optically pure amines and maintaining chirality throughout, the overall yield is significantly improved compared to racemic routes, leading to substantial cost savings per kilogram of active ingredient produced. Additionally, the simplified workup procedures reduce the consumption of auxiliary chemicals and solvents, further driving down the operational expenditure required for production.
- Enhanced Supply Chain Reliability: Utilizing readily available reagents such as diglycolic anhydride and common organic bases ensures a stable supply chain that is not dependent on specialized or custom-synthesized building blocks. This accessibility reduces the risk of supply interruptions and allows for more flexible sourcing strategies, enabling manufacturers to secure better pricing and delivery terms from multiple vendors. The robustness of the chemical process also means that production schedules are more predictable, facilitating better inventory management and planning for downstream formulation teams.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing reaction conditions that are easily transferable from laboratory glassware to large-scale stainless steel reactors without significant re-optimization. The reduction in waste generation, due to fewer synthetic steps and higher atom economy, aligns well with modern environmental regulations and sustainability goals, minimizing the burden on waste treatment facilities. This environmental compliance not only mitigates regulatory risk but also enhances the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Levocetirizine synthesis technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the operational feasibility and quality advantages of the method for potential partners and licensees. Understanding these details is crucial for making informed decisions about technology adoption and supply partnerships.
Q: How does this novel method improve optical purity compared to traditional resolution?
A: The patented method utilizes optically pure starting materials like R-(-)-4-chlorobenzhydrylamine, avoiding the low-yield resolution steps (often <13% yield) associated with racemic splitting. This ensures the optical purity is maintained throughout the synthesis without racemization.
Q: What are the key advantages for large-scale commercial production?
A: The process employs readily available solvents such as acetonitrile and toluene, and avoids complex protection-deprotection sequences required in older methods. This simplifies the workflow, reduces waste generation, and enhances scalability for metric-ton production.
Q: Can the intermediate stability be guaranteed during storage and transport?
A: Yes, the patent describes specific crystalline forms and solvent systems (e.g., acetone slurry) that enhance the stability and purity of intermediates like the acetonitrile dihydrochloride salt, ensuring robustness in the supply chain.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levocetirizine Supplier
At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate complex synthetic pathways like the one described in CN101657437A into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from process development to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor every critical quality attribute of the intermediates and final API. Our commitment to quality assurance means that every batch delivered meets the highest international standards, providing peace of mind to our global partners.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability as your trusted partner in the supply of high-quality pharmaceutical intermediates.
