Advanced Palladium-Catalyzed Synthesis of Phenanthridinone Derivatives for OLED Applications
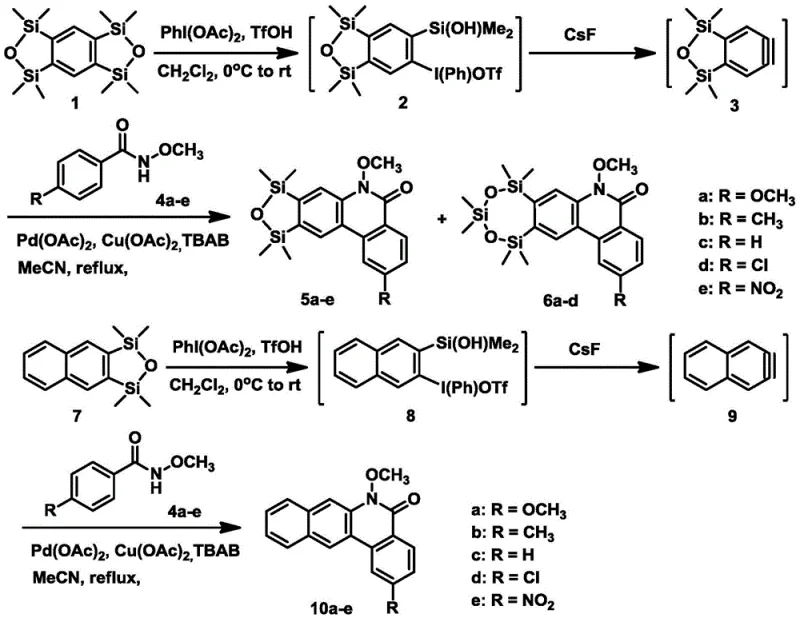
The landscape of organic optoelectronic materials is constantly evolving, driven by the demand for higher efficiency and stability in display technologies. A significant breakthrough in this domain is detailed in patent CN107082761B, which discloses a novel synthetic methodology for constructing phenanthridinone and benzophenanthridinone scaffolds. These fused heterocyclic compounds are not merely academic curiosities; they possess intrinsic photoelectric properties that position them as high-potential candidates for next-generation organic light-emitting materials. The patent outlines a sophisticated palladium-catalyzed coupling strategy that leverages benzyne and naphthyne intermediates generated in situ from stable silyl precursors. This approach represents a paradigm shift from traditional, often hazardous, cyclization methods, offering a streamlined pathway to complex heterocycles that are essential for advanced electronic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenanthridinone cores has relied heavily on the cyclization of biphenylcarbonyl nitro compounds or the Beckmann/Schmidt rearrangement of fluorenone derivatives. These classical routes are fraught with significant operational challenges that hinder their viability for modern, large-scale manufacturing. The reliance on nitro-compounds introduces severe safety hazards due to their potential explosivity and toxicity, necessitating expensive containment infrastructure and rigorous waste management protocols. Furthermore, the reaction conditions required for these traditional cyclizations are often extremely harsh, involving strong acids or high temperatures that can degrade sensitive functional groups, leading to complex impurity profiles that are difficult to purify. The atom economy of these processes is frequently poor, generating substantial amounts of stoichiometric byproducts that increase the environmental footprint and drive up the cost of goods sold (COGS) for the final active pharmaceutical or electronic ingredient.
The Novel Approach
In stark contrast, the methodology presented in the patent utilizes a transition metal-catalyzed C-H/N-H activation strategy mediated by reactive aryne intermediates. By employing benzo-bis(oxydisilocene) or oxydisilenaphthalene as precursors, the process generates benzyne or naphthyne species under remarkably mild conditions. This allows for the simultaneous construction of C-C and C-N bonds in a single pot, drastically reducing the number of synthetic steps required. The use of palladium acetate and copper acetate as co-catalysts, along with cesium fluoride as a fluoride source, enables the reaction to proceed efficiently in acetonitrile under reflux. This modular approach not only enhances the structural diversity accessible to chemists—allowing for easy variation of the R group (methoxy, methyl, chloro, nitro)—but also significantly improves the overall process safety and sustainability profile.
Mechanistic Insights into Pd-Catalyzed Aryne Coupling
The core of this innovation lies in the elegant generation and trapping of the aryne intermediate. The process begins with the activation of the silyl precursor by a hypervalent iodine species, formed in situ from iodobenzene diacetate and trifluoromethanesulfonic acid. Upon treatment with cesium fluoride, the silicon-oxygen bond is cleaved, triggering the elimination of the triflate group to generate the highly reactive benzyne or naphthyne intermediate. This transient species is immediately intercepted by the N-methoxy-4-substituted benzamide derivative. The palladium catalyst plays a pivotal role here, likely facilitating the coordination of the amide nitrogen and the subsequent insertion into the aryne triple bond. This is followed by a C-H activation step that closes the ring to form the phenanthridinone skeleton. The presence of copper acetate and TBAB (tetrabutylammonium bromide) further stabilizes the catalytic cycle and enhances the solubility of the inorganic salts, ensuring a homogeneous reaction environment that maximizes conversion rates.
From an impurity control perspective, this mechanism offers distinct advantages. Because the reaction proceeds at relatively low thermal stress compared to traditional pyrolysis or strong acid treatments, the formation of thermal degradation products is minimized. The specificity of the palladium-catalyzed insertion ensures that the C-N bond forms regioselectively, reducing the occurrence of isomeric byproducts that often plague electrophilic aromatic substitution reactions. Furthermore, the use of N-methoxy amides serves as a traceless directing group that facilitates the cyclization and can be easily managed during the workup phase. This high level of selectivity translates directly to higher crude purity, reducing the burden on downstream purification processes such as column chromatography or recrystallization, which are often the bottlenecks in the production of high-purity electronic chemicals.
How to Synthesize Phenanthridinone Derivatives Efficiently
The synthesis protocol described in the patent provides a robust framework for producing these valuable heterocycles. The procedure involves a two-stage sequence: first, the preparation of the hypervalent iodine-silyl intermediate at low temperature (0°C to room temperature) to ensure stability, and second, the coupling reaction with the benzamide substrate under reflux conditions. This separation of steps allows for precise control over the generation of the reactive aryne species, preventing premature decomposition or polymerization. For detailed operational parameters, stoichiometry, and purification techniques, please refer to the standardized synthesis guide below.
- Generate the hypervalent iodine species by reacting iodobenzene diacetate with trifluoromethanesulfonic acid in dichloromethane at 0°C, then add the silyl precursor to form the triflic acid iodide intermediate.
- Prepare the reaction mixture by combining the intermediate with N-methoxy-4-substituted benzamide, cesium fluoride, palladium acetate, copper acetate, and TBAB in acetonitrile.
- Heat the mixture under reflux for 2 hours to facilitate the in situ generation of benzyne/naphthyne and subsequent cyclization, followed by purification to isolate the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling economic and logistical benefits. The shift away from hazardous nitro-starting materials and harsh cyclization conditions fundamentally alters the risk profile of the manufacturing process. By utilizing stable, commercially available silyl precursors and standard transition metal catalysts, the supply chain becomes more resilient and less susceptible to regulatory disruptions associated with controlled or dangerous substances. The modularity of the reaction also means that a single set of reaction conditions can be used to produce a library of derivatives simply by changing the benzamide input, allowing for agile response to market demands for specific OLED material variants without retooling the entire production line.
- Cost Reduction in Manufacturing: The elimination of multi-step sequences and the reduction in purification complexity lead to substantial cost savings. Traditional methods often require extensive downstream processing to remove toxic byproducts and isomers; this new route's high selectivity minimizes waste generation and solvent consumption. Additionally, the catalysts used (Pd and Cu) are well-established in the industry, allowing for potential recovery and recycling strategies that further lower the raw material costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: The precursors, such as benzo-bis(oxydisilocene), are synthetically accessible and stable, reducing the risk of supply interruptions common with unstable intermediates. The mild reaction conditions (reflux in MeCN) are compatible with standard glass-lined or stainless steel reactors found in most fine chemical facilities, removing the need for specialized high-pressure or corrosion-resistant equipment. This compatibility ensures that production can be scaled rapidly across multiple manufacturing sites without significant capital expenditure.
- Scalability and Environmental Compliance: The process aligns with green chemistry principles by improving atom economy and reducing the use of hazardous reagents. The waste stream is significantly cleaner, primarily consisting of benign silanol byproducts and recoverable salts, which simplifies wastewater treatment and lowers environmental compliance costs. This 'greener' profile is increasingly critical for suppliers aiming to meet the stringent sustainability criteria of major multinational electronics and pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the feasibility of this synthesis route for your specific application needs.
Q: What are the key advantages of using silyl-aryne precursors over traditional nitro-compound cyclization?
A: The silyl-aryne approach described in patent CN107082761B operates under significantly milder conditions (reflux in MeCN vs. harsh acidic cyclization) and avoids the use of unstable or hazardous nitro-intermediates, leading to improved safety profiles and easier impurity control.
Q: Can this synthesis method be scaled for commercial production of OLED materials?
A: Yes, the method utilizes robust catalysts (Pd/Cu) and stable silyl precursors with yields ranging from 48% to 83%. The absence of extreme temperatures or pressures makes it highly suitable for commercial scale-up in electronic chemical manufacturing.
Q: What specific optoelectronic properties do these derivatives exhibit?
A: The synthesized phenanthridinone and benzophenanthridinone derivatives demonstrate distinct absorption and emission spectra in the UV-Vis range, with certain compounds exhibiting high quantum yields, making them promising candidates for organic light-emitting diode (OLED) applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenanthridinone Supplier
As the global demand for high-performance organic light-emitting materials continues to surge, securing a reliable supply chain for complex heterocyclic intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN107082761B to deliver superior quality products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of phenanthridinone or benzophenanthridinone derivative meets the exacting standards required for electronic applications.
We invite you to collaborate with us to optimize your material sourcing strategy. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative manufacturing capabilities can drive value and efficiency in your supply chain.
