Advanced Manufacturing of High-Purity Tamsulosin Hydrochloride: A Technical Breakthrough for Global Supply Chains
The pharmaceutical landscape for urological treatments continues to demand higher standards of chemical purity and manufacturing efficiency, particularly for alpha-1 adrenoceptor antagonists like Tamsulosin. Patent CN1902166A introduces a transformative methodology for the preparation of R-5-(2-(2-(2-ethoxyphenoxyethylamino)propyl)-2-methoxybenzenesulphonamide hydrochloride, commonly known as Tamsulosin Hydrochloride. This intellectual property discloses a robust synthetic route that circumvents the traditional reliance on expensive chiral excesses and toxic solvents, instead leveraging a stoichiometric excess of an achiral reagent combined with a sophisticated thermal crystallization protocol. For global procurement leaders and R&D directors, this represents a pivotal shift towards more sustainable and cost-effective API intermediate manufacturing. The core innovation lies in the ability to achieve optical purity exceeding 99% enantiomeric excess early in the synthesis, thereby eliminating the need for complex downstream chiral separations that typically erode profit margins and extend lead times.
Furthermore, the patent details a purification strategy capable of elevating crude product purity from approximately 86% to greater than 99.8% through merely two crystallization steps. This level of efficiency is critical for a reliable pharmaceutical intermediates supplier aiming to secure long-term contracts with multinational generic drug manufacturers. By addressing the twin challenges of raw material cost and environmental compliance simultaneously, this technology offers a compelling value proposition for the commercial scale-up of complex pharmaceutical intermediates. The following analysis dissects the technical nuances of this process, highlighting its superiority over legacy methods and its potential to redefine supply chain dynamics in the urology therapeutic sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Tamsulosin, such as those described in EP 380,144, were fundamentally constrained by their reliance on the chiral amine starting material to serve a dual purpose: as a reactant and as a base. This necessitated using a molar excess of the optically active (R)-5-(2-amino-1-propyl)-2-methoxybenzenesulfonamide, which is significantly more expensive than its achiral counterparts due to the complexity of its own synthesis and resolution. Consequently, a substantial portion of high-value chiral material was consumed merely to neutralize the hydrobromic acid byproduct, leading to poor atom economy and inflated production costs. Moreover, these conventional routes often inevitably generated significant quantities of by-products and impurities, mandating purification via column chromatography—a technique that is notoriously difficult to scale industrially and poses severe bottlenecks for commercial production volumes.
Additionally, alternative methods disclosed in documents like WO 03/35608 attempted to mitigate reagent excess issues but introduced new liabilities by employing ecologically unacceptable and costly solvents such as dialkylamides, N-methylpyrrolidone (NMP), and sulfolane. These solvents not only increase the direct cost of goods sold but also impose heavy burdens on waste treatment facilities and regulatory compliance teams due to their toxicity and persistence. The combination of expensive chiral waste, difficult purification scaling, and hazardous solvent usage created a fragile supply chain vulnerable to raw material price volatility and regulatory scrutiny. For a procurement manager, these factors translate into unpredictable pricing and potential supply disruptions, making the optimization of this synthetic route a strategic imperative for any organization seeking cost reduction in pharmaceutical manufacturing.
The Novel Approach
The methodology presented in CN1902166A fundamentally inverts the stoichiometric logic of the alkylation step. Instead of wasting expensive chiral amine, the process utilizes a molar excess of the achiral reagent, 1-(2-bromoethoxy)-2-ethoxybenzene (Compound 3), typically in a ratio ranging from 1.5:1 to 2:1 relative to the chiral amine (Compound 2). Remarkably, this reaction proceeds efficiently in lower alkyl alcohols, preferably methanol, without the addition of any external base. The excess achiral bromo-ether effectively drives the reaction equilibrium toward the formation of Tamsulosin while acting as a sacrificial reagent that is far cheaper and easier to remove than the chiral amine. This strategic shift eliminates the need for column chromatography entirely, replacing it with a scalable precipitation and filtration workflow that is inherently suitable for multi-ton production campaigns.
Complementing this synthetic improvement is a novel purification regime based on thermal recrystallization using binary solvent systems. The patent demonstrates that specific mixtures of methanol and ethanol, particularly at a 1:1 ratio, possess unique solubility characteristics that allow for the selective exclusion of over-alkylated impurities. While conventional wisdom might suggest that excess alkylating agent would lead to rampant di-alkylation, this process surprisingly limits the formation of N,N-dialkylated byproducts to less than 2% in the crude stage. Through the optimized crystallization protocol, these trace impurities are reduced to levels below 0.1% w/w, yielding a final product of exceptional chemical and optical purity. This approach not only simplifies the manufacturing process but also aligns perfectly with green chemistry principles by utilizing benign, renewable alcohol solvents instead of chlorinated or amide-based alternatives.
Mechanistic Insights into Nucleophilic Substitution and Impurity Control
The core chemical transformation in this synthesis is a nucleophilic substitution where the primary amine of the chiral sulfonamide attacks the terminal carbon of the bromo-ether side chain. In the absence of an added base, the reaction mechanism relies on the inherent nucleophilicity of the amine in the alcoholic solvent. The use of methanol facilitates the ionization of the bromide leaving group while stabilizing the transition state. Crucially, the stoichiometry is tuned to ensure that the concentration of the electrophile (bromo-ether) remains sufficiently high to drive the mono-alkylation to completion, yet controlled enough to minimize the kinetic probability of a second alkylation event on the newly formed secondary amine or the sulfonamide nitrogen. The patent data indicates that a ratio of approximately 1:1.75 is the sweet spot, balancing reaction rate with impurity profile.
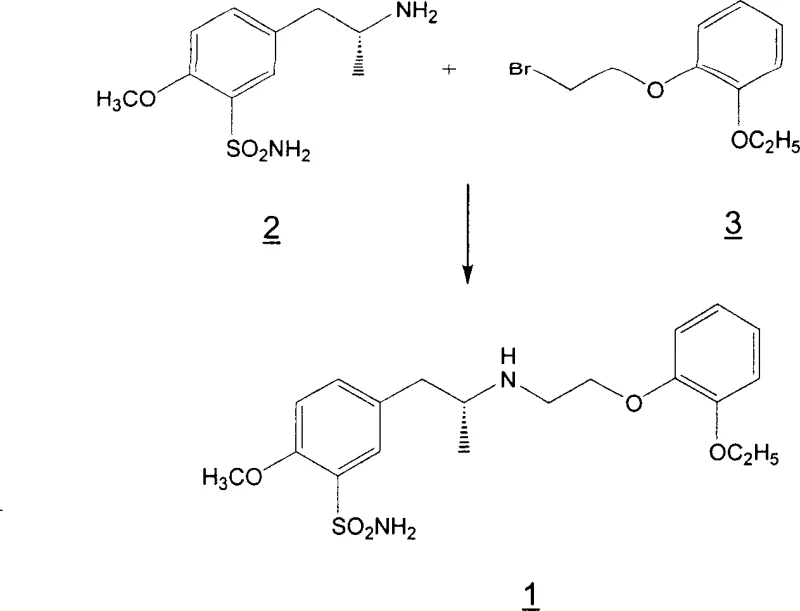
Understanding the impurity profile is vital for R&D directors focused on regulatory filing stability. The primary impurities of concern are the over-alkylated species: the N-alkylated sulfonamide derivative (Impurity 5) and the N,N-dialkylated amine derivative (Impurity 4). As illustrated in the structural diagrams, these molecules possess slightly different polarities and solubility profiles compared to the target Tamsulosin molecule. The brilliance of the disclosed purification method lies in exploiting these subtle physicochemical differences. By heating the crude product in a methanol/ethanol mixture and subsequently cooling it, the crystal lattice of the Tamsulosin hydrochloride forms with high selectivity, excluding the structurally similar but distinct over-alkylated contaminants. This thermal recrystallization acts as a highly efficient filter at the molecular level, ensuring that the final API meets stringent pharmacopeial standards without the need for preparative HPLC or extensive chromatographic polishing.
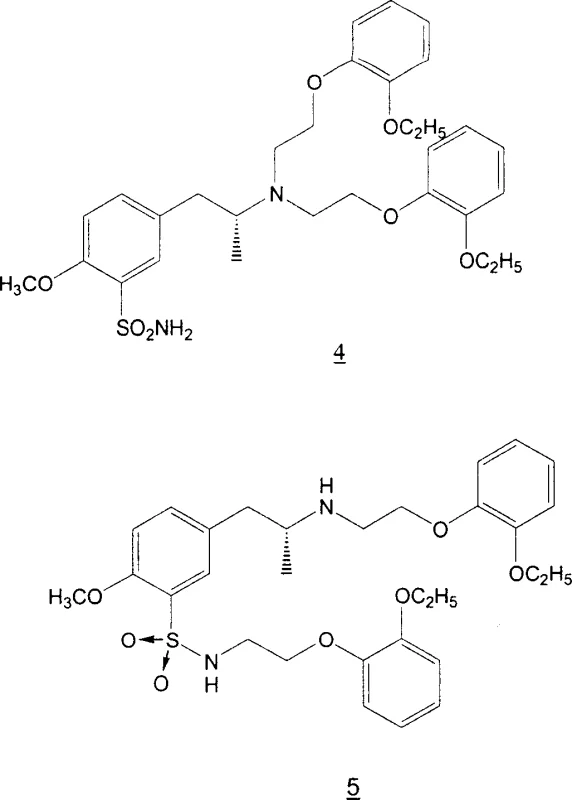
Furthermore, the process demonstrates remarkable robustness against variations in starting material quality. Even when the bromo-ether reagent contains up to 8% of the dimeric impurity 1,2-bis(2-ethoxyphenoxy)ethane, the final product contains less than 0.2% of this contaminant. This indicates that the crystallization steps effectively purge not only reaction byproducts but also carry-over impurities from upstream raw materials. For a technical team, this implies a wider operating window and reduced sensitivity to supplier fluctuations, enhancing the overall reliability of the manufacturing process. The ability to consistently produce material with less than 0.1% total over-alkylated products after just two crystallizations validates the scalability of this route for commercial deployment.
How to Synthesize Tamsulosin Hydrochloride Efficiently
The operational execution of this synthesis requires precise control over reaction stoichiometry and crystallization parameters to maximize yield and purity. The process begins with the reflux of the chiral amine and the achiral bromo-ether in methanol for an extended period, typically around 43 to 45 hours, to ensure complete conversion. Following the reaction, the solvent is removed, and the residue is treated with aqueous base and extracted into an organic phase before conversion to the hydrochloride salt using ethanolic HCl. The resulting crude solid is then subjected to the critical purification sequence. Detailed standard operating procedures regarding temperature ramps, solvent addition rates, and seeding strategies are essential for reproducibility. The standardized synthesis steps outlined below provide a roadmap for implementing this high-efficiency route in a GMP environment.
- React (R)-5-(2-amino-1-propyl)-2-methoxybenzenesulfonamide with a 1.5: 1 to 2:1 molar excess of 1-(2-bromoethoxy)-2-ethoxybenzene in methanol under reflux without added base.
- Isolate the crude hydrochloride salt by treating the reaction mixture with ethanolic HCl, followed by filtration and washing to remove bulk impurities.
- Purify the crude product via two-stage thermal recrystallization using optimized methanol/ethanol solvent mixtures to achieve >99.8% chemical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic advantages that extend beyond simple unit cost savings. By eliminating the requirement for excess chiral starting material, the bill of materials is significantly optimized, as the expensive enantiomer is no longer consumed as a stoichiometric base. This structural change in the cost drivers allows for a more competitive pricing model that can withstand market fluctuations in chiral building blocks. Additionally, the replacement of hazardous solvents like NMP with common alcohols reduces the regulatory burden associated with solvent residuals and waste disposal, leading to substantial cost savings in environmental compliance and waste treatment operations. These factors collectively enhance the margin profile of the final API, making it a more attractive asset for generic drug portfolios.
- Cost Reduction in Manufacturing: The inversion of reagent stoichiometry is the primary driver for cost efficiency. By using an excess of the inexpensive achiral bromo-ether instead of the costly chiral amine, the direct material cost per kilogram of product is drastically reduced. Furthermore, the elimination of column chromatography removes a major bottleneck that typically requires expensive silica gel, large volumes of solvent, and significant labor hours. The switch to thermal crystallization utilizes standard stainless steel reactors and filtration equipment, lowering capital expenditure requirements and reducing the energy intensity associated with solvent recovery in chromatographic processes. This streamlined workflow ensures that cost reduction in pharmaceutical manufacturing is achieved through fundamental process design rather than temporary supplier negotiations.
- Enhanced Supply Chain Reliability: The robustness of the purification process directly impacts supply continuity. Because the thermal recrystallization method is highly effective at removing impurities even from lower-quality crude inputs, the manufacturing process is less sensitive to variations in raw material specifications. This tolerance allows procurement teams to source starting materials from a broader range of qualified vendors without compromising final product quality. Moreover, the use of commodity solvents like methanol and ethanol ensures that solvent supply is never a limiting factor, unlike specialized solvents which may face geopolitical or logistical constraints. This resilience builds a more dependable supply chain capable of meeting the rigorous delivery schedules of global pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling a process that relies on chromatography is notoriously difficult and risky, often requiring linear increases in equipment footprint and waste generation. In contrast, this crystallization-based approach scales naturally with reactor volume, facilitating the commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production with minimal re-engineering. The environmental footprint is also significantly reduced; alcohol solvents are biodegradable and easier to incinerate or recover compared to amides and sulfoxides. This alignment with green chemistry principles not only satisfies corporate sustainability goals but also future-proofs the manufacturing site against tightening environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on how this technology resolves historical pain points in Tamsulosin production. Understanding these specifics is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the value proposition of this manufacturing method.
Q: How does this process improve upon conventional methods regarding chiral reagent usage?
A: Unlike prior art (e.g., EP 380,144) which required a molar excess of the expensive chiral amine to act as a base, this novel method utilizes an excess of the inexpensive achiral bromo-ether reagent. This inversion of stoichiometry drastically reduces raw material costs while maintaining high optical purity.
Q: What is the strategy for controlling over-alkylated impurities?
A: The process controls over-alkylation by optimizing the reagent ratio to approximately 1:1.75. Furthermore, the specific thermal recrystallization protocol using methanol/ethanol mixtures effectively separates polar over-alkylated byproducts (Impurities 4 and 5) from the target API, reducing them to below 0.1% w/w.
Q: Why is the methanol/ethanol solvent system preferred for purification?
A: The binary solvent system allows for fine-tuning of polarity. A 1:1 ratio of methanol to ethanol has been found to uniformly remove both non-polar and polar impurities while maximizing product recovery yield, avoiding the need for ecologically hazardous solvents like NMP or sulfolane used in older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tamsulosin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1902166A are fully realized in your supply chain. We operate stringent purity specifications and maintain rigorous QC labs equipped to detect impurities at trace levels, guaranteeing that every batch of Tamsulosin Hydrochloride meets the highest global pharmacopeial standards. Our commitment to quality assurance means that we do not just supply chemicals; we deliver validated solutions that de-risk your regulatory filings and protect your brand reputation.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your sourcing strategy. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic impact of switching to this more efficient manufacturing method. We are prepared to provide specific COA data from our pilot batches and comprehensive route feasibility assessments to demonstrate our capability to serve as your long-term partner. Let us collaborate to optimize your supply chain for high-purity pharmaceutical intermediates, driving value through innovation and operational excellence.
