Advanced Manufacturing of Tamsulosin Hydrochloride via Safe Reductive Amination
Introduction to Next-Generation Tamsulosin Synthesis
The global demand for benign prostatic hyperplasia (BPH) treatments continues to drive the need for robust, scalable, and safe manufacturing processes for Tamsulosin Hydrochloride. Patent CN111320559A introduces a transformative preparation method that fundamentally alters the synthetic landscape of this critical alpha-blocker. By shifting away from traditional high-pressure hydrogenation techniques in the initial stages, this technology addresses long-standing safety concerns and impurity profiles associated with heavy metal catalysts. For pharmaceutical manufacturers, this represents a pivotal opportunity to enhance process safety while simultaneously improving the economic viability of production. The disclosed method leverages mild chemical reduction strategies that are inherently more controllable and less hazardous than gas-phase hydrogenation, aligning perfectly with modern green chemistry principles and stringent regulatory requirements for residual metals in active pharmaceutical ingredients.
This technical insight report analyzes the specific advantages of this novel route, focusing on its applicability for large-scale commercial production. The transition from hazardous hydrogen gas to solid-state reducing agents not only mitigates operational risks but also simplifies the engineering controls required for manufacturing facilities. As a reliable pharmaceutical intermediates supplier, understanding these mechanistic shifts is crucial for evaluating supply chain resilience. The patent details a comprehensive pathway that maintains high stereochemical fidelity while drastically reducing the complexity of the reaction infrastructure, making it an ideal candidate for cost reduction in API manufacturing where safety and purity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tamsulosin intermediates has relied heavily on the methodology described in earlier patents such as EP0380144B. This conventional approach necessitates the use of molecular hydrogen gas in the presence of nickel or platinum catalysts to effect the reductive amination of the sulfonamide ketone precursor. 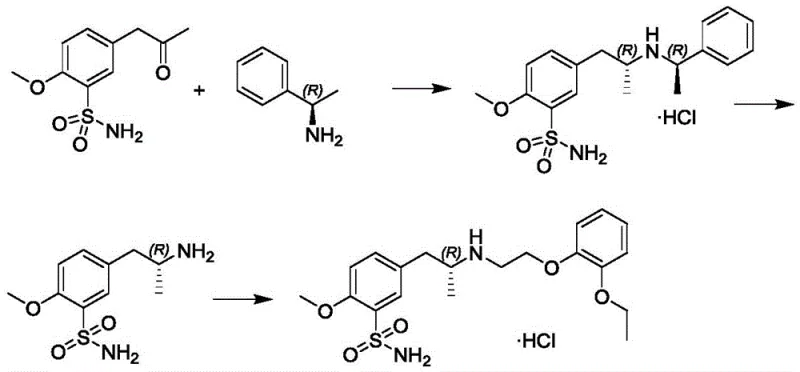 The reliance on high-pressure hydrogenation introduces severe safety liabilities, including the risk of explosion and the requirement for specialized, expensive autoclave reactors capable of withstanding significant pressure. Furthermore, the use of nickel and platinum catalysts creates a persistent challenge regarding heavy metal residues. Removing trace amounts of these toxic metals to meet pharmacopeial standards often requires additional purification steps, such as scavenging or extensive recrystallization, which inevitably erodes overall yield and increases production costs. The harsh conditions required for these hydrogenation reactions can also lead to side reactions, complicating the impurity profile and necessitating rigorous quality control measures.
The reliance on high-pressure hydrogenation introduces severe safety liabilities, including the risk of explosion and the requirement for specialized, expensive autoclave reactors capable of withstanding significant pressure. Furthermore, the use of nickel and platinum catalysts creates a persistent challenge regarding heavy metal residues. Removing trace amounts of these toxic metals to meet pharmacopeial standards often requires additional purification steps, such as scavenging or extensive recrystallization, which inevitably erodes overall yield and increases production costs. The harsh conditions required for these hydrogenation reactions can also lead to side reactions, complicating the impurity profile and necessitating rigorous quality control measures.
The Novel Approach
In stark contrast, the method disclosed in CN111320559A replaces the hazardous hydrogenation step with a chemoselective reduction using sodium triacetoxyborohydride (STAB) or similar borohydride derivatives. This shift allows the reductive amination to proceed under mild, atmospheric conditions, typically in solvents like dichloromethane at temperatures ranging from 0°C to 50°C. By eliminating the need for pressurized hydrogen gas, the process inherently becomes safer and more operable in standard glass-lined or stainless steel reactors without the need for high-pressure ratings. The avoidance of nickel and platinum in this critical first step significantly lowers the initial heavy metal load entering the synthetic stream. This novel approach not only enhances operator safety by removing flammable gases but also streamlines the downstream purification process, as the absence of difficult-to-remove nickel residues simplifies the path to high-purity intermediates. The result is a more robust and economically attractive manufacturing process that retains the necessary stereochemistry without the baggage of traditional catalytic hydrogenation.
Mechanistic Insights into Borohydride-Mediated Reductive Amination
The core innovation of this synthesis lies in the mechanism of the initial reductive amination between 5-acetonyl-2-methoxybenzenesulfonamide and (R)-1-phenylethylamine. In the presence of a mild acid source like acetic acid, the ketone and amine condense to form an iminium ion intermediate. Sodium triacetoxyborohydride acts as a hydride donor that selectively reduces this iminium species to the corresponding secondary amine without reducing other sensitive functional groups that might be present. This selectivity is crucial for maintaining the integrity of the sulfonamide moiety. The reaction proceeds with high efficiency, as evidenced by the patent examples which report yields exceeding 70% with excellent chemical purity of 99.7%. The mild acidic conditions facilitate the formation of the iminium ion while preventing the decomposition of the reducing agent, creating an optimal kinetic window for the transformation. This mechanistic pathway ensures that the chiral center introduced by the (R)-1-phenylethylamine is preserved, setting the stage for the subsequent stereospecific transformations required to build the Tamsulosin backbone.
Following the formation of the protected intermediate (Formula IV), the process employs a catalytic transfer hydrogenation strategy for deprotection. 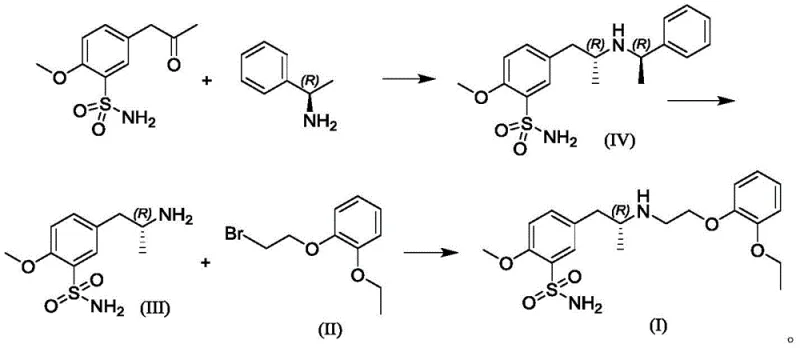 Instead of high-pressure hydrogen, the patent utilizes ammonium formate as a hydrogen donor in the presence of a palladium on carbon catalyst. This transfer hydrogenation generates hydrogen in situ, which is immediately consumed in the hydrogenolysis of the benzyl group. This method effectively cleaves the chiral auxiliary to reveal the primary amine (Formula III) while maintaining the (R)-configuration at the adjacent carbon. The final alkylation step involves the nucleophilic attack of this primary amine on o-ethoxyphenoxyethyl bromide. The use of potassium carbonate as a base and potassium iodide as a catalyst facilitates this SN2 reaction in polar aprotic solvents like acetonitrile. This sequence demonstrates a logical and efficient progression where each step is optimized to minimize side products and maximize the recovery of the desired enantiomer, ensuring a high-purity pharmaceutical intermediate suitable for final salt formation.
Instead of high-pressure hydrogen, the patent utilizes ammonium formate as a hydrogen donor in the presence of a palladium on carbon catalyst. This transfer hydrogenation generates hydrogen in situ, which is immediately consumed in the hydrogenolysis of the benzyl group. This method effectively cleaves the chiral auxiliary to reveal the primary amine (Formula III) while maintaining the (R)-configuration at the adjacent carbon. The final alkylation step involves the nucleophilic attack of this primary amine on o-ethoxyphenoxyethyl bromide. The use of potassium carbonate as a base and potassium iodide as a catalyst facilitates this SN2 reaction in polar aprotic solvents like acetonitrile. This sequence demonstrates a logical and efficient progression where each step is optimized to minimize side products and maximize the recovery of the desired enantiomer, ensuring a high-purity pharmaceutical intermediate suitable for final salt formation.
How to Synthesize Tamsulosin Hydrochloride Efficiently
The synthesis of Tamsulosin Hydrochloride via this novel route involves a carefully orchestrated sequence of reactions designed to maximize safety and yield. The process begins with the reductive amination in dichloromethane, followed by isolation of the intermediate hydrochloride salt. Subsequent deprotection is carried out in ethanol using ammonium formate, a reagent that is easy to handle and store compared to compressed hydrogen cylinders. The final coupling reaction utilizes standard alkylation conditions that are well-understood in process chemistry. For detailed operational parameters, including specific stoichiometric ratios, temperature ramps, and workup procedures, operators should refer to the standardized protocols derived from the patent examples. These protocols ensure reproducibility and consistency across different batches, which is essential for regulatory compliance. The integration of these steps into a cohesive manufacturing workflow allows for the seamless production of high-quality Tamsulosin Hydrochloride with minimal environmental impact.
- Perform reductive amination of 5-acetonyl-2-methoxybenzenesulfonamide with (R)-1-phenylethylamine using sodium triacetoxyborohydride.
- Execute catalytic hydrogenolysis using palladium on carbon and ammonium formate to remove the chiral auxiliary group.
- Conduct nucleophilic substitution with o-ethoxyphenoxyethyl bromide followed by salt formation to yield the final API.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere technical feasibility. The elimination of high-pressure hydrogenation equipment in the first step represents a significant reduction in capital expenditure (CAPEX) for manufacturing facilities. Standard reactors can be utilized instead of costly autoclaves, lowering the barrier to entry for production and increasing the flexibility of existing asset networks. Furthermore, the removal of nickel and platinum catalysts from the initial stage reduces the dependency on volatile precious metal markets and simplifies the supply chain for raw materials. This shift contributes to substantial cost savings by reducing the need for expensive metal scavengers and complex filtration systems required to meet strict residual metal limits. The overall process stability ensures a consistent supply of intermediates, reducing the risk of production delays caused by equipment maintenance or safety incidents associated with high-pressure gas handling.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts (Pt/Ni) and high-pressure hydrogen gas with sodium triacetoxyborohydride significantly lowers raw material costs and operational expenses. The process avoids the need for specialized high-pressure reactors, allowing production in standard multipurpose vessels which reduces energy consumption and maintenance costs. Additionally, the simplified purification steps required to remove heavy metals result in higher overall yields and reduced waste disposal costs, directly impacting the bottom line.
- Enhanced Supply Chain Reliability: By removing the dependency on compressed hydrogen gas and specialized catalytic hydrogenation services, the supply chain becomes more resilient and less prone to logistical disruptions. The reagents used, such as sodium triacetoxyborohydride and ammonium formate, are stable, widely available commodities that do not require hazardous material transport protocols associated with flammable gases. This stability ensures continuous production capabilities and reduces the lead time for high-purity pharmaceutical intermediates, securing the supply pipeline for downstream API manufacturing.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic nickel residues make this process highly scalable and environmentally friendly. The reduction in heavy metal waste simplifies effluent treatment and aligns with increasingly stringent environmental regulations regarding metal discharge. The ability to scale from kilogram to multi-ton production without changing the fundamental reaction physics ensures that commercial scale-up of complex pharmaceutical intermediates can be achieved rapidly and safely, meeting global demand without compromising on sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is essential for R&D teams evaluating the feasibility of technology transfer and for quality assurance teams assessing the impurity profile of the final product. The answers are derived directly from the technical disclosures and experimental data provided in the patent literature, ensuring accuracy and relevance for decision-makers. These insights provide a clear framework for integrating this advanced manufacturing method into existing production portfolios.
Q: Why is the new synthesis route safer than the conventional method?
A: The conventional method relies on high-pressure hydrogen gas and nickel or platinum catalysts, posing significant explosion risks. The novel route utilizes sodium triacetoxyborohydride under atmospheric pressure, eliminating the need for specialized high-pressure autoclaves and flammable gas handling.
Q: How does this process ensure high chiral purity?
A: Chirality is introduced early using enantiomerically pure (R)-1-phenylethylamine. The mild reductive amination conditions prevent racemization, and the subsequent deprotection steps are optimized to maintain the stereochemical integrity of the propylamine chain.
Q: Does this method eliminate heavy metal residues completely?
A: It significantly reduces heavy metal burden. The critical first step avoids nickel and platinum entirely. While palladium is used in the second deprotection step, it is easily removed via filtration, resulting in a much cleaner impurity profile compared to methods using nickel throughout.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tamsulosin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for high-value pharmaceutical intermediates like Tamsulosin Hydrochloride. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and compliant. We are committed to delivering products with stringent purity specifications, utilizing our rigorous QC labs to monitor every step of the synthesis, from the initial reductive amination to the final salt formation. Our infrastructure is designed to accommodate the specific requirements of this novel route, including the handling of borohydride reagents and the precise control of reaction parameters necessary to maintain chiral integrity.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to request specific COA data and route feasibility assessments to verify the superiority of this non-hydrogenation approach. Together, we can establish a robust supply partnership that prioritizes safety, quality, and economic efficiency in the production of essential urological medications.
