Advanced Asymmetric Synthesis of Tamsulosin Hydrochloride for Commercial Scale-Up
The global demand for benign prostatic hyperplasia (BPH) treatments continues to drive innovation in the synthesis of alpha-1 adrenoceptor inhibitors, specifically tamsulosin hydrochloride. As detailed in patent CN101462987A, a novel asymmetric preparation method has been developed that fundamentally shifts the paradigm from wasteful resolution techniques to efficient chiral induction. This technology leverages the unique stereoelectronic properties of ferrocenyl derivatives to achieve high optical purity without the excessive costs associated with traditional enzymatic or chromatographic separations. For pharmaceutical manufacturers, this represents a critical opportunity to optimize the production of this high-value pharmaceutical intermediate. The structural complexity of tamsulosin hydrochloride, characterized by its sulfonamide and ether linkages, requires precise control over stereochemistry to ensure therapeutic efficacy and safety profiles.

Furthermore, the integration of this asymmetric route addresses significant environmental concerns prevalent in the fine chemical industry. By utilizing a recyclable chiral auxiliary, the process minimizes the generation of hazardous waste streams typically associated with the disposal of unwanted enantiomers. This approach not only aligns with green chemistry principles but also enhances the overall economic viability of the manufacturing process. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for evaluating the long-term sustainability and scalability of the supply chain. The following analysis dissects the technical merits of this invention, providing a roadmap for implementing this superior synthetic strategy in commercial operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tamsulosin hydrochloride has relied heavily on chemical resolution methods, which are inherently inefficient and economically burdensome. Traditional processes involve synthesizing racemic tamsulosin and subsequently separating the desired (R)-enantiomer using chiral acids such as camphorsulfonic acid or tartaric acid. This approach suffers from a theoretical maximum yield of only 50%, as the unwanted (S)-enantiomer is discarded or requires complex recycling procedures that often degrade the material. Moreover, the repeated recrystallization steps necessary to achieve high optical purity result in substantial losses of active pharmaceutical ingredients, driving up the cost of goods sold (COGS). Additionally, alternative asymmetric methods utilizing proline derivatives have demonstrated insufficient optical purity, often failing to meet the stringent regulatory requirements for chiral drugs, with reported enantiomeric excess values as low as 39%.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN101462987A introduces a robust asymmetric synthesis utilizing (R)-ferrocenylethylamine as a chiral building block. This novel approach bypasses the need for post-synthesis resolution by establishing the correct stereochemistry at the outset of the reaction sequence. The ferrocenyl moiety acts as a powerful chiral director, inducing high diastereoselectivity during the critical reduction step, with diastereomeric ratios reaching as high as 95:5. This precision eliminates the waste associated with discarding half of the product batch. Furthermore, the process is designed with a built-in recovery mechanism for the chiral auxiliary, allowing the ferrocenyl group to be cleaved and regenerated for reuse. This closed-loop system drastically reduces the consumption of expensive chiral starting materials, offering a distinct competitive advantage in cost reduction in API manufacturing.
Mechanistic Insights into Ferrocenyl-Mediated Asymmetric Induction
The core of this innovative synthesis lies in the strategic application of organometallic chemistry to control stereochemical outcomes. The process initiates with the condensation of (R)-ferrocenylethylamine and 5-acetonyl-2-methoxybenzenesulfonamide to form a chiral imine intermediate. This imine serves as the substrate for a subsequent reduction reaction, where the bulky ferrocenyl group shields one face of the molecule, directing the hydride attack to the opposite face. Whether utilizing sodium borohydride or catalytic hydrogenation with Raney Nickel or Pd/C, the steric hindrance provided by the ferrocene unit ensures the formation of the desired diastereomer with exceptional fidelity. This step is crucial, as it sets the absolute configuration of the propylamine side chain found in the final drug substance.
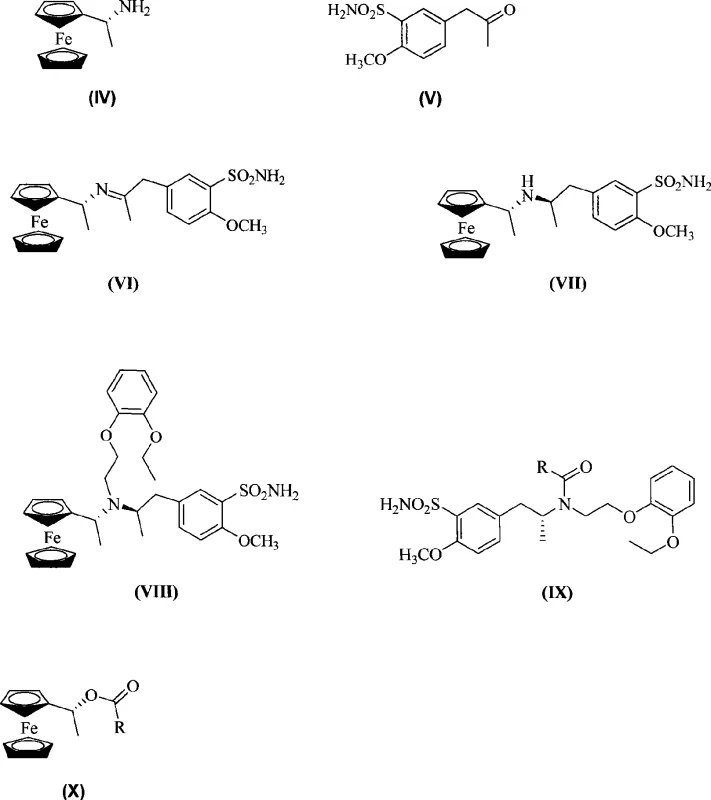
Following the establishment of the chiral center, the intermediate undergoes alkylation with o-2-bromoethoxyphenethyl ether to introduce the phenoxyethyl side chain. The final transformation involves a clever deprotection strategy using acid anhydrides, such as acetic anhydride or trifluoroacetic anhydride. This step cleaves the bond between the nitrogen and the ferrocenyl-ethyl group, releasing the free amine of tamsulosin while converting the auxiliary into ferrocenyl ethanol carboxylate. This byproduct is not waste; it is a valuable precursor that can be ammoniated to regenerate the original (R)-ferrocenylethylamine. This mechanistic elegance ensures that the chiral information is preserved and recycled, minimizing impurity profiles and simplifying the purification workflow. The ability to recover the auxiliary transforms what is typically a stoichiometric expense into a catalytic-like investment, significantly enhancing the atom economy of the overall process.
How to Synthesize Tamsulosin Hydrochloride Efficiently
The implementation of this asymmetric route requires careful attention to reaction parameters to maximize yield and optical purity. The synthesis is divided into distinct stages: condensation, reduction, alkylation, deprotection, and hydrolysis. Each step utilizes common industrial solvents such as methanol, THF, or methyl tert-butyl ether, facilitating easy scale-up. The reduction step, pivotal for stereocontrol, can be tuned by selecting appropriate reducing agents, with NaBH4 offering a balance of cost and effectiveness. The subsequent alkylation requires basic conditions, typically employing potassium carbonate or triethylamine, to facilitate the nucleophilic substitution. Detailed standard operating procedures for each stage, including specific molar ratios and temperature controls, are essential for reproducibility.
- Condense (R)-ferrocenylethylamine with 5-acetonyl-2-methoxybenzenesulfonamide to form the chiral imine intermediate.
- Reduce the imine compound using NaBH4 or catalytic hydrogenation to establish the second chiral center with high diastereoselectivity.
- Perform alkylation with o-2-bromoethoxyphenethyl ether, followed by acid anhydride treatment to cleave the auxiliary and recover the chiral amine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ferrocenyl-mediated synthesis offers tangible benefits beyond mere technical superiority. The primary advantage is the drastic simplification of the supply chain for chiral materials. By eliminating the need for external chiral resolving agents and the associated disposal of unwanted enantiomers, manufacturers can streamline their raw material inventory. The recyclability of the ferrocenyl auxiliary means that the effective cost per kilogram of the chiral inducer decreases significantly over multiple batches, leading to substantial long-term savings. This stability in raw material usage translates directly to more predictable pricing models for the final high-purity pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The elimination of resolution steps removes the inherent 50% yield loss associated with racemic synthesis, effectively doubling the output from the same amount of achiral starting materials. Furthermore, the recovery and reuse of the ferrocenyl auxiliary reduce the dependency on purchasing expensive chiral amines for every batch. The use of standard reducing agents like sodium borohydride instead of specialized enzymatic catalysts further lowers reagent costs. These factors combine to create a highly efficient production model that minimizes waste disposal fees and maximizes resource utilization, driving down the overall cost of production without compromising quality.
- Enhanced Supply Chain Reliability: Relying on commercially available starting materials such as 5-acetonyl-2-methoxybenzenesulfonamide and robust ferrocenyl derivatives ensures a stable supply base. Unlike proprietary biocatalysts or scarce natural chiral pools, these chemical reagents are readily sourced from multiple suppliers, mitigating the risk of supply disruptions. The robustness of the chemical steps, which tolerate mild reaction conditions and standard equipment, reduces the likelihood of batch failures due to sensitive process parameters. This reliability is critical for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, utilizing solvents and reagents that are manageable in large-scale reactors. The avoidance of heavy metal catalysts for chiral induction (relying instead on the stoichiometric but recyclable ferrocenyl group) simplifies the removal of metal impurities, a common bottleneck in API manufacturing. Additionally, the reduced waste generation aligns with increasingly strict environmental regulations regarding solvent discharge and hazardous waste. The ability to recycle the chiral auxiliary internally reduces the carbon footprint of the manufacturing process, supporting corporate sustainability goals and enhancing the marketability of the final product to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the asymmetric synthesis of tamsulosin hydrochloride. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What is the primary advantage of using ferrocenyl ethylamine in tamsulosin synthesis?
A: The use of (R)-ferrocenylethylamine allows for high diastereoselectivity (up to 95:5) during the reduction step, eliminating the need for difficult chiral resolution and significantly reducing raw material waste compared to traditional racemic methods.
Q: Can the chiral auxiliary be recycled in this process?
A: Yes, a key feature of this patent technology is the recovery of the ferrocenyl group as ferrocenyl ethanol carboxylate after the deprotection step, which can be converted back to (R)-ferrocenylethylamine, creating a sustainable cycle.
Q: Is this synthesis method suitable for industrial scale-up?
A: Absolutely. The process utilizes mild reaction conditions, common solvents like methanol and THF, and avoids expensive noble metal catalysts for chiral induction, making it highly viable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tamsulosin Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced asymmetric synthesis technologies like the ferrocenyl-mediated route for tamsulosin hydrochloride. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are seamlessly translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation to verify every batch. Our infrastructure is designed to handle complex chiral intermediates with the utmost care, guaranteeing consistency and reliability for our global partners.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your operational efficiency. Let us be your partner in securing a sustainable and cost-effective supply of high-quality pharmaceutical intermediates.
