Advanced Synthesis of Pemetrexed Disodium Intermediates for Commercial Scale-Up
Introduction to Novel Pemetrexed Intermediate Technology
The pharmaceutical landscape for oncology treatments relies heavily on the efficient production of antifolate agents like pemetrexed disodium. Patent CN101560206A introduces a groundbreaking methodology for synthesizing a critical intermediate, designated as Formula (I), which serves as the precursor to the final active pharmaceutical ingredient. This technology addresses long-standing challenges in the field, specifically targeting the low yields and operational complexities associated with traditional ring-closure reactions. By implementing a strategic sequence of condensation, Heck coupling, and protective group chemistry, this route achieves a ring-closure yield exceeding 85%, a substantial improvement over previous methods. For R&D directors and process chemists, this represents a viable pathway to enhance purity profiles and reduce impurity loads in the final drug substance.
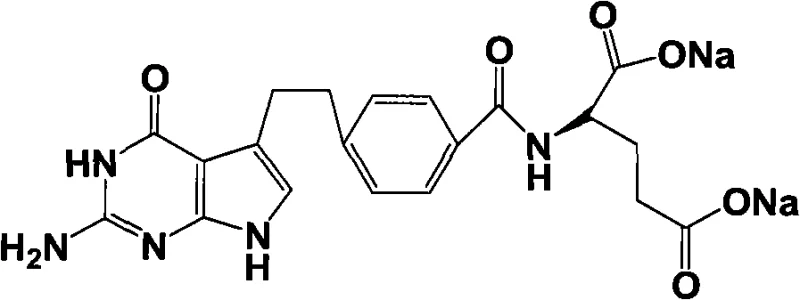
The structural integrity of the final product is paramount, and the intermediate described in this patent ensures that the pyrrolo[2,3-d]pyrimidine core is constructed with high fidelity. The innovation lies not just in the final step, but in the cumulative efficiency of the six-step precursor synthesis. This approach minimizes waste generation and maximizes atom economy, aligning with modern green chemistry principles while maintaining the rigorous quality standards required for injectable oncology medications. As a reliable pemetrexed intermediate supplier, understanding these mechanistic nuances is essential for guaranteeing supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for pemetrexed disodium have historically suffered from inefficient ring-closure steps, often yielding products with significant impurity profiles that require costly purification. Traditional methods frequently struggle with the stability of aldehyde intermediates, which are prone to oxidation, leading to decreased overall recovery rates. In many existing processes, the conversion of the open-chain precursor to the cyclic pyrrolo-pyrimidine system is the bottleneck, limiting the total throughput of manufacturing facilities. Furthermore, the reliance on harsh conditions or expensive catalysts in older pathways often complicates the scale-up process, making it difficult to achieve consistent batch-to-batch reproducibility. These factors collectively drive up the cost of goods sold (COGS) and extend lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in CN101560206A circumvents these issues through a cleverly designed protection strategy. By converting the susceptible aldehyde group into a 1,3-dioxolane derivative early in the sequence, the synthesis prevents unwanted side reactions and oxidation. 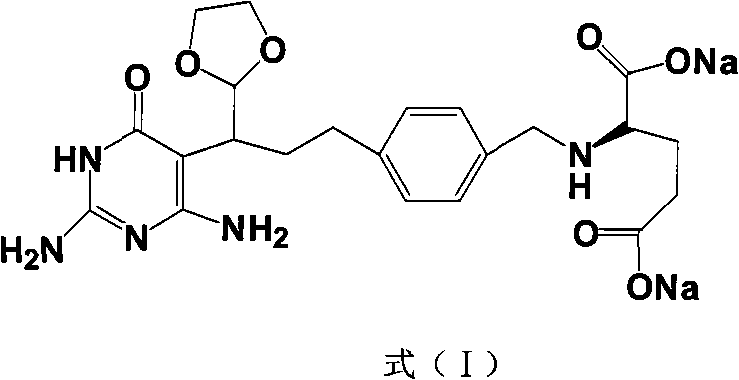 This stabilization allows subsequent reactions, such as the condensation with ethyl cyanoacetate and the final cyclization with guanidine, to proceed with much higher efficiency. The result is a robust process where the critical ring-closure step achieves yields greater than 85%, drastically reducing material loss. This novel approach transforms the manufacturing landscape by offering a route that is not only chemically superior but also economically advantageous for cost reduction in API manufacturing.
This stabilization allows subsequent reactions, such as the condensation with ethyl cyanoacetate and the final cyclization with guanidine, to proceed with much higher efficiency. The result is a robust process where the critical ring-closure step achieves yields greater than 85%, drastically reducing material loss. This novel approach transforms the manufacturing landscape by offering a route that is not only chemically superior but also economically advantageous for cost reduction in API manufacturing.
Mechanistic Insights into the Multi-Step Synthesis
The synthesis begins with the activation of 4-bromobenzoic acid or 4-iodobenzoic acid using carbonyl diimidazole (CDI), followed by condensation with L-glutamate diethyl ester at controlled temperatures of -5 to 0°C. This initial step establishes the amide bond with high stereochemical retention. Subsequently, a palladium-catalyzed Heck reaction introduces the carbon chain necessary for the pyrrole ring formation. This step utilizes Pd(OAc)2 and lithium salts in DMF at 80-85°C, demonstrating the robustness of transition metal catalysis in building complex molecular architectures. The careful control of stoichiometry, with a molar ratio of substrate to 3-butenol between 1:1.05 and 1:1.15, ensures complete conversion while minimizing homocoupling byproducts.
Following the Heck coupling, the process employs 5,5-dibromobarbituric acid (DBBA) and hydrogen bromide for selective bromination at 0-5°C, generating the alpha-bromo aldehyde intermediate. The immediate protection of this aldehyde with ethylene glycol in toluene under reflux conditions is a critical mechanistic pivot point. This acetal formation shields the reactive carbonyl from nucleophilic attack during the subsequent base-mediated cyclization. The final construction of the pyrimidine ring involves reacting the protected intermediate with ethyl cyanoacetate and cuprous iodide, followed by cyclization with guanidine hydrochloride in sodium ethoxide. This sequence elegantly assembles the tricyclic core, ensuring that the amino and oxo functionalities are correctly positioned for the final hydrolysis and salt formation steps.
How to Synthesize Pemetrexed Disodium Intermediate Efficiently
The execution of this synthesis requires precise adherence to the reaction parameters defined in the patent to ensure optimal yield and purity. The process integrates standard unit operations such as extraction, distillation, and crystallization, making it highly adaptable to existing pilot and production plants. Operators must pay close attention to temperature controls during the exothermic condensation and the moisture-sensitive cyclization steps. The detailed standardized synthesis steps见下方的指南.
- Condense 4-bromobenzoic acid with L-glutamate diethyl ester using CDI catalyst at -5 to 0°C to form Compound (1).
- Perform Heck reaction with 3-butenol using Pd(OAc)2 catalyst at 80-85°C, followed by hydrolysis to generate aldehyde Compound (2).
- Execute selective bromination using DBBA and HBr at 0-5°C to obtain Compound (3), then protect the aldehyde with ethylene glycol.
- React the protected intermediate with ethyl cyanoacetate and CuI at 80-85°C, followed by cyclization with guanidine hydrochloride to yield Formula (I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits beyond mere chemical elegance. The primary advantage is the drastic simplification of the purification train; higher yields in the key ring-closure step mean less starting material is wasted, directly translating to significant cost savings. The use of readily available commodity chemicals like 4-bromobenzoic acid, ethylene glycol, and guanidine hydrochloride ensures that the supply chain is not dependent on exotic or single-source reagents. This availability enhances supply chain reliability, mitigating the risk of production stoppages due to raw material shortages. Furthermore, the elimination of difficult-to-remove transition metal residues, thanks to efficient filtration steps described in the patent, reduces the burden on downstream processing.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chromatographic purifications often required in lower-yielding routes. By achieving a ring-closure yield of over 85%, the consumption of high-value precursors is minimized, leading to substantial cost savings per kilogram of produced intermediate. The ability to recycle solvents like toluene and DMF further contributes to a leaner cost structure, making the final API more competitive in the global market.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on robust chemistry that is less sensitive to minor fluctuations in reaction conditions, ensuring consistent output. The starting materials are bulk chemicals with stable market pricing, shielding the project from volatile cost spikes associated with specialized reagents. This stability allows for better long-term planning and inventory management, ensuring that delivery schedules for clinical and commercial batches are met without delay.
- Scalability and Environmental Compliance: The route avoids the use of highly toxic reagents or extreme pressures, facilitating easier scale-up from pilot to commercial tonnage. The waste streams generated are primarily aqueous and organic solvents that can be treated using standard effluent protocols, simplifying environmental compliance. This scalability ensures that the manufacturing capacity can be rapidly expanded to meet surging demand for pemetrexed disodium without requiring massive capital investment in new reactor types.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this intermediate into their supply chain. The answers are derived directly from the experimental data and claims presented in the patent documentation.
Q: What is the key advantage of the Formula (I) intermediate synthesis?
A: The primary advantage is the significantly improved yield of the ring-closure reaction, which exceeds 85%, compared to lower yields in prior art methods. Additionally, the use of aldehyde protection prevents oxidation issues common in earlier routes.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the patent explicitly states the method is applicable to industrialized production due to its reasonable design, high yield, low cost, and ease of operation using standard solvents and reagents.
Q: What are the critical reaction conditions for the Heck coupling step?
A: The Heck reaction requires heating to 80-85°C under nitrogen protection using palladium acetate as a catalyst, with a molar ratio of Compound (1) to 3-butenol between 1:1.05 and 1:1.15.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pemetrexed Disodium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop to marketplace. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of pemetrexed intermediate meets the highest international standards. Our commitment to technical excellence ensures that the complex chemistry described in CN101560206A is executed with precision and reliability.
We invite you to collaborate with us to leverage this advanced synthesis route for your upcoming projects. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your strategic goals. Let us be your partner in delivering high-purity pharmaceutical intermediates with unmatched consistency and speed.
