Advanced Synthesis of Trifluoromethylthio Compounds via Aryl Silane Activation for Commercial Scale-Up
Advanced Synthesis of Trifluoromethylthio Compounds via Aryl Silane Activation for Commercial Scale-Up
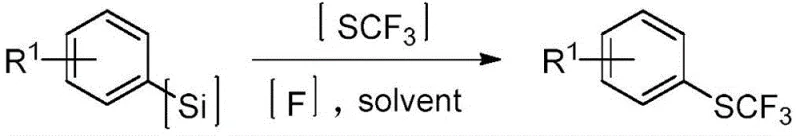
The introduction of the trifluoromethylthio (SCF3) group into organic molecules represents a pivotal strategy in modern medicinal chemistry and agrochemical design, primarily due to the group's exceptional lipophilicity and strong electron-withdrawing capabilities. Patent CN112409115B, published in October 2021, discloses a groundbreaking preparation method that utilizes substituted aryl silanes as raw materials to efficiently construct these valuable motifs. This innovation addresses long-standing challenges in the field by providing a direct, one-step synthetic route that bypasses the need for hazardous precursors or complex multi-step sequences. For R&D directors and process chemists, this patent offers a robust platform for generating diverse libraries of fluorinated compounds with high structural fidelity. The methodology leverages the unique reactivity of the silicon-carbon bond, activated by fluoride ions, to facilitate a nucleophilic attack on electrophilic sulfur species, thereby streamlining the synthesis of high-value intermediates used in next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the installation of the trifluoromethylthio group has been fraught with significant technical hurdles that impede efficient manufacturing and scale-up. Traditional indirect methods often require the conversion of other functional groups, such as thiols or halides, into the target SCF3 moiety, a process that frequently demands harsh reaction conditions, strong bases, or elevated temperatures that can degrade sensitive substrates. Furthermore, early direct trifluoromethylthiolation strategies relied heavily on trifluoromethanesulfenyl chloride (CF3SCl), a reagent notorious for its extreme toxicity, volatility, and difficulty in storage, posing severe safety risks in a production environment. While later developments introduced metal-based reagents like AgSCF3 or CuSCF3, these approaches introduced new bottlenecks, including the high cost of precious metal catalysts and the stringent requirement for heavy metal removal to meet pharmaceutical purity standards. These legacy methods often result in complex impurity profiles and low atom economy, making them economically unviable for the cost-sensitive production of bulk active pharmaceutical ingredients (APIs) or agrochemical intermediates.
The Novel Approach
In stark contrast to these cumbersome legacy protocols, the novel approach detailed in the patent utilizes substituted aryl silanes as stable, commercially available starting materials to achieve direct trifluoromethylthiolation under remarkably mild conditions. This method employs a fluoride-mediated activation strategy where the silicon group acts as a latent leaving group, enabling the generation of a reactive aryl anion species in situ without the need for cryogenic temperatures or pyrophoric reagents. By coupling this activation with stable, electrophilic trifluoromethylthio reagents, the process achieves high conversion rates and excellent selectivity in a single operational step. The reaction proceeds smoothly in common organic solvents such as tetrahydrofuran (THF) at moderate temperatures ranging from 50°C to 80°C, eliminating the energy intensity associated with high-heat processes. This streamlined workflow not only simplifies the operational complexity for plant operators but also drastically reduces the formation of side products, thereby enhancing the overall yield and purity of the final trifluoromethylthio compound.
Mechanistic Insights into Fluoride-Mediated Desilylative Trifluoromethylthiolation
The core mechanistic advantage of this technology lies in the precise control of nucleophilicity generated through fluoride-induced desilylation. When the substituted aryl silane, represented generally by Formula (II), is treated with a fluoride source such as tetrabutylammonium fluoride (TBAF), the strong affinity between silicon and fluorine drives the cleavage of the carbon-silicon bond. This cleavage generates a transient aryl anion or a hypervalent silicate intermediate that possesses significant nucleophilic character at the ipso-carbon position. This reactive species then immediately engages with the electrophilic sulfur atom of the trifluoromethylthio reagent, which acts as a source of the SCF3+ equivalent. The reaction is driven by the formation of a stable carbon-sulfur bond and the expulsion of the leaving group from the SCF3 reagent, typically a stabilized anion derived from saccharin or similar heterocyclic scaffolds. This mechanism ensures that the trifluoromethylthio group is installed regioselectively at the position previously occupied by the silyl group, preserving the integrity of other functional groups on the aromatic ring that might be sensitive to nucleophilic attack under different conditions.
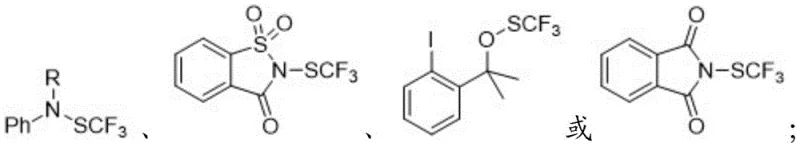
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or metal-catalyzed pathways. Because the reaction proceeds through a polar, ionic mechanism rather than a radical chain process, the formation of homocoupling byproducts (biaryls) or random radical substitution products is minimized. The use of well-defined electrophilic SCF3 reagents, such as N-trifluoromethylthio-saccharin, ensures that the sulfur source is pure and stoichiometrically controlled, preventing the introduction of sulfide or disulfide impurities that are common when using elemental sulfur or thiols. Furthermore, the byproduct of the reaction is typically a neutral, organic soluble species (like saccharin) that can be easily separated from the product during the aqueous workup or silica gel chromatography. This clean reaction profile is critical for pharmaceutical applications, where regulatory agencies impose strict limits on genotoxic impurities and residual metals, thus reducing the burden on downstream purification teams and quality control laboratories.
How to Synthesize Trifluoromethylthio Compounds Efficiently
The practical implementation of this synthesis route is designed for ease of execution in both laboratory and pilot plant settings, requiring standard equipment and readily available reagents. The process begins with the dissolution of the substituted aryl silane in a suitable solvent system, with tetrahydrofuran (THF) being the preferred medium due to its ability to solubilize both the organic substrate and the fluoride salt. A fluoride reagent, such as TBAF, is then added to the mixture under stirring to initiate the desilylation process, typically requiring a molar ratio of 1:1.5 to 1:3.0 relative to the silane substrate to ensure complete activation. Once the reactive intermediate is formed, the electrophilic trifluoromethylthio reagent is introduced, and the reaction mixture is heated to a moderate temperature of approximately 60°C for a duration of 2 to 4 hours. Detailed standardized synthesis steps, including specific workup procedures and purification parameters, are provided in the guide below to ensure reproducible results.
- Mix substituted aryl silane with a fluoride reagent (e.g., TBAF) in a solvent like THF to generate the reactive aryl anion species.
- Add the electrophilic trifluoromethylthio reagent (e.g., N-SCF3 saccharin) to the reaction mixture and heat to 50-80°C.
- Upon completion, perform aqueous workup involving extraction with ethyl acetate, washing with acid and brine, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this aryl silane-based methodology presents a compelling value proposition centered on cost efficiency and supply security. The elimination of expensive transition metal catalysts, such as silver or copper salts, removes a significant variable cost component from the bill of materials, while simultaneously eradicating the need for costly and time-consuming metal scavenging steps during purification. The reliance on stable, solid electrophilic reagents instead of gaseous or volatile liquids like CF3SCl enhances workplace safety and reduces the regulatory burden associated with handling hazardous materials, leading to lower insurance and compliance costs. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side reactions, effectively improving the overall mass balance and reducing the volume of chemical waste that requires disposal. These factors combine to create a manufacturing process that is not only economically superior but also more resilient to supply chain disruptions caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the use of commodity-grade aryl silanes and the avoidance of precious metal catalysts, which significantly lowers the raw material expenditure per kilogram of product. The mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure or cryogenic equipment, reducing capital expenditure (CAPEX) requirements for new production lines. Additionally, the simplified workup procedure, which involves standard liquid-liquid extraction and crystallization or chromatography, reduces labor hours and solvent consumption compared to complex multi-step syntheses. This holistic reduction in operational complexity translates directly into a lower cost of goods sold (COGS), providing a competitive margin advantage in the global market for fluorinated intermediates.
- Enhanced Supply Chain Reliability: Supply chain stability is bolstered by the use of aryl silanes, which are robust, shelf-stable compounds that can be sourced from multiple global suppliers, mitigating the risk of single-source dependency. The electrophilic SCF3 reagents used in this method, such as N-SCF3 saccharin derivatives, are also stable solids that do not require special cold-chain logistics or inert atmosphere storage during transport, unlike many traditional sulfurizing agents. This robustness ensures consistent availability of key inputs even during logistical bottlenecks or geopolitical tensions that might affect the transport of hazardous gases or unstable liquids. Consequently, manufacturers can maintain continuous production schedules with greater confidence, ensuring reliable delivery timelines to downstream pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns perfectly with modern green chemistry principles, facilitating easier regulatory approval and community acceptance for manufacturing sites. The absence of heavy metals in the reaction mixture means that wastewater streams are cleaner and easier to treat, reducing the load on effluent treatment plants and minimizing the risk of environmental violations. The high atom economy and reduced solvent usage contribute to a lower carbon footprint for the manufacturing process, supporting corporate sustainability goals and ESG (Environmental, Social, and Governance) reporting requirements. As regulatory pressures on chemical manufacturing intensify globally, adopting such a clean and scalable technology future-proofs the supply chain against tightening environmental regulations and potential carbon taxes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylthiolation technology, based on the data disclosed in the patent literature. These insights are intended to clarify the operational feasibility and strategic benefits for potential partners and licensees evaluating this synthetic route. Understanding these details is crucial for making informed decisions about process adoption and technology transfer.
Q: What are the primary advantages of using aryl silanes for trifluoromethylthiolation?
A: This method avoids harsh conditions and toxic reagents like CF3SCl. It utilizes stable aryl silanes and electrophilic SCF3 reagents, offering high selectivity and operational safety suitable for industrial scale-up.
Q: Which fluoride sources are compatible with this synthesis route?
A: The process supports various fluoride sources including tetrabutylammonium fluoride (TBAF), silver fluoride, potassium fluoride, sodium fluoride, and cesium fluoride, with TBAF being preferred for optimal efficiency.
Q: Is this method suitable for large-scale pharmaceutical production?
A: Yes, the method is highly scalable due to its green profile, absence of expensive transition metal catalysts, and simple post-treatment procedures which significantly reduce purification costs and environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylthio Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this advanced fluorination technology in accelerating the development of novel bioactive molecules. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including dedicated containment systems and rigorous QC labs capable of verifying stringent purity specifications for complex intermediates. We are committed to delivering high-quality trifluoromethylthio compounds that meet the exacting standards of the global pharmaceutical industry, leveraging our technical expertise to optimize yields and minimize impurities.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this method can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data for our catalog items or to discuss route feasibility assessments for your custom synthesis needs. Let us help you secure a reliable supply of high-performance fluorinated intermediates for your commercial success.
