Advanced Visible-Light Carboxylation Technology for High-Purity Pharmaceutical Intermediates
The global pharmaceutical and fine chemical industries are increasingly prioritizing sustainable synthetic methodologies that minimize environmental impact while maximizing efficiency. In this context, patent CN111777477A presents a transformative approach to the synthesis of high-value carboxylic acids, specifically succinic acid derivatives and 3-arylpropionic acids. This technology leverages visible-light photocatalysis to activate carbon dioxide (CO2), a abundant and inexpensive C1 building block, enabling its incorporation into organic frameworks under exceptionally mild conditions. Unlike traditional carboxylation methods that often demand harsh temperatures, high pressures, or toxic reagents, this innovation utilizes a 30W blue LED light source at room temperature and atmospheric pressure. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, this patent outlines a pathway to produce critical building blocks with superior atom economy and reduced operational hazards, marking a significant leap forward in green chemistry applications for drug discovery and development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of succinic acid and its derivatives has relied on six primary categories of methods, each fraught with significant technical and economic drawbacks. Traditional routes often involve the catalytic oxidation of paraffin or the hydrogenation of maleic anhydride, processes that necessitate the use of expensive noble metal catalysts and operate under severe thermal conditions. Furthermore, alternative pathways such as the electrolytic oxidation of maleic acid utilize lead plate electrodes, introducing severe risks of heavy metal residue and environmental pollution, which are unacceptable in modern GMP-compliant pharmaceutical manufacturing. Similarly, the synthesis of 3-arylpropionic acids traditionally employs catalytic hydrogenation of cinnamic acid or oxidation of propylbenzene with chromates. These legacy methods not only require high-pressure equipment that increases capital expenditure but also suffer from poor atom economy and the generation of hazardous waste streams. The reliance on stoichiometric oxidants and precious metals creates a volatile supply chain susceptible to price fluctuations and regulatory scrutiny regarding metal impurities in final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these archaic techniques, the methodology disclosed in CN111777477A offers a paradigm shift by employing a metal-free, visible-light-driven system. This novel approach utilizes readily available acrylate compounds or aryl vinyl compounds as substrates, reacting them with CO2 in the presence of a thiophenol mediator and a base such as sodium tert-butoxide. The reaction proceeds smoothly at room temperature under 1 atmosphere of CO2, driven by 30W blue LED irradiation. This eliminates the need for high-pressure autoclaves and energy-intensive heating protocols, drastically simplifying the reactor setup and reducing energy consumption. The use of CO2 as a feedstock not only lowers raw material costs compared to carbon monoxide or other C1 sources but also contributes to carbon capture utilization strategies. By avoiding transition metals entirely, this method ensures that the resulting succinic acid derivatives and 3-arylpropionic acids are free from difficult-to-remove metal contaminants, streamlining the purification process and enhancing the safety profile of the final product for downstream pharmaceutical applications.
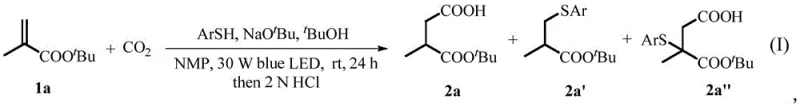
Mechanistic Insights into Visible-Light Mediated Carboxylation
The core of this technological breakthrough lies in the unique activation mode of carbon dioxide facilitated by the synergy between visible light, a base, and a thiophenol additive. Mechanistically, the reaction is believed to proceed through a radical pathway where the thiophenol acts as a hydrogen atom transfer (HAT) catalyst or a radical mediator. Under blue LED irradiation, the interaction between the base and the thiophenol likely generates a thiyl radical species capable of adding to the electron-deficient double bond of the acrylate substrate. This addition generates a carbon-centered radical at the beta-position, which is sufficiently nucleophilic to attack the thermodynamically stable CO2 molecule. This step is critical, as CO2 is kinetically inert and typically requires high energy to activate. The subsequent protonation and termination steps yield the carboxylated product with high regioselectivity. Crucially, the presence of the thiophenol and the specific reaction conditions suppress competing side reactions, such as the Michael addition of the thiol to the acrylate or alpha-carboxylation, which are common pitfalls in traditional radical carboxylations. This precise control over the reaction trajectory ensures high purity and yield, addressing a major pain point for process chemists who often struggle with complex impurity profiles in radical chemistry.
Furthermore, the system exhibits remarkable chemoselectivity and functional group tolerance, which is essential for the synthesis of complex pharmaceutical intermediates. The mild basic conditions and neutral pH environment post-reaction allow for the compatibility of sensitive functional groups such as esters, ethers, halogens, and even heterocycles like indoles and thiophenes. For instance, substrates containing O-allyl groups or benzyl alcohol moieties remain intact during the carboxylation process, demonstrating that the radical species generated are highly selective for the intended C-C bond formation with CO2. This selectivity minimizes the formation of by-products, thereby reducing the burden on downstream purification units like chromatography columns. The ability to tolerate diverse substituents on the aromatic ring of aryl vinyl substrates, including electron-donating and electron-withdrawing groups, further expands the utility of this method. Such robustness implies that this technology can be applied to late-stage functionalization of complex drug candidates, offering a versatile tool for medicinal chemists aiming to introduce carboxylic acid motifs without protecting group manipulations.
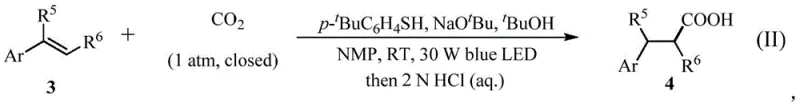
How to Synthesize Succinic Acid Derivatives Efficiently
The practical implementation of this visible-light carboxylation protocol is straightforward and designed for ease of adoption in both laboratory and pilot-scale settings. The process begins with the preparation of a reaction mixture in a dry Schlenk tube or similar vessel, where a base like sodium tert-butoxide is combined with a polar aprotic solvent such as NMP. Key to the success of the reaction is the rigorous exclusion of oxygen, achieved through freeze-pump-thaw degassing cycles, followed by the introduction of CO2 gas to maintain a positive pressure of 1 atmosphere. The addition of the thiophenol mediator and the olefin substrate initiates the catalytic cycle upon exposure to blue LED light. After a reaction period of approximately 24 hours at room temperature, the mixture is quenched with dilute acid and extracted. The detailed standardized synthesis steps for producing these high-value intermediates are provided in the guide below, ensuring reproducibility and consistency for technical teams looking to integrate this green chemistry solution into their existing workflows.
- Prepare the reaction mixture by adding a base such as sodium tert-butoxide into a dry reaction vessel under an inert atmosphere, followed by the addition of solvent, thiophenol mediator, and the acrylate substrate.
- Degas the reaction solution thoroughly using freeze-pump-thaw cycles to remove oxygen, then fill the vessel with CO2 gas to a pressure of 0.1 MPa (1 atm).
- Irradiate the reaction mixture with a 30W blue LED light source at room temperature for 24 hours, then quench with acid and purify the resulting succinic acid derivative via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN111777477A offers compelling strategic advantages that extend beyond mere technical feasibility. The most significant benefit is the drastic reduction in manufacturing costs driven by the elimination of expensive noble metal catalysts such as palladium or rhodium, which are subject to volatile market pricing and supply constraints. By replacing these precious metals with inexpensive organic thiophenols and common bases, the raw material cost structure is significantly optimized. Additionally, the operation at ambient temperature and atmospheric pressure removes the need for specialized high-pressure reactors and energy-intensive heating systems, leading to substantial savings in both capital expenditure (CAPEX) for equipment and operational expenditure (OPEX) for energy. This simplification of the process infrastructure enhances the overall economic viability of producing succinic acid derivatives and 3-arylpropionic acids on a large scale, making it an attractive option for cost-sensitive bulk manufacturing.
- Cost Reduction in Manufacturing: The transition to a metal-free photocatalytic system fundamentally alters the cost dynamics of carboxylic acid production. By removing the requirement for transition metal catalysts, manufacturers avoid the substantial costs associated with catalyst procurement, recovery, and the extensive purification steps needed to meet strict residual metal limits in pharmaceutical products. Furthermore, the use of CO2 as a C1 source is inherently cheaper than using carbon monoxide or formic acid derivatives, providing a low-cost feedstock advantage. The mild reaction conditions also translate to lower energy bills, as there is no need for prolonged heating or cooling cycles, allowing for a more efficient allocation of utility resources within the production facility.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as acrylates, styrenes, and simple thiophenols ensures a robust and stable supply chain. Unlike specialized catalysts that may have single-source suppliers or long lead times, the reagents for this process are widely available from multiple global vendors, mitigating the risk of supply disruptions. The scalability of the process, demonstrated successfully at the gram level in the patent data, suggests that scaling to kilogram or ton quantities is feasible without encountering significant engineering bottlenecks. This reliability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of downstream pharmaceutical clients who require consistent quality and volume.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with modern green chemistry principles and increasingly stringent environmental regulations. The absence of heavy metals eliminates the generation of toxic metal-containing waste streams, simplifying waste treatment and disposal procedures and reducing associated compliance costs. The use of CO2, a greenhouse gas, as a raw material contributes to carbon footprint reduction goals, enhancing the sustainability profile of the manufactured intermediates. This eco-friendly attribute is becoming a key differentiator in the market, as end-users increasingly prefer suppliers who can demonstrate a commitment to sustainable manufacturing practices, thereby future-proofing the supply chain against evolving regulatory landscapes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this visible-light carboxylation technology. These answers are derived directly from the experimental data and specifications outlined in patent CN111777477A, providing clarity on the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs and for procurement officers assessing the long-term value proposition of this innovative synthetic route.
Q: Does this visible-light carboxylation method require expensive noble metal catalysts?
A: No, the method described in patent CN111777477A operates without transition metal catalysts. It utilizes an organic thiophenol mediator and a base under visible light irradiation, which significantly reduces raw material costs and eliminates the risk of heavy metal contamination in the final pharmaceutical intermediate.
Q: What is the substrate scope for this CO2 fixation reaction?
A: The process demonstrates excellent compatibility with a wide range of substrates, including various acrylate esters (primary, secondary, and tertiary) and aryl vinyl compounds. It tolerates diverse functional groups such as halogens, ethers, and heterocycles, making it suitable for synthesizing complex drug intermediates.
Q: Is this photochemical process scalable for industrial production?
A: Yes, the patent explicitly validates the scalability of the method. Successful gram-scale reactions (e.g., 10 mmol scale) have been demonstrated with maintained yields, indicating that the mild reaction conditions (room temperature, 1 atm CO2) are conducive to scaling up for commercial manufacturing without requiring high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Succinic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the visible-light carboxylation technology detailed in CN111777477A for the production of high-purity succinic acid derivatives and 3-arylpropionic acids. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory-scale protocol into robust, commercial-scale manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this green chemistry method—such as high selectivity and mild conditions—are preserved and optimized at larger volumes. We are equipped with state-of-the-art photochemical reactors and rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Whether you are looking to reduce the cost of goods for an existing API or develop a new sustainable route for a complex intermediate, our technical procurement team is ready to assist. Please contact us to request a Customized Cost-Saving Analysis tailored to your specific molecule. We encourage you to reach out for specific COA data and route feasibility assessments to see how our capabilities align with your supply chain goals. Let us help you secure a reliable supply of high-quality intermediates while advancing your sustainability objectives through innovative chemical manufacturing.
