Scalable Green Synthesis of 1,2,3-Triazoles: A Breakthrough in Aqueous Click Chemistry for Industrial Applications
Scalable Green Synthesis of 1,2,3-Triazoles: A Breakthrough in Aqueous Click Chemistry for Industrial Applications
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to synthesize nitrogen-containing heterocycles, particularly 1,2,3-triazoles, which serve as critical bioisosteres and pharmacophores in modern drug design. A pivotal advancement in this domain is detailed in patent CN103880762A, which discloses a novel preparation method for 1,2,3-triazole compounds utilizing a unique pullulan triazole/cuprous chloride catalytic system. This technology represents a significant departure from traditional copper-catalyzed azide-alkyne cycloaddition (CuAAC) protocols by enabling the reaction to proceed in pure water with exceptional efficiency. By leveraging a polymer-stabilized copper catalyst, this method addresses long-standing challenges regarding catalyst stability, toxicity, and separation, offering a robust solution for the commercial scale-up of complex pharmaceutical intermediates.
The core innovation lies in the application of a pullulan polymer functionalized with triazole groups, which acts not merely as a support but as an active stabilizing ligand for cuprous ions. This specific interaction prevents the rapid oxidation of Cu(I) to Cu(II), a common failure mode in aqueous click chemistry that typically necessitates the use of excess reducing agents or inert atmospheres. Consequently, the process operates under mild conditions ranging from room temperature to 100°C and atmospheric pressure, eliminating the need for energy-intensive heating or specialized high-pressure equipment. For R&D directors and process chemists, this translates to a simplified workflow that maintains high atom economy while drastically reducing the operational complexity associated with moisture-sensitive reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of 1,2,3-triazoles via the Huisgen cycloaddition has historically relied on copper salts such as cuprous chloride or cuprous bromide, often dissolved in organic solvents or mixed solvent systems to ensure solubility and reactivity. However, direct use of cuprous salts presents severe limitations for industrial application; these salts are inherently unstable in the presence of oxygen and moisture, rapidly oxidizing to catalytically inactive cupric species. To mitigate this, conventional protocols frequently require the addition of expensive and toxic stabilizing ligands like TBTA (tris(benzyltriazolylmethyl)amine) or phosphines, which complicate the downstream purification process and introduce potential heavy metal contaminants into the final API. Furthermore, standard methods often demand catalyst loadings between 1 mol% and 10 mol%, which, when scaled to metric ton production, results in substantial raw material costs and generates significant volumes of copper-contaminated waste that requires expensive remediation.
The Novel Approach
The methodology described in patent CN103880762A fundamentally re-engineers the catalytic environment by introducing a pullulan triazole polymer as a macromolecular ligand. This approach creates a protective microenvironment around the cuprous ions, effectively shielding them from oxidative degradation without the need for hazardous organic additives. The result is a catalytic system that functions optimally in water, the cheapest and safest solvent available, thereby eliminating the fire hazards and environmental liabilities associated with volatile organic compounds (VOCs). Moreover, this system demonstrates remarkable efficiency with catalyst loadings as low as 0.001 mol%, representing a reduction of several orders of magnitude compared to standard practices. This drastic reduction in catalyst usage, combined with the ability to recycle the aqueous catalyst phase multiple times without loss of activity, offers a compelling value proposition for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pullulan-Stabilized Cu(I) Catalysis
The mechanistic superiority of this system stems from the specific coordination chemistry between the triazole moieties on the pullulan backbone and the copper center. In standard aqueous CuAAC reactions, the disproportionation of Cu(I) into Cu(0) and Cu(II) is a major pathway for catalyst deactivation. The pullulan triazole polymer acts as a multidentate ligand that kinetically stabilizes the Cu(I) oxidation state, maintaining a high concentration of the active catalytic species throughout the reaction timeline. This stabilization allows the reaction to proceed through the standard metallacycle mechanism—forming a copper-acetylide intermediate that subsequently reacts with the organic azide—but with enhanced turnover numbers (TON) due to the prolonged lifespan of the catalyst. The polymer matrix also likely facilitates a phase-transfer effect, bringing the hydrophobic organic substrates into closer proximity with the hydrophilic catalyst complex, thereby accelerating reaction kinetics even in a purely aqueous medium.
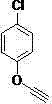
From an impurity control perspective, this mechanism offers distinct advantages. The high selectivity of the stabilized Cu(I) species minimizes side reactions such as Glaser coupling (oxidative homocoupling of alkynes), which is a common impurity in copper-catalyzed alkyne chemistry. The use of water as the sole solvent further simplifies the impurity profile, as there are no solvent-derived byproducts or residual organic solvents to remove. The product, being organic and hydrophobic, naturally separates from the aqueous catalyst phase upon completion, allowing for simple extraction. This inherent ease of separation ensures that the final 1,2,3-triazole product meets stringent purity specifications with minimal downstream processing, a critical factor for regulatory compliance in the production of high-purity OLED materials or API intermediates.
How to Synthesize 1,2,3-Triazole Efficiently
The synthesis protocol outlined in the patent provides a straightforward, scalable route for producing diverse 1,2,3-triazole derivatives. The process begins with the preparation of the pullulan triazole ligand, followed by its combination with cuprous chloride in water to form the active catalyst. Substrates, including various terminal alkynes and organic azides, are then introduced to the aqueous mixture. The reaction proceeds under mild thermal conditions, and upon completion, the product is isolated via organic extraction, leaving the catalyst in the water for reuse. This streamlined workflow eliminates the need for complex inert gas setups or anhydrous conditions, making it highly accessible for both laboratory optimization and plant-scale production. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Prepare the catalyst system by mixing pullulan triazole and cuprous chloride in water, ensuring the molar ratio supports Cu(I) stabilization.
- Combine the terminal alkyne compound and organic azide substrate in the aqueous catalyst mixture at room temperature to 100°C.
- Stir the reaction until completion, then extract the product with ethyl acetate, allowing the aqueous catalyst phase to be recycled for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this pullulan-based catalytic system offers transformative economic and logistical benefits. The shift from organic solvents to water immediately reduces raw material procurement costs and eliminates the regulatory burdens associated with VOC emissions and hazardous waste disposal. Furthermore, the extreme stability of the catalyst system means that reagents can be stored for longer periods without degradation, reducing inventory shrinkage and ensuring supply continuity. The ability to run reactions at ambient pressure and moderate temperatures also lowers energy consumption, contributing to a smaller carbon footprint and reduced utility costs per kilogram of product. These factors collectively drive significant cost savings while enhancing the overall resilience of the supply chain against raw material volatility.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic reduction in catalyst loading. Traditional methods often require 1-10 mol% of copper and expensive ligands, whereas this technology achieves quantitative yields with as little as 0.001 mol% of cuprous chloride. This represents a massive decrease in the cost of goods sold (COGS) related to catalytic materials. Additionally, the elimination of costly phosphine ligands and reducing agents like sodium ascorbate further strips away unnecessary expenses. The simplicity of the workup—simple extraction without column chromatography—reduces labor hours and silica gel consumption, leading to substantial operational cost reductions that directly improve profit margins.
- Enhanced Supply Chain Reliability: Reliance on water as a solvent removes the supply chain risks associated with fluctuating prices and availability of specialty organic solvents. Water is universally available, inexpensive, and non-flammable, simplifying storage and handling logistics. Moreover, the catalyst system's reusability means that the effective consumption of copper salts is negligible over multiple batches. This decouples production capacity from the immediate availability of fresh catalyst batches, allowing for continuous manufacturing runs. For global supply chains, this robustness ensures that production schedules are not disrupted by minor shortages of specialized reagents, guaranteeing on-time delivery to downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this aqueous method inherently mitigates those risks. Running reactions in water eliminates the risk of solvent fires and explosions, a critical safety advantage for large-scale reactors. From a compliance standpoint, the process generates significantly less hazardous waste, as the aqueous phase containing the polymer-bound copper can be treated or recycled rather than disposed of as toxic sludge. This alignment with green chemistry principles facilitates easier permitting for new production lines and helps companies meet increasingly strict environmental, social, and governance (ESG) targets without compromising on yield or quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this pullulan-triazole catalytic technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a transparent view of the process capabilities. Understanding these details is essential for evaluating the feasibility of integrating this method into existing manufacturing workflows.
Q: How does the pullulan triazole ligand improve the Huisgen cycloaddition reaction?
A: The pullulan triazole polymer acts as a stabilizing additive for cuprous ions (Cu+), preventing their oxidation to inactive Cu(II) species. This stabilization allows for significantly lower catalyst loadings (down to 0.001 mol%) and enables the reaction to proceed efficiently in water without the need for toxic organic ligands or inert atmospheres.
Q: Can the catalyst system be reused for multiple reaction cycles?
A: Yes, the pullulan triazole/cuprous chloride system is highly reusable. Since the catalyst remains in the aqueous phase while the product is extracted into an organic solvent (like ethyl acetate), the water layer containing the active catalyst can be directly reused for at least four consecutive cycles with maintained high yields (>98%).
Q: What are the environmental advantages of this synthesis method compared to traditional methods?
A: This method replaces volatile organic solvents (VOCs) with water, drastically reducing environmental impact and disposal costs. Additionally, the ultra-low catalyst loading and elimination of expensive phosphine ligands reduce heavy metal waste and raw material costs, aligning with green chemistry principles for sustainable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2,3-Triazole Supplier
The technological potential of water-based click chemistry is immense, yet translating patent concepts into consistent, high-quality commercial supply requires deep expertise in process engineering and quality control. NINGBO INNO PHARMCHEM stands at the forefront of this translation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our team specializes in adapting innovative catalytic systems like the pullulan/CuCl complex to meet the rigorous demands of the global pharmaceutical market. We understand that consistency is key; therefore, our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of 1,2,3-triazole intermediate meets stringent purity specifications, free from residual heavy metals or solvent impurities.
We invite R&D directors and procurement specialists to collaborate with us to evaluate the feasibility of this green synthesis route for your specific projects. Whether you require custom synthesis of novel triazole derivatives or scale-up of existing intermediates, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples that demonstrate the superior quality and economic advantages of our manufacturing capabilities.
