Advanced Synthesis of Nitropyridine Compounds for Commercial Pharmaceutical Manufacturing
Introduction to Novel Nitropyridine Synthesis Technologies
The chemical landscape for producing high-value heterocyclic intermediates is constantly evolving, driven by the need for safer, more efficient, and regioselective pathways. A pivotal advancement in this domain is documented in patent CN102020606B, which outlines a sophisticated synthesis method for nitropyridine compounds. This technology represents a significant departure from classical electrophilic aromatic substitution, offering a strategic route to access 3-nitropyridine derivatives that are notoriously difficult to synthesize via direct nitration due to the electron-deficient nature of the pyridine ring. By leveraging a diazotization-deamination strategy starting from 2-amino-nitropyridine precursors, this method achieves the target molecular architecture under remarkably mild conditions. For R&D directors and process chemists, this approach opens new avenues for constructing complex nitrogen-containing scaffolds essential for modern drug discovery, particularly where traditional methods fail to provide adequate yields or selectivity.
The core innovation lies in the transformation of an amino group into a leaving group via nitrous acid ester mediation, effectively allowing for the installation of the nitro group at the desired position prior to the removal of the directing amine. This two-in-one functional manipulation ensures that the final product retains the critical nitro functionality while eliminating the auxiliary amino group in a streamlined sequence. The implications for the supply chain are profound, as it reduces the reliance on corrosive mixed acids and extreme thermal inputs. As we delve deeper into the technical specifics, it becomes clear that this patent provides a robust framework for the commercial scale-up of complex pharmaceutical intermediates, addressing both purity concerns and operational safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
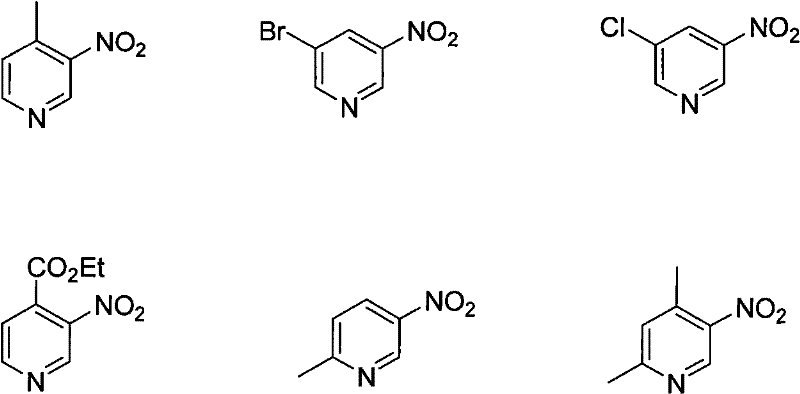
Historically, the synthesis of nitropyridine compounds has been plagued by severe operational challenges and safety hazards inherent to direct nitration protocols. Traditional methods often rely on aggressive nitrating agents such as fuming nitric acid in the presence of sulfuric acid or oleum, necessitating extremely high temperatures to overcome the deactivating effect of the pyridine nitrogen. As illustrated in prior art, some processes require heating up to 270°C in the presence of sulfur trioxide and potassium nitrate, creating an environment that is not only energy-intensive but also highly corrosive to standard reactor materials. Such harsh conditions frequently lead to poor regioselectivity, generating complex mixtures of 2-, 3-, and 4-nitro isomers that are difficult and costly to separate. Furthermore, the use of strong mineral acids generates substantial quantities of acidic waste, complicating environmental compliance and increasing the burden on wastewater treatment facilities.
Beyond the thermodynamic barriers, the economic viability of conventional nitration is often compromised by the need for specialized equipment capable of withstanding high pressure and extreme acidity. The risk of thermal runaway reactions is elevated when dealing with exothermic nitration mixtures at such elevated temperatures, posing significant safety risks to plant personnel. Additionally, the purification of the crude reaction mass often requires extensive chromatographic separation or multiple recrystallization steps, which drastically reduces the overall throughput and increases the cost of goods sold (COGS). For procurement managers, these factors translate into higher raw material costs, longer lead times due to complex purification, and a less reliable supply chain vulnerable to equipment downtime and regulatory scrutiny regarding hazardous waste disposal.
The Novel Approach
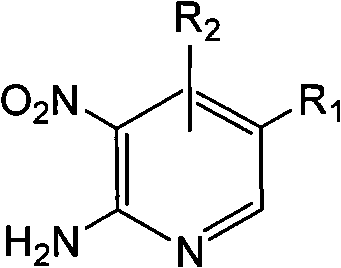
In stark contrast to the brute force of direct nitration, the method disclosed in CN102020606B employs a subtle and highly effective chemical strategy centered on the reactivity of 2-amino-nitropyridine compounds. By utilizing a pre-functionalized starting material where the amino group acts as a powerful activating and directing group, the nitration step (performed prior to the scope of this specific patent or implied in the precursor synthesis) becomes facile and selective. The patent then focuses on the critical conversion step: the replacement of the amino group with a hydrogen atom while preserving the sensitive nitro group. This is achieved through a reaction with nitrous acid esters in an ether solvent, followed by acidic hydrolysis. The result is a process that operates at a gentle 40-60°C, a dramatic reduction from the 270°C required by older methods. This mildness preserves the integrity of other functional groups that might be present on the pyridine ring, such as halogens or esters, thereby expanding the scope of accessible chemical space.
The operational simplicity of this novel approach cannot be overstated. It eliminates the need for handling large volumes of corrosive mixed acids during the final transformation step, replacing them with organic nitrites and dilute hydrochloric acid. The reaction proceeds in a homogeneous or semi-homogeneous phase in solvents like tetrahydrofuran (THF), which are easier to recover and recycle compared to sulfuric acid sludge. For the supply chain, this translates to a more robust and scalable process. The reduction in thermal stress on equipment extends the lifespan of reactors and reduces maintenance frequency. Moreover, the simplified workup procedure involving phase separation and recrystallization allows for faster batch turnover times, enabling manufacturers to respond more agilely to market demands for high-purity nitropyridine intermediates.
Mechanistic Insights into Diazotization-Mediated Deamination
To fully appreciate the technical merit of this synthesis, one must understand the mechanistic underpinnings of the diazotization-deamination sequence. The process begins with the interaction between the primary amine of the 2-amino-nitropyridine substrate and the nitrous acid ester (such as isopentyl nitrite) in an ether solvent. Under the mildly acidic or neutral conditions initially present, the nitrous acid ester generates the active nitrosating species in situ. This species attacks the nucleophilic nitrogen of the amino group, forming a diazonium intermediate. In classical aqueous diazotization, this intermediate is often unstable and prone to decomposition; however, in the organic solvent system described, the stability is managed to allow for the subsequent transformation. The presence of the electron-withdrawing nitro group on the ring influences the electronics of the diazonium moiety, making it susceptible to nucleophilic attack or reductive elimination depending on the specific conditions.
Upon the addition of hydrochloric acid in the second stage, the reaction environment shifts, facilitating the hydrolysis or reduction of the diazonium group. The protonation of the intermediate likely triggers the loss of nitrogen gas (N2), a highly favorable entropic driver that pushes the reaction to completion. The resulting cationic species on the pyridine ring is then quenched, effectively replacing the -N2+ group with a proton (from the solvent or acid) to yield the final nitropyridine product. This mechanism is crucial for impurity control because it avoids the radical mechanisms often associated with high-temperature nitration, which can lead to poly-nitrated byproducts or ring-oxidized impurities. By controlling the stoichiometry of the nitrous acid ester (preferably 1:3 molar ratio) and the temperature (optimized at 60°C), the process minimizes side reactions, ensuring a cleaner crude profile that simplifies downstream purification.
How to Synthesize Nitropyridine Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and purity. The protocol is designed to be straightforward, minimizing the need for exotic catalysts or anhydrous conditions beyond standard solvent drying. The key to success lies in the careful selection of the nitrous acid ester and the precise control of the acid addition rate during the workup phase. Operators should note that while the reaction is tolerant of various ether solvents, THF provides the best balance of solubility for both the starting amino compound and the intermediate species. The following guide summarizes the critical operational steps derived from the patent examples, serving as a foundational reference for process development teams looking to adopt this technology.
- Dissolve the 2-amino-nitropyridine starting material in an ether solvent such as THF and add a nitrous acid ester.
- Heat the reaction mixture to 40-60°C and maintain for 10-24 hours to complete the diazotization and substitution.
- Cool the solution, add hydrochloric acid for hydrolysis, separate layers, and purify the organic phase via washing and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and logistical continuity, the shift to this diazotization-based synthesis offers tangible economic benefits that extend beyond simple yield metrics. The primary advantage is the drastic reduction in operational expenditure (OPEX) associated with energy and waste management. By operating at 60°C instead of 270°C, the energy demand for heating reactors is significantly lowered, and the cooling requirements for quenching exotherms are more manageable. This thermal efficiency directly correlates to lower utility costs per kilogram of product. Furthermore, the avoidance of concentrated sulfuric acid and oleum eliminates the need for expensive corrosion-resistant alloys (like Hastelloy) in favor of standard glass-lined or stainless steel reactors, representing a substantial capital expenditure (CAPEX) saving for new production lines.
- Cost Reduction in Manufacturing: The elimination of harsh mixed acids and the simplification of the workup procedure lead to significant cost savings. Traditional nitration requires neutralization of vast amounts of acid, generating tons of salt waste that must be disposed of as hazardous material. In contrast, this method generates organic waste streams that are often easier to treat or incinerate for energy recovery. Additionally, the use of commercially available nitrites and ether solvents ensures stable raw material pricing, shielding the supply chain from the volatility often seen in specialty acid markets. The streamlined purification process, often requiring only recrystallization, reduces solvent consumption and labor hours, further driving down the unit cost of the final API intermediate.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the robustness of the reaction conditions. High-temperature processes are prone to fouling and coking, which necessitate frequent shutdowns for cleaning and maintenance. The mild nature of this synthesis minimizes reactor fouling, allowing for longer campaign runs and higher asset utilization rates. Moreover, the reagents used, such as isopentyl nitrite and THF, are commodity chemicals with well-established global supply chains, reducing the risk of raw material shortages. This stability ensures that production schedules can be met consistently, a critical factor for pharmaceutical customers who require just-in-time delivery of key intermediates to maintain their own drug manufacturing timelines.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, particularly with exothermic nitrations. This method's mild thermal profile makes it inherently safer to scale from kilogram to tonne quantities, as the heat of reaction is easier to dissipate. From an environmental perspective, the reduction in acidic waste aligns with increasingly stringent global regulations on industrial effluents. Companies adopting this greener chemistry can improve their sustainability profiles, potentially qualifying for green manufacturing incentives and avoiding fines associated with hazardous waste discharge. The ability to recycle the ether solvent further enhances the environmental footprint, making this a sustainable choice for long-term production strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitropyridine synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent literature, providing a transparent view of the method's capabilities and limitations. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the advantages of this nitropyridine synthesis method over traditional nitration?
A: Unlike traditional mixed-acid nitration which requires harsh conditions and high temperatures (up to 270°C), this method utilizes a mild diazotization pathway at 60°C, significantly reducing energy consumption and equipment corrosion risks.
Q: What solvents are compatible with this synthesis protocol?
A: The patent specifies ether solvents including diethyl ether, MTBE, isopropyl ether, THF, and 1,4-dioxane, with THF being the preferred choice for optimal solubility and reaction kinetics.
Q: Can this method be scaled for industrial production of 3-nitropyridine derivatives?
A: Yes, the process is designed for industrial applicability by simplifying operations, shortening reaction times, and utilizing commercially available reagents like isopentyl nitrite, making it suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous process engineering. Our team specializes in bridging this gap, offering comprehensive CDMO services tailored to the synthesis of complex heterocyclic intermediates like nitropyridines. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of mild diazotization chemistry are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs, guaranteeing that every batch meets the exacting standards required by the global pharmaceutical industry.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains. By leveraging our expertise in this specific synthesis method, we can help you achieve a Customized Cost-Saving Analysis that identifies specific efficiencies in your current sourcing strategy. We encourage you to contact our technical procurement team to request specific COA data for our nitropyridine portfolio and to discuss route feasibility assessments for your custom projects. Together, we can build a resilient, cost-effective, and high-quality supply chain for your critical drug intermediates.
