Advanced One-Pot Synthesis of Benzoxazole Derivatives for Commercial Pharmaceutical Applications
Introduction to Next-Generation Benzoxazole Synthesis
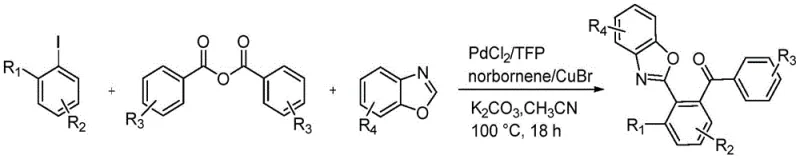
The pharmaceutical and fine chemical industries are constantly seeking more efficient routes to access complex heterocyclic scaffolds, particularly those containing benzoxazole motifs which are prevalent in bioactive molecules. A significant breakthrough in this domain is documented in patent CN107382894B, which discloses a robust preparation method for o-benzophenone benzoxazole derivatives. This technology leverages a sophisticated palladium-phosphine complex catalytic system assisted by norbornene to achieve a highly selective one-pot synthesis. By integrating halogenated benzene, substituted benzoic anhydride, and substituted benzoxazole under mild alkaline conditions, this process circumvents the multi-step limitations of traditional approaches. The strategic use of norbornene as a transient mediator allows for precise ortho-C-H activation, a feat that is notoriously difficult to achieve with standard cross-coupling protocols. For R&D directors and process chemists, this represents a pivotal shift towards atom-economical and operationally simple methodologies that can be seamlessly integrated into existing manufacturing pipelines for high-value intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of benzoxazole derivatives has relied heavily on classical condensation reactions or direct coupling strategies that suffer from significant drawbacks in terms of efficiency and environmental impact. Traditional synthetic routes often employ o-aminophenol and benzaldehyde precursors undergoing cyclization under the influence of tin dioxide, a process that generates substantial heavy metal waste and requires rigorous purification steps to meet pharmaceutical purity standards. Furthermore, alternative methods involving the direct coupling of benzoxazole to aryl halides using cuprous iodide or nickel catalysis frequently struggle with poor regioselectivity, leading to complex mixtures of ortho- and para-substituted isomers that are difficult to separate. These legacy processes not only inflate the cost of goods due to low yields and expensive separation technologies but also pose serious supply chain risks associated with the handling of toxic reagents and the disposal of hazardous byproducts. The inability to directly introduce functional groups at the ortho-position without pre-functionalization adds unnecessary synthetic steps, prolonging lead times and reducing overall process throughput.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a paradigm-shifting one-pot strategy that utilizes a palladium-phosphine complex in conjunction with norbornene to orchestrate a cascade of reactions with exceptional precision. This novel approach effectively merges C-H activation and acylation into a single operational unit, eliminating the need for isolated intermediates and reducing solvent consumption. The reaction proceeds through a unique mechanism where norbornene acts as a transient bridge, facilitating the activation of the ortho-carbon-hydrogen bond adjacent to the halogen substituent on the benzene ring. This allows for the simultaneous coupling of the benzoxazole nitrogen and the introduction of the benzoyl group from the anhydride, resulting in the formation of the target o-benzophenone benzoxazole derivative with high fidelity. The use of inexpensive and readily available starting materials, such as iodotoluene and benzoic anhydride, combined with a catalytic amount of palladium, drastically simplifies the supply chain logistics and reduces the raw material cost burden, making it an attractive option for commercial scale-up.
Mechanistic Insights into Pd/Norbornene Catalyzed Ortho-Acylation
The core of this technological advancement lies in the intricate catalytic cycle driven by the synergy between the palladium catalyst, the phosphine ligand, and the norbornene mediator. The reaction initiates with the oxidative addition of the aryl iodide to the active Pd(0) species, forming an aryl-palladium(II) intermediate. Crucially, the presence of norbornene triggers an insertion into the Pd-C bond, followed by a concerted metalation-deprotonation (CMD) process that activates the ortho-C-H bond of the aromatic ring. This creates a palladacycle intermediate that is poised for further functionalization. Subsequently, the substituted benzoic anhydride undergoes oxidative addition or ligand exchange with this intermediate, introducing the carbonyl functionality. The final stages involve reductive elimination to forge the C-C bond at the ortho-position and the release of norbornene, regenerating the active catalyst for the next cycle. Simultaneously, the benzoxazole moiety couples via a copper-mediated pathway, likely involving a transmetallation step with the CuBr co-catalyst, ensuring the formation of the C-N bond. This dual-activation strategy ensures that both the carbon and nitrogen connections are made with high regiocontrol, minimizing the formation of unwanted side products.
From an impurity control perspective, this mechanism offers distinct advantages by limiting the number of reactive intermediates that can diverge into side pathways. The specificity of the norbornene-mediated C-H activation ensures that substitution occurs exclusively at the ortho-position relative to the iodine leaving group, preventing the formation of meta- or para-isomers that typically plague electrophilic aromatic substitution reactions. Furthermore, the use of potassium carbonate as a mild base helps to maintain a neutral to slightly basic environment, reducing the risk of acid-catalyzed degradation of the sensitive benzoxazole ring or the anhydride reagent. The choice of tris(2-furyl)phosphine (TFP) as a ligand is also critical, as its electron-rich nature stabilizes the palladium center during the oxidative addition steps while remaining labile enough to allow for the necessary coordination changes during the catalytic turnover. This fine-tuned balance results in a cleaner reaction profile, simplifying downstream purification and enhancing the overall purity of the final API intermediate.
How to Synthesize o-Benzophenone Benzoxazole Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reaction parameters to maximize yield and reproducibility. The protocol dictates a specific molar ratio of reactants, typically maintaining a 1:2:3 ratio of halogenated benzene to benzoxazole to benzoic anhydride to drive the equilibrium towards product formation. The reaction is conducted in polar aprotic solvents such as acetonitrile, 1,4-dioxane, or DMF, with acetonitrile showing particularly favorable results in terms of solubility and reaction rate. Temperature control is vital, with the process optimized at 100 °C for a duration of 16 to 18 hours to ensure complete consumption of the starting materials. Detailed standardized synthesis steps, including precise weighing, inert atmosphere setup, and workup procedures, are essential for achieving the reported yields of up to 94% in optimized examples.
- Charge a dry reaction vessel with iodotoluene, benzoic anhydride, benzoxazole, PdCl2, TFP ligand, CuBr, norbornene, K2CO3, and acetonitrile solvent.
- Stir the reaction mixture under nitrogen protection at 100 °C for 16 to 18 hours to ensure complete conversion.
- Filter off insoluble matter, remove solvent, and purify the crude product via silica gel column chromatography to obtain the target derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis method translates into tangible strategic benefits that extend beyond mere chemical curiosity. The primary advantage lies in the drastic simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply security. By consolidating multiple synthetic transformations into a single pot, the method eliminates the need for intermediate isolation, drying, and storage, thereby reducing the physical footprint required for production and minimizing the risk of material loss during transfer operations. The reliance on commodity chemicals like iodotoluene and benzoic anhydride ensures a stable and diversified supply base, mitigating the risks associated with sourcing specialized or proprietary reagents that may be subject to market volatility or geopolitical constraints. Furthermore, the catalytic nature of the palladium system means that expensive precious metals are used in sub-stoichiometric amounts, significantly lowering the raw material cost per kilogram of product compared to stoichiometric organometallic methods.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the elimination of expensive and toxic reagents such as tin dioxide or specialized organolithium compounds that are often required in conventional routes. By utilizing a small amount of palladium catalyst and cheap phosphine ligands, the direct material cost is substantially lowered. Additionally, the one-pot nature of the reaction reduces energy consumption associated with heating and cooling cycles for multiple steps, as well as the labor costs linked to monitoring and manipulating sequential reactions. The simplified workup procedure, which involves basic filtration and chromatography, further decreases the consumption of silica gel and solvents, contributing to a leaner and more cost-effective manufacturing model that improves overall profit margins for high-volume production.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by reducing the complexity of the bill of materials. Since the key starting materials are bulk chemicals available from multiple global suppliers, the risk of single-source dependency is minimized. The reaction conditions are relatively mild and do not require extreme pressures or cryogenic temperatures, allowing the process to be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility with existing infrastructure accelerates technology transfer and scale-up timelines, ensuring that production can be ramped up quickly to meet surging demand without the need for capital-intensive equipment upgrades. The high selectivity of the reaction also means less waste generation, simplifying regulatory compliance and waste disposal logistics.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this method offers a greener alternative to traditional benzoxazole synthesis. The avoidance of heavy metal waste streams, particularly tin residues, simplifies wastewater treatment and reduces the environmental liability of the manufacturing site. The use of acetonitrile, while requiring proper handling, is a well-understood solvent in the industry with established recovery and recycling protocols. The high atom economy of the coupling reaction ensures that a greater proportion of the input mass ends up in the final product, aligning with green chemistry principles and corporate sustainability goals. This environmental friendliness not only aids in meeting stringent regulatory standards but also enhances the brand reputation of the end-product in markets that increasingly value sustainable manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzoxazole synthesis technology. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on reaction scope, optimization, and practical application. Understanding these nuances is critical for process development teams evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: What are the key advantages of this Pd/norbornene catalytic system over traditional tin dioxide methods?
A: The Pd/norbornene system avoids toxic tin reagents and harsh cyclization conditions, enabling a milder, one-pot synthesis with better regioselectivity for ortho-substitution.
Q: Can this method accommodate diverse substituents on the benzoxazole ring?
A: Yes, the patent demonstrates successful synthesis with various substituents including methyl, tert-butyl, and chloro groups on the benzoxazole moiety, indicating broad substrate tolerance.
Q: Is the catalyst loading economically viable for large-scale production?
A: The method utilizes a small amount of palladium catalyst (molar ratio 0.05-0.15:1) relative to the substrate, which significantly lowers metal costs compared to stoichiometric reagent methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the Pd/norbornene system described in CN107382894B for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle sensitive palladium-catalyzed reactions under strictly controlled conditions, guaranteeing consistent quality and adherence to stringent purity specifications. With our rigorous QC labs and dedicated process chemistry team, we are committed to delivering high-purity benzoxazole derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data for our catalog compounds or to discuss route feasibility assessments for your custom synthesis needs. Together, we can optimize your supply chain and bring life-saving medicines to market faster and more efficiently.
