Advanced One-Pot Synthesis of Florfenicol Intermediates for Commercial Scale-Up
Advanced One-Pot Synthesis of Florfenicol Intermediates for Commercial Scale-Up
The pharmaceutical and agrochemical industries constantly seek more efficient pathways for producing critical active ingredients, and the synthesis of Florfenicol intermediates represents a prime area for innovation. Patent CN1097583C discloses a highly advantageous process for preparing compounds of Formula I, which serve as pivotal precursors in the manufacture of Florfenicol, a broad-spectrum antibacterial agent widely used in veterinary medicine. This technology addresses longstanding inefficiencies in traditional synthetic routes by introducing a streamlined one-pot methodology that converts serine ester derivatives directly into stable oxazoline structures. For R&D directors and procurement specialists, understanding this patented approach is essential for evaluating potential cost reductions and supply chain optimizations in the production of high-purity veterinary pharmaceutical intermediates. The method not only simplifies the operational workflow but also enhances the overall economic viability of scaling these complex chemical transformations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating Florfenicol precursors often involve multi-step sequences where the aminodiol sulfone (ADS) intermediate must be isolated and purified before proceeding to the cyclization stage. This isolation step is technically demanding because the ADS intermediate can be unstable or difficult to handle, leading to significant material loss and increased processing time. Furthermore, the requirement for separate reaction vessels and extensive solvent exchanges between the reduction and cyclization steps inflates the operational expenditure and complicates waste management protocols. In a commercial manufacturing setting, these inefficiencies translate to higher production costs, longer lead times, and a larger environmental footprint due to excessive solvent usage. The necessity to purify the intermediate also introduces additional variables that can affect the final stereochemical purity, which is critical for the biological efficacy of the end product.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a telescoped one-pot strategy that seamlessly integrates the reduction of the ester group and the subsequent cyclization with a nitrile. By conducting both reactions in the same vessel, the process eliminates the need to isolate the sensitive aminodiol intermediate, thereby preserving yield and maintaining stereochemical integrity. This methodology allows for the direct conversion of the starting serine ester into the desired oxazoline derivative with minimal handling. The operational simplicity of this route means that manufacturers can achieve substantial cost reduction in veterinary drug manufacturing by reducing labor hours, equipment utilization time, and solvent volumes. The ability to drive the reaction to completion without intermediate workup represents a significant technological leap forward for industrial chemists aiming to optimize their production lines.
Mechanistic Insights into One-Pot Reductive Cyclization
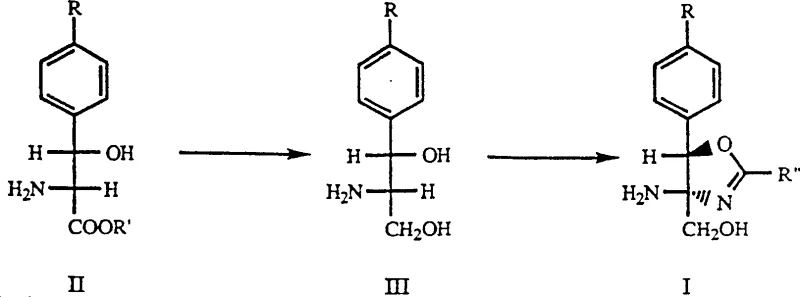
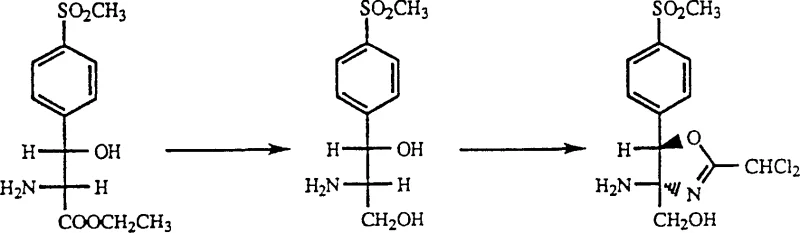
The core of this innovative process lies in the careful selection of reaction conditions that allow two distinct chemical transformations to occur sequentially without interference. Initially, the serine ester precursor (Formula II) undergoes reduction using a borohydride species, such as potassium borohydride, in a protic solvent like methanol. This step selectively reduces the ester functionality to a primary alcohol, generating the aminodiol species (Formula III) in situ. The choice of reducing agent is critical, as it must be potent enough to drive the reduction efficiently while remaining compatible with the other functional groups present on the aromatic ring, such as the methylsulfonyl moiety. Following the reduction, the reaction mixture is treated to remove the bulk solvent and quench excess reducing agent, often using glycerol, which prepares the system for the next phase without requiring a full isolation of the intermediate.
Once the reduction is complete, a nitrile component (Formula IV), such as benzonitrile or dichloroacetonitrile, is introduced directly into the same reaction vessel. Under elevated temperatures, typically ranging from 25°C to 115°C depending on the specific nitrile used, the aminodiol undergoes a cyclization reaction to form the oxazoline ring. This intramolecular condensation is driven by the nucleophilic attack of the amino and hydroxyl groups on the nitrile carbon. The process is robust enough to tolerate various substituents on the nitrile, allowing for the synthesis of diverse oxazoline derivatives. The mechanistic elegance of this route ensures that the stereochemistry established in the starting material is preserved throughout the transformation, resulting in high-purity intermediates suitable for downstream pharmaceutical applications.
How to Synthesize Florfenicol Oxazoline Intermediates Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and purity while minimizing byproduct formation. The protocol begins with the suspension of the reducing agent in a suitable alcohol solvent, followed by the addition of the serine ester substrate. Monitoring the reduction progress via HPLC is recommended to ensure complete conversion before proceeding to the cyclization step. Once the reduction is confirmed, the solvent is removed, and the nitrile is added to the residue. The mixture is then heated for an extended period, typically around 18 hours, to ensure full cyclization. Detailed standard operating procedures for this specific transformation are outlined below to guide process engineers in replicating these results.
- Reduce the serine ester precursor (Formula II) using potassium borohydride in a protic solvent like methanol to generate the aminodiol intermediate.
- Remove the reaction solvent via distillation and add glycerol to quench excess reducing agent before heating the mixture.
- Introduce the nitrile component (Formula IV) directly into the same vessel and maintain elevated temperature to effect cyclization into the final oxazoline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this one-pot synthesis method offers compelling strategic benefits that extend beyond simple chemistry. By eliminating the isolation of the intermediate ADS, the process drastically simplifies the manufacturing workflow, which directly correlates to reduced operational overheads and faster throughput times. The reduction in unit operations means less equipment is tied up per batch, allowing facilities to increase their production capacity without significant capital investment in new reactors. Furthermore, the simplified workup, which often involves mere filtration and washing of the precipitated product, reduces the demand for specialized purification infrastructure and skilled labor. These factors combine to create a more resilient and cost-effective supply chain for critical veterinary drug intermediates.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the elimination of the intermediate isolation step, which traditionally consumes large volumes of solvents and requires extensive energy for drying and purification. By telescoping the reduction and cyclization, manufacturers can achieve substantial cost savings through reduced solvent purchase and disposal costs. Additionally, the higher overall yield resulting from avoiding material loss during isolation translates directly into a lower cost of goods sold. The use of common, inexpensive reagents like potassium borohydride and methanol further ensures that raw material costs remain low and predictable, providing a stable financial baseline for long-term production planning.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials, such as serine esters and common nitriles, mitigates the risk of supply disruptions that often plague complex synthetic routes dependent on exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent systems, ensures consistent production output even when minor variations in raw material quality occur. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, preventing costly production stoppages. Moreover, the simplified process flow reduces the likelihood of human error during complex transfer operations, further stabilizing the supply chain and ensuring on-time delivery of high-quality intermediates.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns perfectly with modern green chemistry principles by minimizing waste generation and solvent usage. The ability to recover and reuse solvents like methanol via distillation adds another layer of sustainability and cost efficiency. Scaling this process from pilot plant to commercial production is straightforward because it avoids the bottlenecks associated with filtering and drying unstable intermediates on a large scale. The reduced environmental footprint not only lowers compliance costs but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for global pharmaceutical companies seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific production needs. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: The primary advantage is the elimination of the isolation step for the unstable aminodiol sulfone (ADS) intermediate. By performing the reduction and cyclization in a single vessel, the process significantly reduces solvent consumption, processing time, and potential yield losses associated with purification.
Q: Which reducing agents are compatible with this protocol?
A: The patent specifies several compatible hydride reducing agents, including sodium borohydride (NaBH4), calcium borohydride, lithium borohydride, and preferably potassium borohydride (KBH4). These reagents effectively reduce the ester functionality to the required alcohol without compromising the stereochemistry.
Q: How is the reaction workup simplified in this new process?
A: The workup is simplified because the final oxazoline product often precipitates directly from the reaction mixture upon cooling or addition of water. This allows for recovery via simple filtration and washing, avoiding complex extraction or chromatographic purification steps typically required for sensitive intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Florfenicol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the competitive landscape of veterinary pharmaceuticals. Our team of expert chemists has extensively analyzed technologies like the one described in CN1097583C to ensure we can deliver superior intermediates with consistent quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Florfenicol intermediate meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
