Advanced Synthesis of 2,6-Dihydroxytoluene: Green Chemistry for High-Purity Pharmaceutical Intermediates
The chemical industry is currently undergoing a paradigm shift towards sustainable manufacturing, driven by stringent environmental regulations and the need for cost-effective production of high-value intermediates. A pivotal development in this arena is detailed in patent CN113214051A, which discloses a novel method for synthesizing 2,6-dihydroxytoluene by effectively utilizing waste acid. This technology addresses the critical bottlenecks of traditional synthesis routes, specifically the excessive generation of hazardous waste and low overall yields that have long plagued the production of this essential pharmaceutical and agrochemical building block. By integrating microchannel reaction technology with a closed-loop acid recycling system, the process transforms a linear, waste-intensive workflow into a circular, efficient operation. For R&D directors and supply chain leaders, this represents a significant opportunity to secure a reliable 2,6-dihydroxytoluene supplier capable of delivering high-purity materials while adhering to green chemistry principles. The implications extend beyond mere compliance; they offer a tangible pathway to reducing the total cost of ownership for complex fine chemical intermediates through process intensification and waste minimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,6-dihydroxytoluene has been fraught with technical and environmental challenges that hinder scalable industrial production. Traditional routes, such as the hydrogenation of 2,6-dinitrotoluene or the sulfonation of 4-methyl benzoic acid, often require extreme conditions involving high temperatures and pressures, leading to significant energy consumption and equipment corrosion. Specifically, the conventional route starting from 3-chloro-2-methylaniline, while chemically feasible, suffers from severe inefficiencies in its downstream processing. As illustrated in the reaction scheme below, the standard process generates substantial quantities of acidic wastewater and requires multiple rounds of decolorization using activated carbon, which not only lowers the yield through adsorption losses but also creates a secondary solid waste stream that is costly to dispose of.
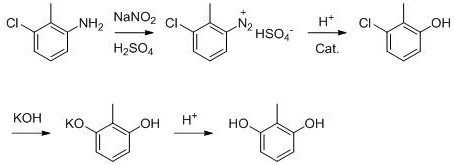
Furthermore, the reliance on batch processing for the diazotization step in conventional methods poses inherent safety risks due to the accumulation of unstable diazonium salts and the difficulty in controlling exothermic reactions. The yield in these legacy processes typically hovers around 60-65%, meaning that over one-third of the raw material value is lost to side reactions or purification steps. For procurement managers, this inefficiency translates directly into higher raw material costs and volatile supply availability. The extensive use of strong acids and bases without effective recovery mechanisms results in a heavy environmental burden, with traditional plants generating approximately 35 tons of acidic wastewater per ton of product. This level of waste generation is increasingly unsustainable in modern regulatory environments, forcing manufacturers to seek alternative technologies that can decouple production growth from environmental impact.
The Novel Approach
The innovative method described in patent CN113214051A fundamentally reengineers the synthesis pathway by introducing a continuous flow microchannel reactor for the critical diazotization step and implementing a robust acid recycling strategy. Instead of discarding the sulfuric acid solution after the hydrolysis of the diazonium salt, the process captures and reuses this acidic stream for subsequent batches, maintaining an optimal concentration of 30-50% for effective hydrolysis. This closed-loop approach drastically reduces the consumption of fresh sulfuric acid and virtually eliminates the discharge of acidic wastewater, addressing one of the most significant pain points in fine chemical manufacturing. The transition from batch to continuous flow in the initial stage allows for precise control over reaction parameters, such as temperature and residence time, ensuring that the unstable diazonium intermediate is consumed immediately, thereby enhancing safety and selectivity.
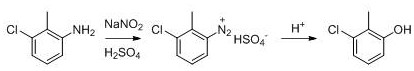
In addition to the upstream improvements, the downstream refining process has been optimized to replace the traditional activated carbon decolorization with a solvent extraction technique using dichloroethane or toluene. This modification prevents the physical adsorption loss of the product, which is a common issue with activated carbon treatments, thereby pushing the total yield up to the 80-90% range. The elimination of activated carbon also removes the generation of hazardous solid waste, simplifying the waste management protocol and reducing associated disposal costs. For supply chain heads, this means a more predictable and stable production output with fewer interruptions for waste treatment maintenance. The combination of yield enhancement and waste reduction creates a compelling economic case, positioning this technology as a superior choice for cost reduction in pharmaceutical intermediate manufacturing where margin compression is a constant challenge.
Mechanistic Insights into Microchannel Diazotization and Alkali Fusion
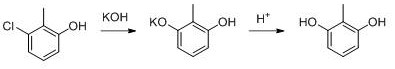
The core of this technological breakthrough lies in the precise manipulation of reaction kinetics within a microchannel environment. In the diazotization of 3-chloro-2-methylaniline, the reaction is highly exothermic and the resulting diazonium salt is thermally unstable. By utilizing a microchannel reactor, the surface-area-to-volume ratio is significantly increased, allowing for instantaneous heat removal and preventing local hot spots that could lead to decomposition or explosive runaway. The reaction is conducted at a controlled temperature of 0-20°C with a residence time of merely 6-20 seconds, which is sufficient to complete the conversion while minimizing side reactions. The immediate transfer of the diazo liquid into the hydrolysis zone containing recycled sulfuric acid ensures that the intermediate is consumed as soon as it is formed, maximizing the efficiency of the transformation into 3-chloro-2-methylphenol.
Following the formation of the phenol intermediate, the process employs a high-pressure alkali fusion step catalyzed by cuprous chloride and sodium sulfite. This step is critical for the nucleophilic substitution of the chlorine atom with a hydroxyl group. The use of a specific catalyst system under elevated pressure (0.8-1.0 MPa) and temperature (170-180°C) facilitates the breaking of the carbon-chlorine bond, which is otherwise resistant to hydrolysis under mild conditions. The subsequent acidification using the recovered acidic wastewater from the previous step not only neutralizes the reaction mixture but also precipitates the crude product. The final purification via solvent extraction leverages the differential solubility of the product and impurities, ensuring that the final 2,6-dihydroxytoluene achieves a purity of 99.0-99.5% without the need for chromatographic separation or carbon treatment. This mechanistic elegance ensures that impurity profiles are tightly controlled, a key requirement for R&D directors validating materials for clinical applications.
How to Synthesize 2,6-Dihydroxytoluene Efficiently
The synthesis of 2,6-dihydroxytoluene via this patented route involves a sequence of highly optimized unit operations designed for maximum efficiency and minimal waste. The process begins with the preparation of reactant streams where 3-chloro-2-methylaniline is dissolved in sulfuric acid and sodium nitrite is prepared as an aqueous solution. These streams are then fed into a microchannel reactor where the diazotization occurs under strictly controlled thermal conditions. The resulting diazonium solution is directly hydrolyzed using recycled acid, and the organic product is extracted. The second phase involves converting the extracted phenol into its sodium salt, followed by the high-pressure catalytic fusion. Finally, the reaction mass is acidified and refined through solvent extraction to isolate the pure product. The detailed standardized synthesis steps, including specific flow rates, mixing ratios, and safety protocols required for commercial implementation, are outlined in the guide below.
- Perform microchannel diazotization of 3-chloro-2-methylaniline with sodium nitrite at 0-20°C, followed by hydrolysis using recycled sulfuric acid.
- Conduct high-pressure alkali fusion of the intermediate phenol sodium salt at 170-180°C using cuprous chloride catalyst.
- Execute acidification using recovered acid and solvent extraction refining to achieve 99.0-99.5% purity without activated carbon.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this waste-acid recycling technology offers profound strategic advantages that go beyond simple unit price negotiations. The primary value driver is the drastic reduction in variable production costs associated with waste management and raw material consumption. By recycling sulfuric acid and eliminating the need for activated carbon, the process removes two major cost centers from the P&L statement. The reduction in acidic wastewater generation by 30-40 tons per ton of product translates into significantly lower effluent treatment fees and reduced regulatory compliance risks. Furthermore, the increase in yield from roughly 60% to nearly 90% means that less raw material is required to produce the same amount of finished goods, effectively lowering the cost of goods sold (COGS) and improving gross margins.
- Cost Reduction in Manufacturing: The elimination of activated carbon usage removes both the purchase cost of the filtering agent and the disposal cost of the hazardous carbon slag, which traditionally amounts to 0.5 tons per ton of product. Additionally, the recycling of sulfuric acid reduces the demand for fresh acid, a commodity subject to price volatility. The higher yield ensures that the fixed costs of the plant are amortized over a larger volume of saleable product, driving down the unit cost. These factors combine to create a leaner manufacturing model that is more resilient to market fluctuations in raw material pricing.
- Enhanced Supply Chain Reliability: The use of continuous flow technology in the diazotization step enhances process stability and reduces the risk of batch failures due to thermal runaway, which are common in large-scale batch reactors. This reliability ensures consistent delivery schedules, a critical factor for pharmaceutical customers who operate on tight Just-In-Time inventory models. The simplified post-treatment process, which avoids the time-consuming filtration and regeneration of activated carbon beds, shortens the overall cycle time per batch. This increased throughput capacity allows suppliers to respond more agilely to spikes in demand without requiring massive capital investment in new reactor vessels.
- Scalability and Environmental Compliance: The modular nature of microchannel reactors allows for easy scale-up by numbering up rather than scaling up vessel size, maintaining the same heat and mass transfer characteristics at commercial volumes. This facilitates the commercial scale-up of complex pharmaceutical intermediates with minimal re-engineering. From an environmental perspective, the near-zero discharge of acidic wastewater aligns with the strictest global environmental standards, future-proofing the supply chain against tightening regulations. This sustainability profile is increasingly becoming a prerequisite for inclusion in the vendor lists of top-tier multinational corporations committed to Scope 3 emission reductions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,6-dihydroxytoluene using this advanced waste-acid recycling methodology. These answers are derived directly from the experimental data and process descriptions found in patent CN113214051A, providing transparency into the capabilities and limitations of the technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines or for procurement teams assessing the long-term viability of the supply source.
Q: How does the new process reduce environmental impact compared to conventional methods?
A: The patented method recycles sulfuric acid from the hydrolysis step for subsequent batches, reducing acidic wastewater generation by approximately 30-40 tons per ton of product. Additionally, it eliminates the use of activated carbon for refining, removing solid waste disposal issues.
Q: What is the expected yield and purity of 2,6-dihydroxytoluene using this method?
A: The optimized process achieves a total reaction yield of 80-90%, a significant improvement over the conventional 60-65%. The final product purity consistently reaches 99.0-99.5%, meeting stringent pharmaceutical intermediate standards.
Q: Why is microchannel technology used in the diazotization step?
A: Microchannel reactors provide superior heat transfer and mixing efficiency, allowing the highly exothermic diazotization reaction to be controlled precisely at 0-20°C with a short residence time of 6-20 seconds, enhancing safety and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dihydroxytoluene Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient chemical processes is not just an environmental imperative but a business necessity. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN113214051A are fully realized in practical, large-scale manufacturing. Our facilities are equipped with state-of-the-art microchannel reactors and high-pressure fusion units, allowing us to implement this waste-acid recycling technology with precision. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,6-dihydroxytoluene meets the 99.0-99.5% purity threshold required for sensitive pharmaceutical applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain. By leveraging our expertise in process intensification, we can offer a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how the reduction in waste and improvement in yield translates to bottom-line savings. We encourage you to contact us to request specific COA data and route feasibility assessments, ensuring that your project moves forward with a supply partner dedicated to quality, sustainability, and long-term reliability.
