Advanced Synthesis of Trifluoromethyl 1,2,4-Triazines for Scalable Pharmaceutical Manufacturing
Advanced Synthesis of Trifluoromethyl 1,2,4-Triazines for Scalable Pharmaceutical Manufacturing
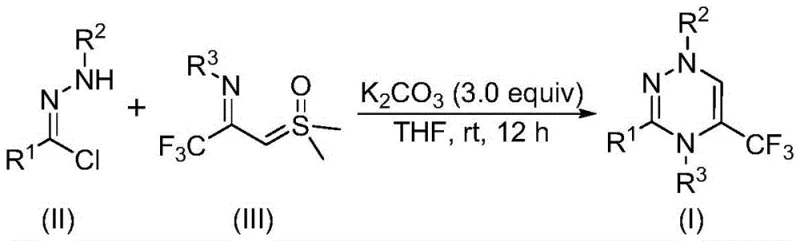
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to construct nitrogen-rich heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in traditional heterocyclic synthesis. This innovation leverages a unique cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive potassium carbonate. For R&D directors and procurement specialists, this represents a significant shift towards greener, more cost-effective manufacturing processes that eliminate the need for toxic heavy metal catalysts and harsh reaction conditions. The ability to synthesize these complex scaffolds under ambient air conditions at room temperature not only simplifies operational protocols but also drastically reduces energy consumption and safety risks associated with high-pressure or high-temperature reactors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of 1,2,4-triazine skeletons has relied heavily on the condensation of amidrazones with 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. These traditional pathways often suffer from significant drawbacks, including the necessity for pre-functionalized substrates that are difficult and expensive to prepare. Furthermore, conventional cyclization methods frequently require elevated temperatures, inert atmospheres, or stoichiometric amounts of hazardous reagents, which complicates waste management and increases the overall cost of goods sold. The structural diversity achievable through these older methods is often limited, restricting the chemical space available for medicinal chemists to explore during lead optimization campaigns. Additionally, the purification of products from these complex reaction mixtures can be arduous, leading to lower isolated yields and extended production timelines that negatively impact supply chain efficiency.
The Novel Approach
In stark contrast, the methodology disclosed in CN116253692A utilizes readily available chlorohydrazones and trifluoroacetyl sulfur ylides as building blocks, reacting them in the presence of potassium carbonate within common organic solvents like tetrahydrofuran. This novel approach operates efficiently at mild temperatures ranging from 20°C to 40°C and tolerates exposure to air, removing the stringent requirement for nitrogen or argon protection. The reaction mechanism proceeds through the generation of a nitrile imine intermediate, which then engages in a synergistic [3+3] cycloaddition with the sulfur ylide to forge the triazine ring while eliminating dimethyl sulfoxide. This streamlined process not only enhances atom economy but also allows for the introduction of diverse substituents at multiple positions on the triazine core, thereby expanding the library of accessible analogs for biological evaluation. The simplicity of the post-treatment, involving basic filtration and column chromatography, further underscores the practical utility of this method for both laboratory discovery and potential industrial application.
Mechanistic Insights into Potassium Carbonate-Promoted Cycloaddition
The mechanistic elegance of this transformation lies in the dual role of the reagents and the mild base promoter. Initially, potassium carbonate acts as a dehydrohalogenation agent, abstracting hydrogen chloride from the chlorohydrazone precursor to generate a highly reactive nitrile imine species in situ. This transient intermediate is crucial for the subsequent ring-closing event. Simultaneously, the trifluoroacetyl sulfur ylide serves as a dipolarophile or a 1,3-dipole equivalent, depending on the specific electronic nature of the substituents. The interaction between the nitrile imine and the sulfur ylide is hypothesized to proceed via a concerted [3+3] cycloaddition pathway, although stepwise nucleophilic addition followed by intramolecular substitution cannot be entirely ruled out. The driving force for the reaction is partly attributed to the formation of the stable aromatic triazine system and the expulsion of dimethyl sulfoxide as a leaving group. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters, as the stability of the nitrile imine and the nucleophilicity of the ylide are sensitive to solvent polarity and base strength.
From an impurity control perspective, the mildness of the reaction conditions plays a pivotal role in minimizing side reactions. Traditional methods often lead to polymerization of reactive intermediates or over-oxidation of sensitive functional groups due to harsh thermal conditions. By maintaining the reaction at room temperature, the kinetic energy available for non-productive collisions is reduced, thereby favoring the desired cycloaddition pathway. Furthermore, the use of potassium carbonate, a weak inorganic base, prevents the degradation of acid-sensitive moieties that might be present on the aromatic rings of the starting materials. The patent data indicates that a wide range of functional groups, including halogens, alkoxy groups, and trifluoromethyl groups, are well-tolerated, suggesting a clean reaction profile with minimal byproduct formation. This high level of chemoselectivity simplifies downstream purification, ensuring that the final active pharmaceutical ingredient (API) intermediates meet stringent purity specifications required by regulatory bodies.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
The synthesis protocol outlined in the patent provides a straightforward recipe for accessing these valuable heterocycles. The process begins with the precise weighing of chlorohydrazone and trifluoroacetyl sulfur ylide, typically in a molar ratio of 1:2, along with three equivalents of potassium carbonate. These solids are suspended in anhydrous tetrahydrofuran, which has been identified as the optimal solvent for maximizing conversion rates. The mixture is then stirred vigorously at ambient temperature for a period of 10 to 14 hours, allowing sufficient time for the cycloaddition to reach completion. Monitoring the reaction progress via thin-layer chromatography (TLC) is recommended to determine the exact endpoint. Upon completion, the solid inorganic salts are removed by simple filtration, and the filtrate is concentrated. The crude residue is then subjected to flash column chromatography on silica gel to isolate the pure trifluoromethyl-substituted 1,2,4-triazine product. Detailed standardized synthesis steps are provided in the guide below.
- Mix potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent like THF.
- Stir the reaction mixture at room temperature (20-40°C) for 10-14 hours under an air atmosphere.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final triazine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical benefits. The elimination of expensive transition metal catalysts, such as palladium or copper, which are common in cross-coupling approaches to similar scaffolds, results in significant raw material cost savings. Moreover, the absence of heavy metals removes the need for costly and time-consuming metal scavenging steps during purification, which are often mandatory to meet residual metal limits in pharmaceutical products. The use of potassium carbonate, a commodity chemical, as the promoter further drives down the cost of goods, making the process highly attractive for large-scale manufacturing. Additionally, the reaction's tolerance to air and moisture reduces the capital expenditure required for specialized equipment like gloveboxes or rigorous drying systems, allowing for production in standard multipurpose reactors.
- Cost Reduction in Manufacturing: The process utilizes cheap, commercially available starting materials and avoids the use of precious metal catalysts, leading to substantial cost savings in raw material procurement. The simplified workup procedure, which involves basic filtration rather than complex extractions or distillations, reduces labor costs and solvent consumption. Furthermore, the high yields reported in the patent examples, ranging significantly above 60% and reaching up to 87% for certain substrates, ensure efficient utilization of resources and minimize waste disposal costs associated with low-yielding processes.
- Enhanced Supply Chain Reliability: Since all key reagents, including chlorohydrazones and sulfur ylides, can be synthesized from readily available bulk chemicals like acyl chlorides, hydrazines, and trifluoroacetic acid, the supply chain is less vulnerable to shortages of exotic reagents. The robustness of the reaction conditions means that production can be scheduled with greater flexibility, as there is no need to coordinate complex inert gas supplies or maintain strict anhydrous environments. This reliability translates to shorter lead times for custom synthesis projects and a more predictable delivery schedule for key intermediates, ensuring continuity of supply for downstream drug development programs.
- Scalability and Environmental Compliance: The mild reaction conditions and the use of non-toxic inorganic bases align perfectly with green chemistry principles, facilitating easier regulatory approval for manufacturing sites. The process generates dimethyl sulfoxide as a byproduct, which is relatively benign and easy to handle compared to the hazardous waste streams generated by traditional methods. The ability to scale the reaction from milligram to gram levels without loss of efficiency demonstrates its potential for kilogram and ton-scale production. This scalability ensures that the technology can support the growing demand for trifluoromethyl-containing drugs without requiring extensive process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on substrate compatibility and operational parameters. Understanding these details is essential for process development teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What are the key advantages of this new triazine synthesis method?
A: The method operates at room temperature in air without heavy metal catalysts, uses cheap potassium carbonate, and offers high yields (up to 87%) with broad substrate tolerance.
Q: Can this process be scaled for industrial production?
A: Yes, the patent explicitly states the method is convenient for operation and can be expanded to gram levels, indicating strong potential for commercial scale-up due to mild conditions.
Q: What is the role of the sulfur ylide in this reaction?
A: The trifluoroacetyl sulfur ylide acts as a 3-atom component that undergoes a synergistic [3+3] cycloaddition with the nitrile imine intermediate to form the triazine ring.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient heterocyclic chemistry in modern drug discovery. Our team of expert chemists has thoroughly analyzed the potential of the trifluoromethyl 1,2,4-triazine scaffold and is fully equipped to leverage this patented methodology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to pilot plant is seamless. Our state-of-the-art facilities are designed to handle fluorinated chemistry safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee the quality of every batch we deliver.
We invite you to collaborate with us to unlock the full potential of this innovative synthesis route for your pipeline. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss your project specifics,索取 specific COA data, and receive comprehensive route feasibility assessments that will accelerate your development timeline and optimize your manufacturing budget.
