Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Commercial Scale-Up
Advanced Metal-Free Synthesis of Trifluoromethyl 1,2,4-Triazines for Commercial Scale-Up
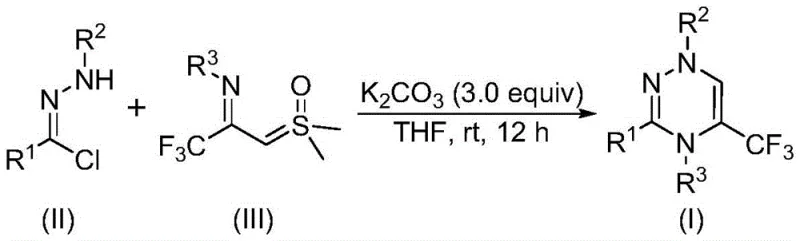
The pharmaceutical and agrochemical industries are constantly seeking efficient pathways to construct nitrogen-rich heterocycles, particularly those incorporating fluorine atoms to enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl-substituted 1,2,4-triazine compounds, addressing critical bottlenecks in current synthetic methodologies. This innovation utilizes a synergistic [3+3] cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, promoted by inexpensive potassium carbonate. Unlike traditional routes that often demand harsh conditions or toxic transition metals, this novel approach operates efficiently at room temperature under an air atmosphere. For R&D directors and procurement managers alike, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value intermediates used in anticancer, antifungal, and antimalarial drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has relied heavily on the condensation of amidrazones with 1,2-diketones or alkynes, as well as multicomponent reactions involving hydrazides and dicarbonyl compounds. These conventional pathways frequently suffer from significant drawbacks, including the necessity for pre-functionalized substrates that are difficult and expensive to synthesize. Furthermore, many established cyclization methods require elevated temperatures, inert atmospheres, or stoichiometric amounts of hazardous reagents, which complicates process safety and waste management. The structural diversity achievable through these older methods is often limited, restricting the ability of medicinal chemists to rapidly explore structure-activity relationships (SAR) around the triazine scaffold. Consequently, the search for a more versatile and operationally simple synthesis has been a priority for leading chemical manufacturers aiming to streamline their supply chains.
The Novel Approach
The methodology disclosed in the patent fundamentally alters the synthetic landscape by employing readily available chlorohydrazones and trifluoroacetyl sulfur ylides as building blocks. This route eliminates the need for complex substrate preparation, allowing for the direct assembly of the heterocyclic ring with high atom economy. The use of potassium carbonate as a promoter is particularly advantageous; it is a benign, odorless, and non-toxic inorganic salt that effectively drives the reaction without introducing heavy metal contamination. By conducting the reaction in common organic solvents like tetrahydrofuran (THF) at ambient temperatures (20-40°C), the process drastically reduces energy consumption and operational complexity. This simplicity not only accelerates the timeline for lead optimization but also paves the way for seamless technology transfer from the laboratory to pilot and commercial scales.
Mechanistic Insights into Potassium Carbonate-Promoted [3+3] Cycloaddition
The mechanistic pathway of this transformation is both elegant and efficient, proceeding through the in situ generation of a reactive nitrile imine intermediate. Under the basic conditions provided by potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to form the nitrile imine species, which serves as a crucial 1,3-dipole. This intermediate then engages in a synergistic [3+3] cycloaddition with the trifluoroacetyl sulfur ylide, a reaction that constructs the six-membered 1,2,4-triazine ring while eliminating a molecule of dimethyl sulfoxide (DMSO). Alternatively, the reaction may proceed via a stepwise mechanism involving intermolecular nucleophilic addition followed by intramolecular nucleophilic substitution. Understanding this mechanism is vital for process chemists, as it highlights the robustness of the reaction against moisture and oxygen, explaining why strict inert gas protection is unnecessary. This tolerance simplifies reactor setup and reduces the risk of batch failures due to atmospheric leaks.
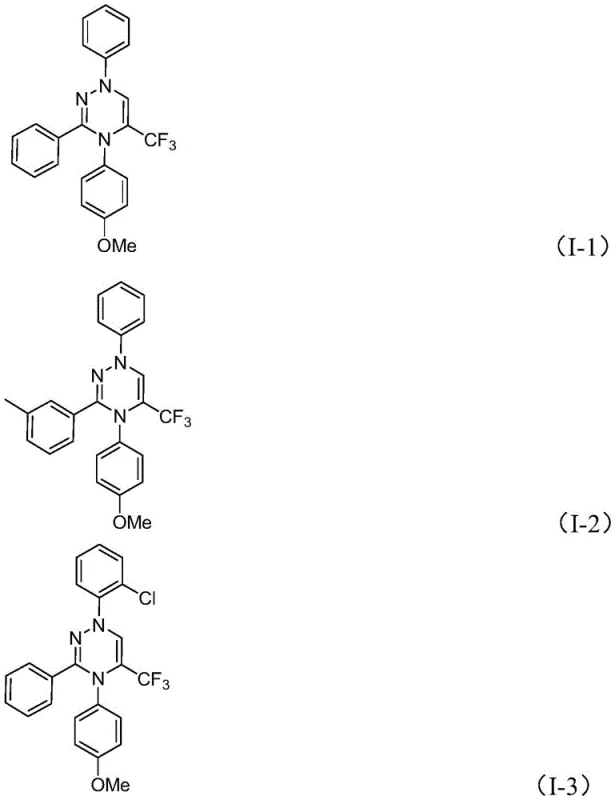
From an impurity control perspective, the absence of transition metal catalysts is a decisive factor in ensuring high product purity. Traditional metal-catalyzed couplings often leave behind trace residues that require rigorous and costly purification steps, such as scavenging or recrystallization, to meet regulatory standards for pharmaceutical intermediates. In this metal-free protocol, the primary byproducts are inorganic salts and DMSO, which are easily removed during the aqueous workup or column chromatography. The broad substrate scope demonstrated in the patent, accommodating various substituents like methyl, methoxy, halogens, and trifluoromethyl groups on the aromatic rings, further underscores the versatility of this mechanism. This flexibility allows for the rapid generation of diverse libraries of trifluoromethyl triazines, enabling comprehensive SAR studies without the need to re-optimize reaction conditions for each new analog.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
To implement this synthesis effectively, operators should adhere to the optimized molar ratios and conditions detailed in the patent examples. The standard protocol involves mixing the chlorohydrazone, trifluoroacetyl sulfur ylide, and 3.0 equivalents of potassium carbonate in THF, followed by stirring at room temperature for approximately 12 hours. Post-reaction processing is straightforward, typically involving filtration to remove inorganic salts, followed by silica gel treatment and purification via column chromatography. This streamlined workflow minimizes unit operations and solvent usage, contributing to a lower overall cost of goods sold (COGS). For detailed standardized synthesis steps and specific experimental parameters, please refer to the guide below.
- Mix potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent such as THF.
- Stir the reaction mixture at room temperature (20-40°C) for 10-14 hours under an air atmosphere.
- Filter the reaction mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of expensive and potentially supply-constrained transition metal catalysts directly translates to significant cost reduction in API manufacturing. By relying on commodity chemicals like potassium carbonate and widely available organic solvents, the process mitigates the risk of raw material price volatility and ensures a stable supply chain. Furthermore, the mild reaction conditions reduce the energy footprint of the manufacturing process, aligning with global sustainability goals and potentially lowering utility costs associated with heating and cooling large-scale reactors.
- Cost Reduction in Manufacturing: The removal of heavy metal catalysts from the process flow eliminates the need for specialized metal scavengers and extensive purification protocols, which are often major cost drivers in fine chemical production. Additionally, the use of inexpensive inorganic bases instead of strong organic bases or exotic reagents lowers the direct material costs substantially. The high atom economy of the [3+3] cycloaddition ensures that a greater proportion of the starting mass is converted into the desired product, minimizing waste disposal fees and maximizing resource efficiency.
- Enhanced Supply Chain Reliability: The starting materials, including chlorohydrazones and sulfur ylides, are derived from commercially available precursors such as acyl chlorides, hydrazines, and trifluoroacetic acid derivatives. This reliance on bulk chemicals ensures that the supply chain is robust and less susceptible to disruptions compared to processes requiring bespoke or proprietary catalysts. The ability to run the reaction under air without stringent moisture control further enhances operational reliability, reducing the likelihood of batch rejection due to environmental factors and ensuring consistent delivery timelines to downstream customers.
- Scalability and Environmental Compliance: The protocol has been successfully demonstrated at the gram level with excellent yields, indicating strong potential for scale-up to kilogram and tonne quantities without fundamental changes to the chemistry. The absence of toxic heavy metals simplifies wastewater treatment and waste stream management, facilitating compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces the environmental impact but also streamlines the regulatory approval process for new drug applications, as the impurity profile is cleaner and easier to characterize.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of trifluoromethyl substituted 1,2,4-triazine compounds. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is essential for partners looking to integrate this technology into their existing manufacturing portfolios or R&D pipelines.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method avoids heavy metal catalysts, operates at room temperature in air, and uses cheap, non-toxic potassium carbonate, significantly simplifying purification and reducing environmental impact.
Q: What is the substrate scope for this trifluoromethyl triazine synthesis?
A: The reaction tolerates a wide range of substituents including alkyl, phenyl, naphthyl, and furyl groups, with various electron-donating and withdrawing groups like methyl, methoxy, halogens, and trifluoromethyl.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the protocol is designed for scalability, utilizing inexpensive reagents and mild conditions that facilitate safe expansion from gram-level laboratory synthesis to commercial production volumes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and assay. Whether you require custom synthesis of novel analogs or large-scale supply of established intermediates, our infrastructure is designed to support your growth from early-stage discovery to full commercialization.
We invite you to collaborate with us to leverage this innovative metal-free synthesis for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and efficiency.
