Scalable Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Pharmaceutical Applications
Scalable Synthesis of Trifluoromethyl 1,2,4-Triazines for Advanced Pharmaceutical Applications
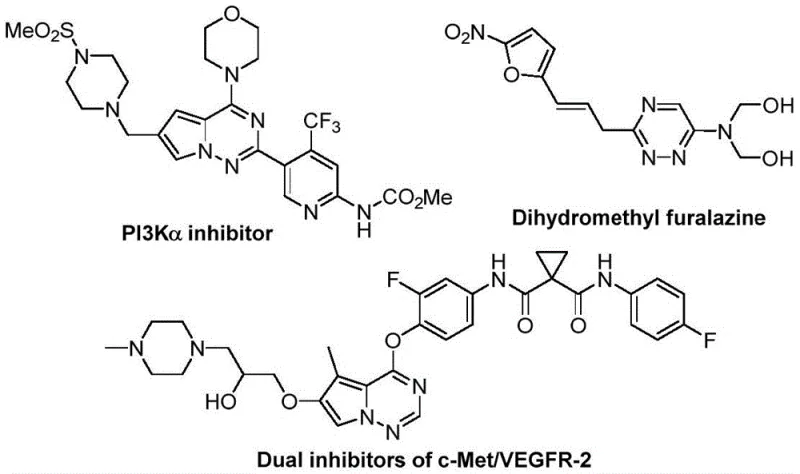
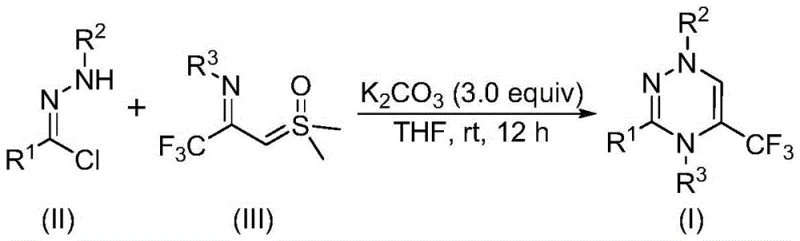
The pharmaceutical industry continuously seeks efficient pathways to construct nitrogen-rich heterocycles, particularly those bearing trifluoromethyl groups which enhance metabolic stability and bioavailability. Patent CN116253692A introduces a groundbreaking preparation method for trifluoromethyl substituted 1,2,4-triazine compounds that addresses critical bottlenecks in modern medicinal chemistry. This technology leverages a novel cycloaddition strategy between chlorohydrazones and trifluoroacetyl sulfur ylides, facilitated by inexpensive potassium carbonate. Unlike traditional methods that often rely on toxic transition metals or extreme thermal conditions, this process operates efficiently at room temperature under an air atmosphere. For R&D directors and procurement specialists, this represents a paradigm shift towards greener, more cost-effective manufacturing of high-value pharmaceutical intermediates. The structural diversity achievable through this route allows for the rapid generation of libraries for drug discovery, specifically targeting anticancer and antifungal applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 1,2,4-triazine core has been fraught with synthetic challenges that hinder large-scale adoption. Traditional condensation reactions between amidrazones and 1,2-diketones often suffer from low atom economy and require rigorous exclusion of moisture and oxygen. Furthermore, many existing protocols necessitate the use of precious metal catalysts or strong oxidants, which introduce significant impurities that are difficult to remove to meet stringent stringent purity specifications required for API production. The need for specialized equipment to handle hazardous reagents increases capital expenditure and operational complexity. Additionally, the substrate scope in conventional methods is frequently limited, restricting the ability of chemists to explore diverse chemical space around the triazine scaffold. These factors collectively contribute to extended lead times and inflated costs in the development of new therapeutic agents.
The Novel Approach
The methodology disclosed in CN116253692A offers a robust alternative by utilizing a [3+3] cycloaddition mechanism that is both operationally simple and chemically elegant. By employing chlorohydrazones and trifluoroacetyl sulfur ylides as building blocks, the reaction proceeds smoothly in common organic solvents like tetrahydrofuran (THF). The use of potassium carbonate as a promoter is particularly advantageous; it is non-toxic, odorless, and significantly cheaper than organic bases or metal catalysts. This approach not only streamlines the workflow by eliminating the need for inert gas protection but also enhances the safety profile of the manufacturing process. The reaction tolerates a wide array of functional groups, including halogens, alkoxy groups, and alkyl chains, enabling the synthesis of complex derivatives without extensive protecting group strategies. This flexibility is crucial for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Metal-Free [3+3] Cycloaddition
The core of this innovation lies in the generation of a reactive nitrile imine intermediate in situ. Under the basic conditions provided by potassium carbonate, the chlorohydrazone undergoes dehydrohalogenation to form the nitrile imine species. This highly reactive intermediate then engages in a synergistic [3+3] cycloaddition with the trifluoroacetyl sulfur ylide. The sulfur ylide acts as a three-carbon synthon, donating its electron density to close the six-membered triazine ring while extruding dimethyl sulfoxide as a benign byproduct. This mechanism avoids the formation of stable metal-complex intermediates that often plague transition-metal catalyzed reactions, thereby simplifying the downstream purification process. The absence of metal residues is a critical quality attribute for pharmaceutical ingredients, reducing the burden on analytical QC labs to detect trace heavy metals.
From an impurity control perspective, the reaction pathway is remarkably clean. The primary byproducts are inorganic salts (potassium chloride) and dimethyl sulfoxide, both of which are easily removed during standard aqueous workup or chromatographic purification. The high regioselectivity of the cycloaddition ensures that the trifluoromethyl group is installed at the desired position on the triazine ring with high fidelity. This precision minimizes the formation of regioisomers, which are notoriously difficult to separate and can compromise the efficacy of the final drug product. The mechanistic clarity provided by this patent allows process chemists to confidently optimize reaction parameters such as stoichiometry and concentration to maximize yield and throughput.
How to Synthesize Trifluoromethyl 1,2,4-Triazine Efficiently
The synthesis protocol described in the patent is designed for ease of execution in both laboratory and pilot plant settings. The procedure involves simply mixing the solid reagents and solvent, followed by stirring at ambient temperature. This simplicity reduces the training burden on operators and minimizes the risk of human error during batch preparation. The reaction time of 10 to 14 hours is compatible with standard overnight shifts, facilitating efficient scheduling in multi-purpose manufacturing facilities. Detailed standardized synthesis steps see the guide below.
- Mix potassium carbonate, chlorohydrazone, and trifluoroacetyl sulfur ylide in an organic solvent like THF.
- Stir the reaction mixture at room temperature (20-40°C) under an air atmosphere for 10-14 hours.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this technology are profound. The shift from noble metal catalysts to earth-abundant inorganic bases represents a direct reduction in raw material costs. Moreover, the elimination of heavy metals removes the need for expensive scavenging resins or specialized filtration units, further driving down the cost of goods sold (COGS). The ability to run the reaction in air rather than under nitrogen or argon significantly reduces utility costs associated with gas consumption and reactor purging cycles. These cumulative efficiencies translate into substantial cost savings in API manufacturing without compromising on quality or yield.
- Cost Reduction in Manufacturing: The replacement of expensive catalysts with potassium carbonate drastically lowers the input cost per kilogram of product. Since the reaction does not require cryogenic cooling or high-pressure equipment, energy consumption is minimized. The simplified workup procedure reduces solvent usage and waste disposal fees, contributing to a leaner and more profitable production model. These factors combined ensure a highly competitive pricing structure for the final intermediate.
- Enhanced Supply Chain Reliability: The starting materials, including chlorohydrazones and sulfur ylides, are derived from commodity chemicals that are readily available from multiple global suppliers. This diversification of the supply base mitigates the risk of shortages that often plague specialized reagents. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or environmental fluctuations. Consequently, manufacturers can offer more reliable lead times and maintain consistent inventory levels for their clients.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, aligning with increasingly strict environmental regulations worldwide. The absence of toxic heavy metals simplifies the handling of effluent streams and reduces the regulatory burden associated with discharge permits. The method has been demonstrated to scale effectively from milligram to gram quantities, indicating a clear path to ton-scale production. This scalability ensures that the technology can meet the growing demand for trifluoromethylated heterocycles in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production pipelines. The answers are derived directly from the experimental data and technical disclosures within the patent documentation.
Q: What are the key advantages of this synthesis method over traditional routes?
A: This method eliminates the need for expensive heavy metal catalysts and harsh reaction conditions. It operates at room temperature in air, using cheap inorganic bases like potassium carbonate, which significantly simplifies purification and reduces environmental impact.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly states the method can be expanded to gram levels and beyond. The use of stable, commercially available starting materials and mild conditions makes it highly amenable to commercial scale-up of complex pharmaceutical intermediates.
Q: What is the typical yield range for these trifluoromethyl triazine derivatives?
A: According to the experimental data in the patent, the reaction yields are consistently high, ranging from 62% to 87% across various substrates, demonstrating robust functional group tolerance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl 1,2,4-Triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis route for the next generation of therapeutics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry and heterocycle synthesis, ensuring that every batch meets stringent purity specifications. Our rigorous QC labs utilize advanced analytical techniques to verify the identity and quality of every intermediate, providing our partners with the confidence they need to advance their drug candidates through clinical trials.
We invite you to collaborate with us to leverage this innovative technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Together, we can accelerate the development of life-saving medicines while optimizing your supply chain efficiency.
