Advanced Synthesis of Carbon-14 Labeled Dinotefuran for Metabolic Research and Commercial Scale-Up
Advanced Synthesis of Carbon-14 Labeled Dinotefuran for Metabolic Research and Commercial Scale-Up
The development of radiolabeled agrochemicals is a cornerstone of modern environmental safety assessment and metabolic pathway analysis. Patent CN114044766B introduces a robust and versatile methodology for the synthesis of radioisotope carbon-14 labeled dinotefuran, a potent neonicotinoid insecticide. This technical disclosure addresses the critical need for high-specific-activity tracers that allow researchers to track the fate of pesticide residues in crops, soil, and livestock with unparalleled precision. By establishing a reliable synthetic route for key intermediates such as the C-14 labeled compound IM-3 and 3-aminomethyl tetrahydrofuran, this technology facilitates comprehensive metabolic studies that are essential for regulatory approval and environmental risk assessment. The innovation lies not merely in the labeling itself, but in the strategic placement of the isotope and the efficiency of the synthetic pathway, which avoids complex and costly precursor sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing radiolabeled neonicotinoids often suffer from significant drawbacks regarding regioselectivity and economic feasibility. Conventional methods may rely on random labeling techniques or the use of extremely expensive, commercially scarce labeled building blocks that drive up the cost of goods sold prohibitively. Furthermore, older synthetic routes frequently involve harsh reaction conditions that can lead to isotopic scrambling or loss of the radioactive label during workup and purification stages. The lack of a dedicated, streamlined process for introducing the carbon-14 atom at the metabolically stable yet traceable position on the tetrahydrofuran side chain has historically limited the availability of high-quality reference standards. These inefficiencies create bottlenecks in the R&D pipeline, delaying critical toxicology studies and extending the time-to-market for new agrochemical formulations.
The Novel Approach
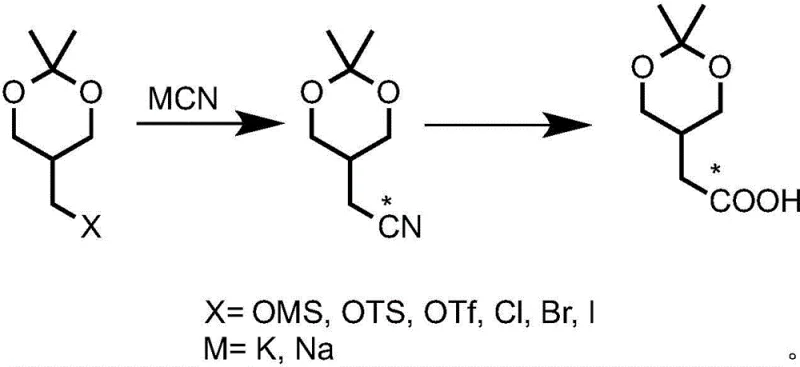
The methodology disclosed in Patent CN114044766B overcomes these hurdles by utilizing readily available dioxane derivatives as starting materials, which serve as effective masked precursors for the tetrahydrofuran ring system. The core innovation involves two distinct pathways for introducing the carbon-14 label: nucleophilic substitution with labeled cyanide or carboxylation of a Grignard reagent with labeled carbon dioxide. This dual-pathway flexibility allows manufacturers to select the most cost-effective and logistically feasible route based on the availability of labeled reagents. The process ensures that the radioactive label is incorporated early in the synthesis, maximizing atom economy and minimizing the handling of high-activity materials in later, more complex steps. This strategic design significantly enhances the overall yield and specific activity of the final dinotefuran product.
Mechanistic Insights into C-14 Labeling Strategies
The chemical elegance of this patent lies in the protection-deprotection strategy employed to construct the tetrahydrofuran ring while preserving the isotopic label. The synthesis begins with 2,2-dimethyl-5-(halogenated methyl)-1,3-dioxane, which acts as a protected form of the hydroxymethyl group. In the first labeling variant, the halogenated precursor undergoes a nucleophilic substitution reaction with potassium cyanide enriched with carbon-14. This SN2 reaction proceeds with high fidelity, installing the labeled nitrile group which is subsequently hydrolyzed to the corresponding carboxylic acid (IM-3). Alternatively, the Grignard route involves the formation of an organomagnesium intermediate which reacts with carbon-14 labeled carbon dioxide. Both pathways converge at the labeled acid intermediate, demonstrating the chemical robustness of the dioxane scaffold under diverse reaction conditions.
Following the introduction of the label, the synthetic sequence meticulously transforms the linear dioxane chain into the cyclic tetrahydrofuran structure required for dinotefuran activity. This involves reduction of the carboxylic acid to a primary alcohol, followed by acid-catalyzed cyclization which simultaneously removes the acetonide protecting group and closes the five-membered ether ring. The resulting 3-hydroxymethyl tetrahydrofuran is then activated, typically via sulfonation, to enable nucleophilic displacement by nitrogen sources such as phthalimide or azides. This step-wise construction allows for rigorous purification of intermediates, ensuring that the final radioisotope labeled dinotefuran meets the stringent purity specifications required for metabolic tracing studies, free from unlabeled impurities that could skew analytical data.
How to Synthesize Carbon-14 Labeled Dinotefuran Efficiently
The complete synthetic workflow described in the patent offers a blueprint for producing high-purity labeled standards suitable for GLP-compliant studies. The process integrates standard unit operations familiar to fine chemical manufacturers, including halogenation, cyanation, hydrolysis, reduction, and heterocycle formation. By breaking down the synthesis into discrete, manageable stages, the protocol minimizes the risk of side reactions and facilitates the isolation of pure intermediates at each step. The detailed experimental examples provided in the patent data serve as a valuable guide for optimizing reaction parameters such as temperature, solvent choice, and stoichiometry to maximize radiochemical yield.
- Convert 2,2-dimethyl-1,3-dioxane-5-methanol to the corresponding iodide or sulfonate leaving group.
- Perform nucleophilic substitution with Potassium Cyanide-14C or react Grignard reagent with Carbon-14 Dioxide to introduce the label.
- Hydrolyze the nitrile or ester to the acid, reduce to alcohol, cyclize to tetrahydrofuran, and couple with nitro-guanidine precursors.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain directors seeking to optimize the sourcing of specialized agrochemical intermediates. The reliance on commodity chemicals such as dioxane derivatives, common solvents like dichloromethane and DMF, and standard reagents implies a supply chain that is resilient and less susceptible to the volatility associated with exotic proprietary catalysts. The ability to choose between cyanide and Grignard labeling routes provides strategic flexibility, allowing production planners to mitigate risks associated with the supply of specific labeled precursors. This adaptability is crucial for maintaining continuous production schedules in the face of global supply chain disruptions.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step labeling sequences found in older methodologies translates directly into lower operational expenditures. By introducing the expensive carbon-14 label at an early stage using relatively inexpensive precursors like KCN or CO2, the process maximizes the utilization of the radioactive material. Furthermore, the use of robust protection groups reduces the formation of byproducts, thereby lowering the costs associated with downstream purification and waste disposal. The streamlined nature of the synthesis reduces labor hours and reactor occupancy time, contributing to a more favorable cost structure for high-value radiochemical products.
- Enhanced Supply Chain Reliability: The starting materials identified in this patent, such as 2,2-dimethyl-1,3-dioxane-5-methanol, are chemically stable and can be sourced from multiple vendors or synthesized in-house with ease. This diversification of raw material sources reduces dependency on single-supplier monopolies often seen in the niche radiochemical market. Additionally, the intermediates generated, such as the labeled acid IM-3, are stable enough to be stockpiled if necessary, providing a buffer against demand fluctuations. This stability ensures that critical metabolic studies can proceed without interruption due to material shortages.
- Scalability and Environmental Compliance: The synthetic steps described utilize standard organic transformations that are well-understood and easily scalable from gram to kilogram quantities in specialized facilities. The avoidance of transition metal catalysts in the key labeling steps simplifies the purification process and reduces the burden of heavy metal removal, which is a significant regulatory hurdle in pharmaceutical and agrochemical manufacturing. Moreover, the efficient atom economy of the nucleophilic substitution and carboxylation reactions minimizes chemical waste generation, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of carbon-14 labeled dinotefuran. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity on the feasibility and advantages of this specific synthetic approach for industry stakeholders.
Q: What is the primary advantage of the C-14 labeling position in this patent?
A: The labeling is specifically positioned on the side chain (acetate/acetonitrile moiety), which is critical for tracing the metabolic cleavage of the tetrahydrofuran ring in biological systems.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the route utilizes standard organic transformations such as iodination, nucleophilic substitution, and reduction, which are well-suited for scale-up in specialized radiochemical facilities.
Q: What are the key intermediates identified in Patent CN114044766B?
A: The key intermediates include the C-14 labeled compound IM-3 (a protected acetic acid derivative) and C-14 labeled 3-aminomethyl tetrahydrofuran.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dinotefuran Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality radioisotope labeled intermediates in advancing agrochemical research and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that even complex radiochemical syntheses can be translated into reliable supply chains. We maintain stringent purity specifications and operate rigorous QC labs equipped to handle radioactive materials safely, guaranteeing that every batch of dinotefuran intermediate meets the exacting standards required for metabolic tracing and regulatory submission.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for labeled agrochemicals. By leveraging our technical expertise, you can achieve Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that your next metabolic study is supported by the highest quality materials available in the market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
