Advanced One-Pot Synthesis of Dinotefuran for Commercial Scale Agrochemical Production
The global demand for next-generation neonicotinoid insecticides continues to surge, driving the need for more efficient and environmentally benign manufacturing processes for key active ingredients like Dinotefuran. Patent CN113214193A introduces a groundbreaking preparation method that fundamentally restructures the synthetic landscape for this critical agrochemical intermediate. Unlike conventional multi-step pathways that rely on hazardous reagents and generate substantial waste, this novel approach leverages a sophisticated one-pot condensation strategy. By utilizing accessible 1,3-dicarbonyl compounds and tetrahydrofuran-3-methylamine in the presence of a synergistic acid and water-absorption catalyst system, the invention achieves a dramatic simplification of the production workflow. This technical breakthrough not only enhances reaction yields but also aligns perfectly with modern green chemistry principles, offering a robust solution for large-scale industrial application.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Dinotefuran has been plagued by significant technical and economic bottlenecks that hinder efficient mass production. Traditional routes, such as the 3-tetrahydrofuryl methyl triflate method or those employing S-methyl-N-nitro-N'-phthaloyl isourea, often suffer from yields consistently below 60 percent, rendering them economically unviable for competitive markets. These legacy processes frequently necessitate the use of expensive, moisture-sensitive reagents and generate copious amounts of toxic byproducts that require complex and costly disposal protocols. Furthermore, the O-methyl-N-nitroisourea method, while more common, involves an excessively long synthetic sequence and consumes large quantities of nitrogen-containing reagents, leading to severe environmental pollution issues. The cumulative effect of these drawbacks is a supply chain that is fragile, costly, and increasingly incompatible with stringent global environmental regulations.
The Novel Approach
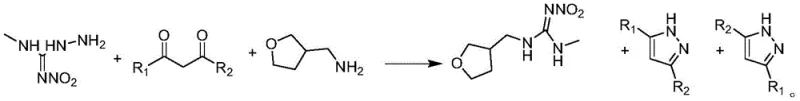
In stark contrast, the methodology disclosed in the patent data presents a paradigm shift by streamlining the synthesis into a highly efficient, convergent process. This new route bypasses the need for unstable intermediates by directly condensing a specifically prepared hydrazine derivative (Compound I) with versatile 1,3-dicarbonyl compounds (Compound II). The reaction is facilitated by a unique dual-catalyst system that simultaneously promotes condensation and removes water, driving the equilibrium towards the desired product with exceptional efficiency. A standout feature of this approach is the generation of pyrazole derivatives as co-products, which possess independent economic value and can be easily separated, thereby transforming a potential waste stream into a revenue source. This holistic improvement in atom economy and process simplicity marks a significant advancement in the manufacturing of high-purity agrochemical intermediates.
Mechanistic Insights into Acid-Water Absorption Catalyzed Condensation
The core innovation of this synthesis lies in the intricate interplay between the acid catalyst and the water-absorbing agent, which creates an optimal microenvironment for the formation of the nitroguanidine moiety. The reaction initiates with the activation of the carbonyl groups in the 1,3-dicarbonyl substrate by the acid catalyst, increasing their electrophilicity towards nucleophilic attack by the hydrazine derivative. Simultaneously, the water-absorbing catalyst, such as activated molecular sieves or anhydrous sodium sulfate, sequesters the water molecules produced during the condensation steps. This continuous removal of water prevents the hydrolysis of sensitive intermediates and shifts the chemical equilibrium decisively towards product formation according to Le Chatelier's principle. The result is a rapid and high-yielding transformation that proceeds smoothly at moderate temperatures, typically ranging from 50 degrees Celsius to the reflux temperature of the solvent.
Furthermore, the structural integrity and purity of the final Dinotefuran molecule are meticulously preserved through precise control of the crystallization dynamics. The patent data highlights that the cooling rate in the final isolation step is a critical parameter, with an optimal range of 1 degree Celsius per minute to 3 degrees Celsius per minute identified for maximizing purity. Rapid cooling can trap impurities within the crystal lattice, whereas overly slow cooling may promote the formation of unwanted polymorphs or degrade thermal stability. By maintaining this controlled thermal gradient, the process ensures that the target molecule precipitates in a highly ordered crystalline form, while soluble impurities and the valuable pyrazole byproducts remain in the mother liquor. This level of mechanistic control is essential for meeting the rigorous quality specifications demanded by the global agrochemical industry.
How to Synthesize Dinotefuran Efficiently
The practical implementation of this synthesis route requires careful attention to reagent stoichiometry and reaction conditions to replicate the high yields reported in the patent examples. The process begins with the preparation of the key hydrazine intermediate, Compound I, which is synthesized by reacting 1-methyl-3-nitroguanidine with hydrazine hydrate in an aqueous medium, followed by acid quenching and crystallization to ensure high purity before entering the main reaction vessel. Once Compound I is secured, it is combined with the chosen 1,3-dicarbonyl compound and the dual-catalyst system in a non-alcoholic solvent such as toluene or xylene. The detailed operational parameters, including specific molar ratios and temperature profiles, are critical for success and are outlined in the standardized guide below for technical teams to follow.
- Prepare Compound I by reacting 1-methyl-3-nitroguanidine with hydrazine hydrate in water, followed by acid quenching and crystallization.
- In a reactor, combine Compound I, a 1,3-dicarbonyl compound (Compound II), an acid catalyst (e.g., formic acid), and a water-absorbing catalyst (e.g., molecular sieves) in a non-alcoholic solvent like toluene.
- Heat the mixture to reflux, add tetrahydrofuran-3-methylamine, continue reaction, then cool slowly to crystallize the product and separate valuable pyrazole byproducts from the filtrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple yield improvements. The shift away from exotic, hazardous reagents like triflates towards commodity chemicals such as acetylacetone and malondialdehyde drastically stabilizes the raw material supply base. This transition mitigates the risk of supply disruptions caused by the limited availability of specialized precursors, ensuring a more consistent and reliable flow of materials for continuous manufacturing operations. Moreover, the simplification of the process flow reduces the number of unit operations required, which directly translates to lower capital expenditure on equipment and reduced operational complexity on the plant floor.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the elimination of expensive and difficult-to-handle reagents that characterized previous methods. By utilizing a dual-catalyst system that can be partially recovered and reused, specifically the solid molecular sieves which are easily filtered and regenerated, the recurring cost of consumables is significantly lowered. Additionally, the ability to isolate and sell the pyrazole byproducts creates a secondary revenue stream that effectively subsidizes the production cost of the primary Dinotefuran product, leading to substantial overall cost savings without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals for the 1,3-dicarbonyl component ensures that the supply chain is resilient against market volatility. Unlike proprietary intermediates that may be sourced from a single supplier, the key starting materials for this route are produced by multiple chemical manufacturers globally, providing procurement teams with greater negotiating power and flexibility. This diversification of the supply base reduces lead times and minimizes the risk of production stoppages due to raw material shortages, thereby enhancing the overall reliability of the supply chain for downstream formulators.
- Scalability and Environmental Compliance: From a scalability perspective, the one-pot nature of the reaction simplifies the scale-up process from pilot plant to commercial production, as there are fewer intermediate isolation and purification steps that typically introduce bottlenecks. The reduction in solvent usage and the avoidance of heavy metal catalysts or highly toxic reagents also streamline waste treatment processes, making it easier to comply with increasingly strict environmental regulations. This environmental compatibility not only reduces disposal costs but also future-proofs the manufacturing facility against regulatory changes, ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Dinotefuran synthesis method. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a reliable foundation for decision-making. Understanding these nuances is crucial for R&D teams evaluating the feasibility of technology transfer and for commercial teams assessing the market potential of the resulting product.
Q: What are the advantages of the new Dinotefuran synthesis route over traditional methods?
A: The new route utilizes a one-pot condensation of readily available 1,3-dicarbonyl compounds, avoiding the use of hazardous triflates or complex thiourea derivatives found in older methods. This significantly shortens the synthetic sequence, reduces pollution, and improves overall production efficiency while generating valuable pyrazole byproducts.
Q: How is the purity of Dinotefuran controlled in this process?
A: Purity is strictly controlled through a dual-catalyst system involving both acid and water-absorbing agents, which drives the equilibrium forward. Furthermore, a precise cooling rate (optimized between 1°C/min and 3°C/min) during crystallization ensures that impurities remain in the mother liquor while high-purity Dinotefuran precipitates.
Q: Can the catalysts and solvents be recovered in this manufacturing process?
A: Yes, the process is designed for sustainability. Solid catalysts like molecular sieves can be recovered from the filter cake, washed, dried, and reused. Additionally, the non-alcoholic solvents such as toluene can be distilled and recycled from the filtrate, contributing to significant cost reduction and waste minimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dinotefuran Supplier
As the agrochemical sector evolves towards more sustainable and efficient manufacturing paradigms, partnering with a technically proficient CDMO is essential for capitalizing on these innovations. NINGBO INNO PHARMCHEM stands at the forefront of this transition, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of this novel condensation chemistry, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that the successful commercialization of complex agrochemical intermediates requires not just chemical expertise, but a deep commitment to quality assurance and process safety.
We invite you to collaborate with us to unlock the full potential of this advanced synthesis route for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this method can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and growth in your agrochemical portfolio.
