Advanced Manufacturing of 3-Aminomethyl Tetrahydrofuran for Global Agrochemical Supply Chains
The global agrochemical sector is witnessing a sustained surge in demand for next-generation neonicotinoid insecticides, particularly Dinotefuran, following the expiration of key compound patents. As the market expands, the pressure on supply chains to deliver high-purity intermediates efficiently has never been greater. Patent CN108424406B introduces a transformative production process for 3-aminomethyl tetrahydrofuran, the critical building block for Dinotefuran, addressing long-standing inefficiencies in traditional synthesis. This innovative methodology leverages a concise three-step sequence starting from readily available 2-butene-1,4-diol, utilizing advanced catalytic systems to achieve superior atom economy. By shifting away from hazardous reagents and multi-step protocols, this technology offers a robust pathway for reliable agrochemical intermediate supplier networks to enhance their production capabilities. The strategic implementation of this process not only mitigates environmental impact but also stabilizes the supply of this essential chemical precursor against market volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminomethyl tetrahydrofuran has been plagued by significant operational hazards and economic inefficiencies inherent to legacy chemical pathways. Traditional routes often rely on the use of extremely toxic reagents such as sodium cyanide (NaCN) or explosive compounds like sodium azide (NaN3), which impose severe safety burdens and require specialized, costly waste treatment infrastructure. Furthermore, many existing methods involve excessive synthetic steps, sometimes exceeding five or six stages, which inevitably leads to cumulative yield losses and increased consumption of solvents and energy. The reliance on corrosive agents like phosphorus oxychloride (POCl3) in Vilsmeier formylation reactions further exacerbates equipment maintenance costs and poses risks to personnel safety. These factors collectively result in a fragmented supply chain where cost reduction in agrochemical intermediate manufacturing is stifled by the complexity and danger of the chemistry involved. Consequently, manufacturers face difficulties in scaling these processes without compromising on purity or incurring prohibitive operational expenses.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the novel process outlined in the patent utilizes a streamlined three-step strategy that fundamentally reimagines the synthetic landscape for this key intermediate. The route begins with a catalytic dehydration cyclization of 2-butene-1,4-diol, followed by a highly selective hydroformylation, and concludes with a straightforward reductive amination. This approach completely eliminates the need for toxic cyanides or explosive azides, replacing them with safer, more manageable catalytic systems that operate under mild conditions. The integration of a specific Rhodium catalyst system alongside a boron-based Lewis acid ensures high regioselectivity and conversion rates, drastically simplifying the purification requirements. By reducing the step count and removing hazardous reagents, the process inherently lowers the barrier for commercial scale-up of complex agrochemical intermediates. This technological leap provides a clear competitive advantage, enabling producers to offer high-purity products with a significantly reduced environmental footprint and enhanced operational safety profile.
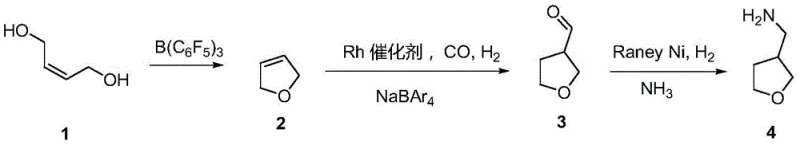
Mechanistic Insights into Catalytic Cyclization and Hydroformylation
The core of this technological breakthrough lies in the sophisticated application of Lewis acid catalysis and transition metal hydroformylation, which work in tandem to construct the tetrahydrofuran ring with precision. The initial step employs tris(pentafluorophenyl)borane, a potent Lewis acid, to facilitate the dehydration cyclization of 2-butene-1,4-diol into 2,5-dihydrofuran. This catalyst is chosen for its ability to activate the hydroxyl groups effectively at moderate temperatures, minimizing side reactions such as polymerization or over-dehydration that often plague acid-catalyzed cyclizations. Following this, the dihydrofuran intermediate undergoes hydroformylation using a Rhodium complex, specifically RhCl(CO)(DPPB), in the presence of a non-coordinating anion source like NaBAr4. This catalytic system is crucial for directing the formyl group to the 3-position of the ring with high regioselectivity, ensuring that the subsequent amination yields the desired aminomethyl structure rather than unwanted isomers. The precise tuning of the ligand environment around the Rhodium center allows the reaction to proceed efficiently at relatively low pressures and temperatures, preserving the integrity of the sensitive furan ring.
Impurity control is intrinsically built into the mechanism of this process through the selection of mild reaction conditions and highly specific catalysts. In traditional routes, harsh acidic or basic conditions often lead to ring-opening side reactions or the formation of polymeric byproducts that are difficult to separate from the final product. However, the use of the boron Lewis acid and the Rhodium hydroformylation system avoids these extreme conditions, thereby suppressing the formation of degradation products. Furthermore, the final reductive amination step utilizes Raney Nickel under controlled hydrogen pressure in an ammonia-methanol solution, which selectively reduces the aldehyde-oxime or imine intermediate without affecting the ether linkage of the tetrahydrofuran ring. This selectivity is paramount for achieving the stringent purity specifications required for pharmaceutical and agrochemical applications. The cumulative effect of these mechanistic advantages is a product profile with a clean impurity spectrum, reducing the need for extensive downstream purification and ensuring consistent quality across large production batches.
How to Synthesize 3-Aminomethyl Tetrahydrofuran Efficiently
The synthesis of this valuable intermediate is achieved through a logical progression of catalytic transformations that prioritize safety and yield. The process begins with the activation of the diol substrate, moves through a carbon-carbon bond-forming hydroformylation, and concludes with nitrogen incorporation. Each step is optimized to maximize throughput while minimizing waste generation, reflecting a modern approach to green chemistry in fine chemical manufacturing. The detailed standardized synthesis steps see the guide below for specific operational parameters and stoichiometry.
- Dehydration cyclization of 2-butene-1,4-diol using B(C6F5)3 catalyst to form 2,5-dihydrofuran.
- Hydroformylation of 2,5-dihydrofuran using Rh catalyst and NaBAr4 to produce 3-formyl tetrahydrofuran.
- Reductive amination of 3-formyl tetrahydrofuran using Raney Ni and ammonia to yield 3-aminomethyl tetrahydrofuran.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route represents a strategic opportunity to optimize costs and secure supply continuity for critical agrochemical ingredients. The elimination of hazardous reagents like sodium cyanide and sodium azide removes the need for expensive safety protocols, specialized storage facilities, and complex hazardous waste disposal procedures, leading to substantial cost savings in overall manufacturing operations. Additionally, the reduction in synthetic steps from five or six down to three directly correlates with lower utility consumption, reduced solvent usage, and decreased labor hours per kilogram of product. This efficiency gain translates into a more competitive pricing structure without sacrificing quality, allowing buyers to negotiate better terms with their suppliers. The robustness of the process also means that production schedules are less likely to be disrupted by safety incidents or regulatory compliance issues related to toxic chemical handling.
- Cost Reduction in Manufacturing: The streamlined three-step process significantly lowers the cost of goods sold by reducing raw material consumption and energy requirements associated with multiple isolation and purification stages. By avoiding expensive and toxic reagents such as Ru/C catalysts and sodium cyanide, the process eliminates the high costs associated with their procurement, safe handling, and neutralization. The high yields achieved in each step further contribute to cost efficiency by maximizing the output from every unit of starting material, reducing the effective cost per kilogram of the final intermediate. This economic advantage is compounded by the use of commercially available and inexpensive starting materials like 2-butene-1,4-diol, ensuring stable input costs even during market fluctuations.
- Enhanced Supply Chain Reliability: The reliance on common, non-restricted raw materials ensures that the supply chain is resilient against regulatory bans or shortages often associated with controlled substances like cyanides. The mild reaction conditions reduce the risk of unplanned shutdowns due to equipment corrosion or safety alarms, leading to more predictable production timelines and shorter lead times for high-purity agrochemical intermediates. Furthermore, the simplified process flow allows for easier scaling of production capacity to meet sudden spikes in demand, providing a buffer against market volatility. Suppliers adopting this technology can offer more reliable delivery schedules, as the process is less susceptible to the bottlenecks caused by complex waste treatment or hazardous material logistics.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, featuring reactions that can be easily transferred from laboratory to pilot and finally to full commercial scale without significant re-engineering. The reduction in three-waste generation aligns with increasingly stringent global environmental regulations, minimizing the risk of fines or production halts due to non-compliance. The absence of heavy metal contaminants from expensive catalysts like Ruthenium simplifies the purification process and ensures the final product meets strict residue limits. This environmental stewardship not only protects the manufacturer's license to operate but also enhances the brand value for downstream customers who are under pressure to reduce the carbon footprint of their own supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this intermediate, based on the specific advantages detailed in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this material into their existing formulations or synthesis pipelines. The answers provided reflect the consensus on the operational benefits and safety profiles associated with this advanced manufacturing route.
Q: What are the primary safety advantages of this new synthesis route?
A: The process eliminates the use of highly toxic sodium cyanide (NaCN) and explosive sodium azide (NaN3) found in conventional methods, significantly improving operational safety and reducing hazardous waste disposal costs.
Q: How does the catalyst system improve cost efficiency?
A: By utilizing a highly active Rhodium catalyst system combined with a specific boron Lewis acid for cyclization, the process achieves high yields in fewer steps, reducing raw material consumption and energy usage compared to five or six-step legacy routes.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the process features mild reaction conditions and generates less three-waste, making it highly suitable for commercial scale-up and continuous manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Aminomethyl Tetrahydrofuran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis plays in the competitiveness of the global agrochemical market. Our team of expert chemists has extensively evaluated emerging technologies like the one described in CN108424406B to ensure our production capabilities remain at the forefront of the industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major multinational corporations. Our commitment to quality is upheld by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards. By leveraging advanced catalytic methods, we are able to offer a product that not only meets but exceeds the performance expectations of downstream pesticide manufacturers.
We invite you to collaborate with us to explore how this optimized supply chain can benefit your specific production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements and logistical constraints. Please contact us directly to request specific COA data and route feasibility assessments for your upcoming projects. Together, we can build a more sustainable and efficient supply chain for the next generation of crop protection solutions.
