Revolutionizing L-Carnitine Production: A Safe, Asymmetric Catalytic Route for Industrial Scale-Up
Revolutionizing L-Carnitine Production: A Safe, Asymmetric Catalytic Route for Industrial Scale-Up
The global demand for L-carnitine, a vital quaternary ammonium compound essential for fatty acid metabolism and widely utilized in pharmaceutical, nutritional, and food industries, continues to surge. However, traditional manufacturing methods have long been plagued by inefficiencies, safety hazards, and environmental concerns. Patent CN110483316B introduces a groundbreaking asymmetric synthesis method that fundamentally redefines the production landscape for this critical nutritional ingredient. By leveraging an innovative in-situ generation of ketene from acetyl chloride, this technology bypasses the inherent dangers of handling unstable gaseous ketene while achieving exceptional enantioselectivity. For R&D directors and procurement managers seeking a reliable L-carnitine supplier, this patent represents a paradigm shift towards safer, more cost-effective, and scalable manufacturing protocols that align with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
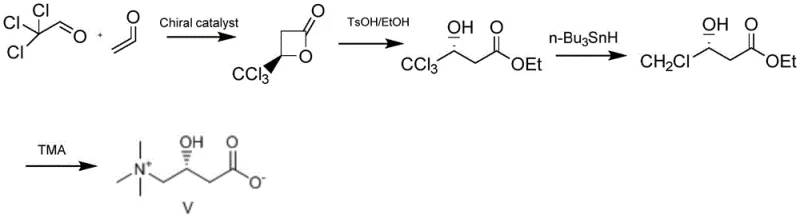
Historically, the industrial preparation of L-carnitine has relied heavily on the resolution of racemic mixtures or microbial fermentation, both of which suffer from significant drawbacks. Resolution methods are inherently inefficient, with a maximum theoretical yield of only 50%, necessitating the disposal or recycling of the unwanted D-enantiomer, which drives up costs and waste. Furthermore, earlier asymmetric synthetic routes, such as the method reported by Song involving trichloroacetaldehyde, required the use of toxic tin compounds for dechlorination and involved lengthy multi-step sequences that complicated purification. Another notable approach by Paul utilized direct ketene, but this introduced severe safety liabilities due to ketene's tendency to polymerize into diketene and its high toxicity, making industrial scale-up hazardous and economically unviable for many manufacturers.
The Novel Approach
The methodology disclosed in CN110483316B offers a robust solution by employing acetyl chloride as a stable precursor for the in-situ generation of ketene under mild basic conditions. This strategic modification eliminates the need to store or transport dangerous ketene gas, thereby significantly enhancing process safety and operational continuity. The generated ketene immediately undergoes an asymmetric intermolecular [2+2] cycloaddition with chloroacetaldehyde in the presence of a chiral catalyst and Lewis acid, directly forming a chiral lactone intermediate with high stereocontrol. This streamlined one-pot transformation not only reduces the number of unit operations but also minimizes solvent consumption and waste generation, offering a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
![Novel asymmetric synthesis route of L-carnitine via in-situ ketene generation and [2+2] cycloaddition](/insights/img/l-carnitine-asymmetric-synthesis-pharma-supplier-20260307082714-02.webp)
Mechanistic Insights into Asymmetric [2+2] Cycloaddition
The core of this technological breakthrough lies in the sophisticated dual-catalytic system that governs the stereoselective formation of the beta-lactone ring. The reaction utilizes a specific class of chiral organic small molecules, specifically compounds IIIb through IIIf, which act as organocatalysts to activate the electrophilic species. These catalysts work in synergy with a Lewis acid, typically lithium perchlorate, to coordinate the chloroacetaldehyde and facilitate the nucleophilic attack by the in-situ generated ketene. This cooperative catalysis creates a highly organized transition state that rigidly controls the facial selectivity of the cycloaddition, ensuring that the S-configuration is predominantly formed. The precision of this mechanism is evidenced by the exceptional enantiomeric excess (ee) values achieved, ranging from 94.5% to 98.5% depending on the specific catalyst variant employed.
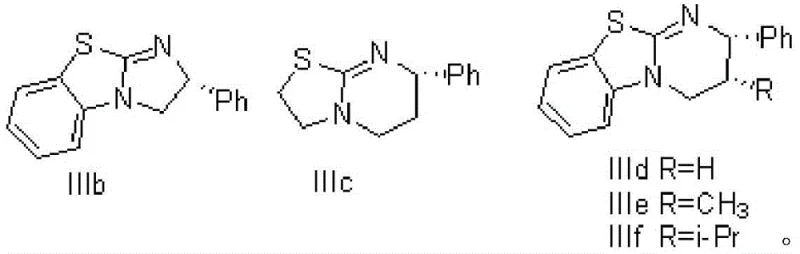
Furthermore, the mechanistic pathway is designed to suppress side reactions that typically plague ketene chemistry, such as dimerization or uncontrolled polymerization. By maintaining the reaction temperature between -70°C and 20°C and carefully controlling the addition rate of reagents, the process ensures that the concentration of free ketene remains low, favoring the desired cross-reaction with chloroacetaldehyde. This level of kinetic control is crucial for maintaining high purity profiles and minimizing the formation of difficult-to-remove impurities. For technical teams evaluating route feasibility, this mechanism demonstrates a high degree of robustness, allowing for consistent product quality even when scaling from laboratory benchtop to commercial production vessels.
How to Synthesize L-Carnitine Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing high-purity L-carnitine suitable for pharmaceutical and nutritional applications. The process begins with the low-temperature dehydrohalogenation of acetyl chloride to generate ketene, followed immediately by the asymmetric cycloaddition to form the chiral lactone. The final stage involves a ring-opening reaction with trimethylamine and subsequent purification via electrodialysis. This sequence is designed to be operationally simple, avoiding complex chromatographic separations and utilizing common industrial solvents. For detailed standard operating procedures and specific reaction parameters, please refer to the technical guide below.
- Generate ketene in-situ from acetyl chloride using an organic base at low temperature (-50 to -20°C) to avoid handling unstable gaseous ketene.
- Perform an asymmetric intermolecular [2+2] cycloaddition between the generated ketene and chloroacetaldehyde using a chiral catalyst and Lewis acid to form a chiral lactone intermediate with >95% ee.
- React the chiral lactone with trimethylamine and sodium hydroxide, followed by electrodialysis and recrystallization to isolate high-purity L-carnitine.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented process addresses several critical pain points associated with traditional L-carnitine manufacturing, offering tangible benefits in terms of reliability and total cost of ownership. By shifting from unstable gaseous reagents to stable liquid precursors like acetyl chloride, the method drastically simplifies raw material logistics and storage requirements. This stability translates directly into enhanced supply chain reliability, as manufacturers are no longer vulnerable to the degradation losses or safety shutdowns often associated with handling hazardous gases. Additionally, the elimination of toxic tin reagents and the reduction in synthetic steps contribute to a significantly greener process profile, which is increasingly important for meeting stringent environmental regulations in global markets.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven primarily by the high atom economy of the [2+2] cycloaddition and the avoidance of expensive resolution agents. Since the asymmetric catalysis yields the desired enantiomer directly with high selectivity, there is no need to purchase, separate, and dispose of the unwanted D-isomer, effectively doubling the theoretical yield compared to resolution methods. Furthermore, the use of commodity chemicals like acetyl chloride and chloroacetaldehyde ensures that raw material costs remain low and predictable, shielding buyers from the volatility often seen with specialized chiral building blocks.
- Enhanced Supply Chain Reliability: The robustness of the in-situ ketene generation technique ensures consistent batch-to-batch quality, which is paramount for maintaining uninterrupted production schedules. Because the reaction conditions are mild and the catalysts are highly effective, the process is less susceptible to variations in raw material quality or minor fluctuations in operating parameters. This reliability allows supply chain managers to forecast lead times with greater accuracy and reduces the risk of production delays caused by safety incidents or complex purification bottlenecks.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to full commercial production is facilitated by the use of standard reactor equipment and the absence of exotic or highly hazardous reagents. The workup procedure, which utilizes electrodialysis for desalting rather than ion-exchange resins or extensive solvent extractions, significantly reduces the volume of wastewater and solid waste generated. This streamlined downstream processing not only lowers waste disposal costs but also simplifies the regulatory approval process for new manufacturing sites, accelerating time-to-market for new capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a transparent view of the process capabilities. Understanding these details is essential for stakeholders evaluating the integration of this method into their existing supply chains.
Q: Why is in-situ ketene generation preferred over using pre-formed ketene?
A: Pre-formed ketene is highly unstable, prone to polymerization into diketene, and poses significant safety risks during storage and transport. The patented method generates ketene in-situ from stable acetyl chloride, drastically improving operational safety and reducing raw material loss.
Q: How does this method improve enantioselectivity compared to traditional resolution?
A: Traditional resolution methods are limited to a maximum theoretical yield of 50% and require expensive resolving agents. This asymmetric catalytic route achieves enantiomeric excess (ee) values exceeding 98.5% directly during the bond-forming step, eliminating the need for wasteful separation processes.
Q: What are the purification advantages of this synthetic route?
A: The process utilizes electrodialysis for desalting instead of complex chromatographic separations. Combined with a straightforward recrystallization step using ethanol and acetone, this ensures high purity (99.5%+) while minimizing solvent waste and processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnitine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the asymmetric synthesis technology described in CN110483316B and are fully equipped to leverage these innovations for our global partners. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are realized in practical, large-volume manufacturing. Our facilities are supported by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of L-carnitine meets the highest standards required for pharmaceutical and high-end nutritional applications.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through this advanced synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
