Scalable Biocatalytic Production of High-Optical-Purity L-Carnitine Intermediates
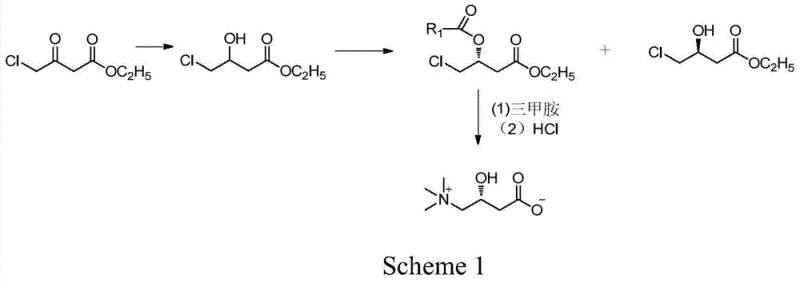
The pharmaceutical and nutritional supplement industries continuously demand higher purity standards for chiral compounds, particularly for metabolic regulators like L-carnitine. A significant technological advancement in this domain is detailed in patent CN103497978A, which outlines a robust preparation method for high-optical-purity L-carnitine. This innovative synthetic route departs from traditional hazardous chemical methodologies by integrating a mild borohydride reduction with a highly selective enzymatic resolution step. The process begins with the readily available raw material, ethyl 4-chloroacetoacetate, which undergoes a controlled reduction to form ethyl 4-chloro-3-hydroxybutyrate. Subsequently, the core innovation lies in the use of immobilized lipase to perform an asymmetric esterification, effectively separating the desired (R)-enantiomer from the (S)-isomer with exceptional precision. This strategic combination of chemocatalysis and biocatalysis not only ensures product quality but also aligns with modern green chemistry principles by minimizing toxic waste. The final stages involve condensation with trimethylamine and hydrolysis to yield the target molecule, establishing a comprehensive pathway that addresses both purity and scalability concerns for industrial manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of L-carnitine has been plagued by significant safety and efficiency challenges that hinder optimal production workflows. Traditional chemical routes frequently rely on the use of epoxy chloropropane as a starting material, a process that necessitates the handling of sodium cyanide, a severely poisonous chemical posing extreme risks to worker safety and environmental compliance. Furthermore, these legacy methods often result in racemic mixtures requiring complex and costly chiral separation steps to isolate the biologically active L-isomer. Alternative approaches utilizing natural chiral compounds like tartaric acid derivatives involve multi-step syntheses with complicated operations and inherently lower yields, making them economically unviable for large-scale manufacturing. Even more recent attempts using transition metal catalysts, such as ruthenium complexes for asymmetric hydrogenation, suffer from limitations including the high cost of noble metals, the complexity of ligand preparation, and the difficulty in completely removing trace metal residues from the final pharmaceutical product. These conventional pathways often struggle to consistently achieve the stringent optical purity levels required for high-end medical applications, typically capping out around 96% ee, which is insufficient for premium market segments.
The Novel Approach
In stark contrast to these legacy issues, the novel approach described in the patent data introduces a streamlined and safer methodology that leverages the specificity of biological enzymes. By employing immobilized lipase, specifically Novozym435, the process achieves a kinetic resolution that selectively esterifies one enantiomer while leaving the other untouched, thereby driving the optical purity well above 99% ee. This biocatalytic step operates under mild reaction conditions, typically between 0°C and 40°C, which drastically reduces energy consumption compared to high-temperature chemical catalysis. The elimination of toxic cyanide reagents and expensive ruthenium catalysts fundamentally alters the cost structure and safety profile of the manufacturing process. Moreover, the strategy turns a potential waste stream into a value-added opportunity; the (S)-enantiomer by-product generated during resolution is not discarded but can be recovered and utilized as a key intermediate for synthesizing Statin class drugs. This holistic view of the synthesis tree transforms the production of L-carnitine from a linear, waste-generating process into a circular, economically efficient operation that maximizes atom economy and minimizes environmental impact.
Mechanistic Insights into Enzymatic Asymmetric Esterification
The cornerstone of this high-purity synthesis is the mechanistic precision offered by the immobilized lipase-catalyzed asymmetric esterification. In this critical step, the racemic ethyl 4-chloro-3-hydroxybutyrate serves as the substrate for the enzyme Candida antarctica lipase B, which is adsorbed onto a macroporous resin to enhance its stability and reusability in organic solvents. The enzyme exhibits profound stereoselectivity, preferentially recognizing and reacting with the hydroxyl group of the (R)-enantiomer in the presence of an acyl donor such as vinyl acetate or methoxy menthyl acetate. This selective acylation converts the (R)-alcohol into an ester while leaving the (S)-alcohol unchanged, creating a distinct chemical difference that allows for easy separation via standard techniques like column chromatography or distillation. The immobilization of the enzyme ensures that it maintains its structural integrity and catalytic activity over multiple cycles, providing a consistent reaction rate and selectivity that homogeneous catalysts often fail to match. The reaction kinetics are carefully tuned by adjusting parameters such as solvent polarity, using non-polar solvents like n-hexane or methyl tert-butyl ether to favor the forward esterification reaction while preventing enzyme deactivation.
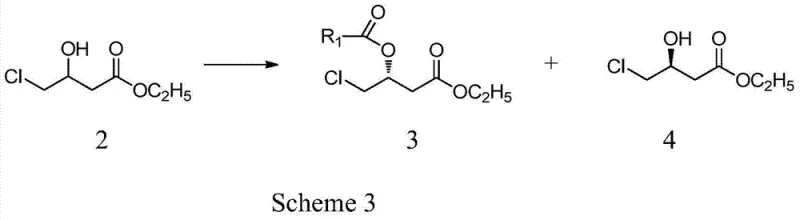
Following the resolution, the control of impurities is inherently managed by the specificity of the biocatalyst, which rejects non-target stereoisomers and side-reactants. Unlike chemical catalysts that might promote non-selective side reactions leading to complex impurity profiles, the lipase operates with a lock-and-key mechanism that strictly adheres to the stereochemical requirements of the active site. This results in a crude product with a significantly cleaner impurity spectrum, reducing the burden on downstream purification processes. The subsequent conversion of the resolved (R)-ester to L-carnitine involves a nucleophilic substitution with trimethylamine, followed by hydrolysis. Because the starting material for this final sequence is already of high optical purity (>99% ee), the final product inherits this quality without the need for further chiral resolution. The hydrolysis step is conducted under controlled acidic conditions to cleave the ester bond without racemizing the chiral center, ensuring that the final L-carnitine retains its biological activity. This mechanistic robustness provides R&D directors with confidence that the process is reproducible and capable of meeting rigorous pharmacopeial standards for chiral drugs.
How to Synthesize L-Carnitine Efficiently
The synthesis of L-carnitine via this patented route is designed for operational simplicity and high throughput, making it an ideal candidate for technology transfer and scale-up. The process initiates with a straightforward borohydride reduction where ethyl 4-chloroacetoacetate is treated with a reducing agent like potassium borohydride in a solvent system such as tetrahydrofuran or ethanol. This step is exothermic and requires careful temperature control, typically maintained between 0°C and 70°C, to ensure complete conversion to the hydroxy-intermediate without over-reduction or decomposition. Once the reduction is complete, the reaction mixture is quenched and worked up to isolate the racemic ethyl 4-chloro-3-hydroxybutyrate, which serves as the feedstock for the critical resolution step. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined below to guide process engineers in replicating this high-efficiency protocol.
- Perform borohydride reduction on ethyl 4-chloroacetoacetate using sodium or potassium borohydride in THF or alcohol solvents to obtain ethyl 4-chloro-3-hydroxybutyrate.
- Conduct asymmetric esterification on the hydroxybutyrate intermediate using immobilized lipase (Novozym435) and an acyl donor to separate (R) and (S) enantiomers.
- React the isolated (R)-ester with trimethylamine followed by acid hydrolysis and purification to yield high-purity L-carnitine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biocatalytic synthesis route presents a compelling value proposition centered on cost stability and supply security. The shift away from volatile and regulated chemicals like sodium cyanide eliminates the need for specialized hazardous material handling permits and expensive waste disposal protocols, directly translating to lower operational overheads. Furthermore, the reliance on immobilized enzymes, which can be recovered and reused multiple times, decouples production costs from the fluctuating prices of precious metals like ruthenium or rhodium often found in alternative catalytic systems. This stability in raw material sourcing ensures that long-term supply contracts can be honored without the risk of sudden price spikes due to geopolitical tensions affecting metal markets. The ability to utilize the (S)-enantiomer by-product for other high-value pharmaceutical applications, such as Statin synthesis, creates an additional revenue stream that offsets the cost of the primary L-carnitine production, effectively subsidizing the manufacturing expense.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and toxic reagents significantly lowers the direct material costs associated with each batch. By avoiding the complex ligand synthesis required for ruthenium-based asymmetric hydrogenation, the process simplifies the supply chain and reduces the number of unit operations, leading to substantial savings in labor and utility consumption. The mild reaction conditions also reduce energy demands for heating and cooling, contributing to a leaner manufacturing budget. Additionally, the high yield and selectivity of the enzymatic step minimize the loss of valuable starting materials, ensuring that a greater proportion of input mass is converted into saleable product.
- Enhanced Supply Chain Reliability: The raw materials for this process, such as ethyl 4-chloroacetoacetate and common borohydrides, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source bottlenecks. The robustness of the immobilized enzyme against varying reaction conditions means that production schedules are less likely to be disrupted by minor process deviations, ensuring consistent on-time delivery. The modular nature of the synthesis allows for flexible production scaling, enabling manufacturers to respond quickly to surges in market demand for L-carnitine supplements or pharmaceutical formulations without requiring massive capital investment in new infrastructure.
- Scalability and Environmental Compliance: The process is inherently scalable due to the use of heterogeneous catalysis, which facilitates easy separation of the catalyst from the reaction mixture, a critical factor for large-scale industrial reactors. The reduction in toxic waste generation aligns with increasingly stringent environmental regulations, minimizing the risk of regulatory fines and shutdowns. The aqueous workup and use of recyclable organic solvents further enhance the sustainability profile of the manufacturing site, making it easier to obtain and maintain necessary environmental permits. This green chemistry approach not only protects the environment but also enhances the brand reputation of the supplier among eco-conscious pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity L-carnitine synthesis method. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and technical feasibility for potential partners. Understanding these details is crucial for evaluating the fit of this technology within existing manufacturing portfolios and supply chain strategies.
Q: How does this biocatalytic method improve optical purity compared to traditional chemical synthesis?
A: Traditional methods often rely on toxic cyanide or expensive ruthenium catalysts which may yield lower enantiomeric excess (ee). This patented biocatalytic route utilizes immobilized lipase for kinetic resolution, achieving optical purity exceeding 99% ee, which is critical for pharmaceutical grade L-carnitine.
Q: What are the cost advantages of using immobilized lipase over transition metal catalysts?
A: Immobilized lipases like Novozym435 are reusable and operate under mild conditions, eliminating the need for expensive noble metals like ruthenium. This significantly reduces raw material costs and simplifies the removal of heavy metal impurities, lowering overall production expenses.
Q: Can the by-products from this L-carnitine synthesis be utilized?
A: Yes, the process generates (S)-4-chloro-3-hydroxybutyrate as a by-product during the resolution step. This chiral intermediate is valuable and can be repurposed for the synthesis of Statin class drugs, enhancing the overall economic efficiency of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Carnitine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic routes to meet the evolving demands of the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering high-purity L-carnitine and its intermediates with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. Our expertise in biocatalysis and chiral resolution allows us to optimize this patented process for maximum yield and minimal environmental impact, providing our clients with a sustainable and cost-effective supply solution.
We invite you to collaborate with us to leverage this innovative technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this route can improve your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data and proven performance. Let us be your partner in navigating the complexities of chiral intermediate manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
