Advanced Nitronium Salt-Promoted Thioamide Synthesis for Scalable Pharmaceutical Intermediate Production
The landscape of organic synthesis is constantly evolving, driven by the need for more efficient and environmentally benign pathways to critical building blocks. A significant breakthrough in this domain is documented in Chinese Patent CN114805152B, which discloses a novel synthetic method for thioamides utilizing nitronium salts. Thioamides are pivotal structural motifs in medicinal chemistry, serving as amide isosteres that can profoundly influence the biological activity, stability, and metabolic profile of drug candidates. This patent introduces a robust protocol that leverages nitronium salts, such as NO2BF4, to promote the reaction between bromides and nitriles in the presence of a sulfur source. This approach represents a paradigm shift from traditional methods, offering a versatile platform for generating high-purity pharmaceutical intermediates. The general reaction scope is illustrated below, highlighting the broad applicability across various substituted benzyl, alkyl, and cycloalkyl groups.
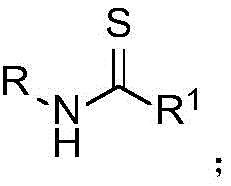
For R&D directors and process chemists, the ability to access diverse thioamide structures under relatively mild conditions is a substantial advantage. The method described in CN114805152B eliminates the need for harsh reagents often associated with sulfur incorporation, thereby simplifying the purification process and improving the overall impurity profile of the final product. By enabling the direct thioamidation of brominated derivatives, including those derived from complex drug molecules, this technology opens new avenues for the rapid synthesis of analog libraries and the optimization of lead compounds. As a reliable pharmaceutical intermediate supplier, understanding and adopting such innovative synthetic strategies is crucial for maintaining a competitive edge in the development of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioamides has been plagued by significant technical and environmental challenges that hinder large-scale production. Traditional methods often rely on the use of Lawesson's reagent or phosphorus pentasulfide (P2S5) to convert amides into thioamides. While effective on a small laboratory scale, these reagents generate substantial amounts of organic phosphorus waste, creating severe disposal issues and complicating the purification process due to the difficulty in separating phosphorus-containing byproducts. Furthermore, alternative routes such as the Willgerodt-Kindler reaction typically require high-temperature conditions which can lead to thermal decomposition of sensitive substrates and result in lower overall yields. Other approaches, like the Beckmann rearrangement of oximes, involve cumbersome multi-step sequences requiring the pre-preparation of oximes, which adds time and cost to the manufacturing process. Additionally, oxidative coupling reactions of amines are often limited to conjugated systems, restricting the structural diversity accessible to chemists. These limitations collectively underscore the urgent need for a more sustainable and efficient synthetic methodology.
The Novel Approach
In stark contrast to these legacy methods, the nitronium salt-promoted strategy outlined in patent CN114805152B offers a streamlined and highly adaptable solution. This novel approach utilizes readily available bulk chemicals such as nitriles and organic bromides, reacting them under the influence of nitronium salts like NO2BF4, NOBF4, or NOPF6. The reaction conditions are remarkably mild, typically proceeding at temperatures ranging from -35°C to 90°C, which preserves the integrity of sensitive functional groups often found in complex pharmaceutical intermediates. The versatility of this method is exemplified by its successful application in the synthesis of thioamide derivatives of complex drug molecules, such as ezetimibe. As shown in the reaction scheme below, the method tolerates intricate molecular architectures, enabling the direct installation of the thioamide functionality without compromising the rest of the molecule.
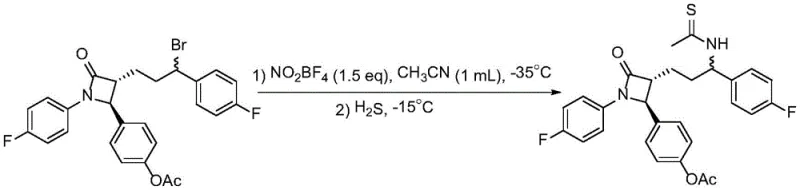
This capability is particularly valuable for cost reduction in API manufacturing, as it reduces the number of synthetic steps and avoids the use of expensive or hazardous reagents. The process is operationally simple, involving the sequential addition of reagents under an inert atmosphere, followed by a straightforward workup procedure that includes filtration and chromatography. The broad substrate scope, encompassing various substituted benzyl bromides, alkyl bromides, and cyclic bromides, ensures that this technology can be applied to a wide array of chemical targets. By overcoming the selectivity and yield issues associated with older methods, this novel approach provides a robust foundation for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nitronium Salt-Promoted Ritter-Type Reaction
The core of this innovative synthesis lies in the unique role of the nitronium salt as a potent promoter for a Ritter-type-like reaction mechanism. In this process, the nitronium cation acts as a strong electrophile that facilitates the activation of the nitrile group or the generation of a reactive carbocation species from the organic bromide. When the bromide substrate is introduced to the solution containing the nitronium salt and nitrile, an intermediate nitrilium ion is likely formed. This highly reactive species is then susceptible to nucleophilic attack by the sulfur source. The use of nitronium salts such as NO2BF4 allows for the generation of these reactive intermediates under much milder conditions than traditional Lewis acids, which often require stoichiometric amounts and generate significant waste. The reaction proceeds through a coordinated sequence where the initial formation of the carbon-nitrogen bond is followed by the incorporation of sulfur, ultimately yielding the thioamide structure after hydrolysis or workup.
From an impurity control perspective, this mechanism offers distinct advantages. The mild reaction temperatures, often maintained between -35°C and 45°C during the sulfurization step, minimize side reactions such as polymerization or decomposition that are common in high-temperature processes. The choice of sulfurizing reagent also plays a critical role in determining the purity of the final product. The patent details the successful use of various sulfur sources including hydrogen sulfide gas (H2S), sodium sulfide (Na2S), sodium hydrosulfide (NaHS), thiourea, and thioacetamide. Each of these reagents interacts with the reaction intermediate differently, allowing chemists to tune the process for optimal yield and purity. For instance, the use of gaseous H2S requires careful handling but can lead to clean conversion, while solid sulfur sources like Na2S offer ease of handling. The ability to remove solid impurities, such as elemental sulfur, via simple filtration through diatomaceous earth further streamlines the purification workflow, ensuring that the final high-purity pharmaceutical intermediates meet stringent quality specifications required for downstream applications.
How to Synthesize Thioamide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety. The general procedure involves dissolving the nitronium salt in a dry nitrile solvent, such as acetonitrile or nitromethane, under an inert argon atmosphere to prevent moisture interference. The organic bromide is then added dropwise at a controlled temperature, which varies depending on the reactivity of the substrate, ranging from room temperature to 90°C. After the initial reaction period, typically around 30 minutes, the mixture is cooled, and the sulfur source is introduced. The reaction is allowed to stir for a short duration, often 20 minutes, before quenching. The detailed standardized synthesis steps, including specific molar ratios and workup procedures for various substrates, are provided in the guide below.
- Dissolve a nitronium salt (such as NO2BF4) in a nitrile compound or a suitable solvent like acetonitrile under inert atmosphere.
- Add the organic bromide substrate to the reaction mixture at temperatures ranging from -35°C to 90°C and allow the reaction to proceed.
- Cool the mixture to between -35°C and 45°C, introduce a sulfurizing reagent such as H2S or Na2S, and stir to complete the thioamidation.
- Filter off solid impurities, remove solvents under reduced pressure, and purify the crude product via column chromatography to obtain the final thioamide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this nitronium salt-promoted synthesis offers tangible benefits that extend beyond mere chemical efficiency. The primary advantage lies in the significant simplification of the supply chain for raw materials. Unlike traditional methods that rely on specialized and often expensive reagents like Lawesson's reagent, this process utilizes commodity chemicals such as simple nitriles and organic bromides which are readily available from multiple global suppliers. This diversification of the supply base reduces the risk of shortages and price volatility, ensuring a more stable and reliable supply of critical intermediates. Furthermore, the elimination of phosphorus-based reagents removes the need for complex waste treatment protocols associated with organic phosphorus pollution, leading to substantial cost savings in environmental compliance and waste disposal.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the reduction in both material and operational costs. By avoiding the use of expensive transition metal catalysts or specialized sulfurizing agents that require rigorous removal steps, the overall cost of goods sold (COGS) is significantly lowered. The mild reaction conditions also translate to lower energy consumption, as there is no need for prolonged heating at high temperatures or cryogenic cooling below -35°C for extended periods. Additionally, the simplified workup procedure, which often involves basic filtration and distillation rather than complex extraction or recrystallization sequences, reduces labor hours and solvent usage. These factors combine to create a more cost-effective manufacturing process that enhances the margin potential for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route contributes directly to supply chain resilience. The tolerance of the reaction to a wide variety of functional groups means that the same core process can be applied to synthesize a diverse range of intermediates without needing to requalify entirely new manufacturing lines. This flexibility allows for faster response times to market demands and reduces the lead time for high-purity pharmaceutical intermediates. Moreover, the use of stable nitronium salts and common solvents ensures that the process is less susceptible to disruptions caused by the scarcity of exotic reagents. This reliability is crucial for maintaining continuous production schedules and meeting the strict delivery timelines expected by multinational pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram-scale laboratory experiments to multi-ton commercial production is facilitated by the inherent safety and simplicity of the reaction conditions. The absence of highly pyrophoric reagents or extreme pressure requirements makes the technology well-suited for standard stainless steel reactors found in most fine chemical manufacturing facilities. From an environmental standpoint, the method aligns with green chemistry principles by minimizing waste generation and avoiding toxic heavy metals. The ability to efficiently remove solid byproducts like elemental sulfur ensures that the effluent load is manageable, simplifying the permitting process for new manufacturing capacities. This combination of scalability and environmental stewardship positions the technology as a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
To address common technical inquiries regarding the implementation and scope of this technology, we have compiled a set of frequently asked questions based on the detailed experimental data provided in the patent documentation. These answers cover critical aspects such as reagent compatibility, reaction conditions, and substrate scope, providing clarity for technical teams evaluating this method for their specific projects. Understanding these nuances is essential for successful technology transfer and process optimization.
Q: What types of sulfurizing reagents are compatible with this nitronium salt method?
A: The patent specifies a wide range of sulfur sources including hydrogen sulfide (H2S), sodium sulfide (Na2S), sodium hydrosulfide (NaHS), thiourea, and thioacetamide, allowing flexibility based on safety and availability requirements.
Q: What is the operational temperature range for this thioamide synthesis?
A: The reaction demonstrates remarkable flexibility, operating effectively between -35°C and 90°C depending on the specific bromide substrate, with the sulfurization step typically occurring between -35°C and 45°C.
Q: Can this method be applied to complex drug molecules?
A: Yes, the methodology has been successfully demonstrated on complex brominated derivatives of active pharmaceutical ingredients such as ezetimibe and salimibe, proving its utility for late-stage functionalization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioamide Supplier
The technological advancements described in patent CN114805152B represent a significant leap forward in the synthesis of thioamide intermediates, offering a pathway to higher purity and greater efficiency. At NINGBO INNO PHARMCHEM, we are committed to leveraging such cutting-edge innovations to serve our global clientele. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from development to full-scale manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of thioamide intermediate delivered meets the highest industry standards for pharmaceutical applications.
We invite you to collaborate with us to explore the full potential of this nitronium salt-promoted synthesis for your specific drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project's unique requirements, demonstrating how this method can optimize your budget without compromising quality. Please contact us to request specific COA data for our existing thioamide portfolio or to discuss route feasibility assessments for your custom synthesis needs. Together, we can accelerate the delivery of life-saving medicines to the market through superior chemical innovation and reliable supply chain execution.
