Advanced Chemical Synthesis of Niraparib Chiral Intermediates for Commercial Scale-Up
Advanced Chemical Synthesis of Niraparib Chiral Intermediates for Commercial Scale-Up
The pharmaceutical landscape for Poly (ADP-ribose) polymerase (PARP) inhibitors has been revolutionized by the introduction of Niraparib, a potent agent for treating recurrent ovarian cancer. As demand for this critical oncology therapy surges globally, the efficiency of its supply chain becomes paramount. Patent CN107311911B discloses a groundbreaking preparation method for the chiral intermediate of Niraparib, addressing critical bottlenecks in traditional manufacturing. This novel approach leverages a sophisticated organic synthesis strategy starting from 4-bromophenylacetic acid and chiral substituted oxazolones, bypassing the limitations of earlier biological or metal-heavy routes. By establishing a robust, non-enzymatic pathway, this technology offers a compelling value proposition for reliable pharmaceutical intermediate supplier partnerships seeking to optimize their API sourcing strategies.
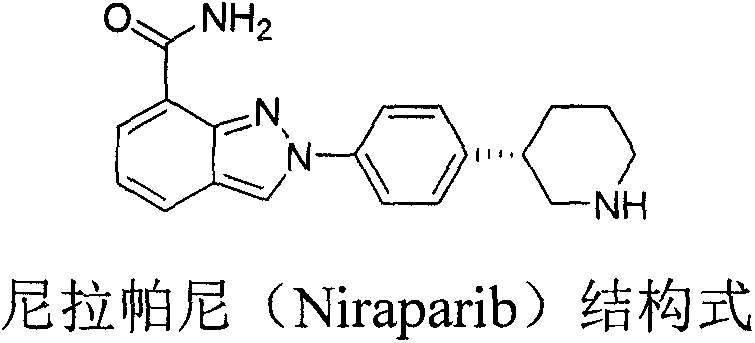
The strategic importance of this patent lies in its ability to deliver high-purity intermediates through a chemically defined process that is inherently more stable and predictable than biocatalytic alternatives. For R&D directors and procurement leaders, understanding the mechanistic advantages of this route is essential for long-term supply security. The method ensures consistent quality while drastically simplifying the purification workflow, which is a key driver for cost reduction in API manufacturing. As we delve into the technical specifics, it becomes clear that this synthesis represents a significant leap forward in the industrial production of complex chiral amines required for next-generation cancer therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the key piperidine intermediate for Niraparib has relied on pathways that introduce substantial operational friction and cost volatility. One prominent prior art route, described in WO2009087381, utilizes a Suzuki coupling reaction followed by hydrogenation with Platinum(IV) oxide (PtO2). While chemically feasible, this approach necessitates the use of expensive precious metal catalysts and, critically, relies on chiral column chromatography for enantiomeric separation. Chiral separation on an industrial scale is notoriously cumbersome, low-yielding, and creates significant solvent waste, making it economically unsustainable for high-volume commercial production. Furthermore, the reliance on transition metals introduces rigorous impurity control requirements to meet stringent pharmaceutical standards.
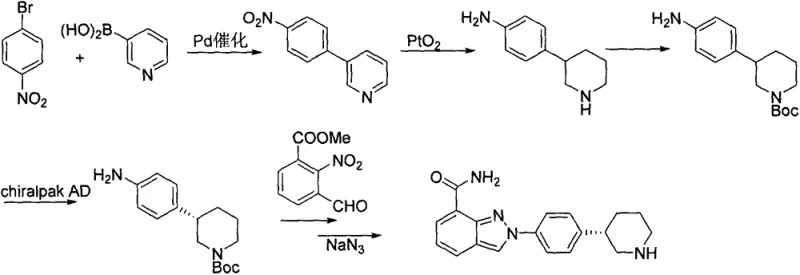
Another existing methodology, outlined in WO2014088983, attempts to solve the chirality issue through enzymatic catalysis. Although enzymes offer high selectivity, they come with their own set of supply chain vulnerabilities. Enzymes are often proprietary, expensive to purchase, and sensitive to reaction conditions such as temperature and pH, leading to batch-to-batch variability. The operational complexity of managing biocatalytic processes, including enzyme recovery and stability issues, results in higher production costs and extended lead times. For a supply chain head, these factors represent significant risks to continuity, as the availability of specific high-grade enzymes can be a single point of failure in the manufacturing timeline.
The Novel Approach
In stark contrast, the method disclosed in CN107311911B employs a purely chemical asymmetric synthesis strategy that elegantly circumvents these hurdles. By utilizing (S)-4-substituted-2-oxazolidinones as chiral auxiliaries, the process installs the necessary stereocenter through a highly diastereoselective Michael addition. This chemical approach eliminates the need for both precious metal hydrogenation catalysts and expensive biocatalysts. The starting materials, particularly 4-bromophenylacetic acid, are commodity chemicals with stable global supply chains, ensuring raw material availability. The route is designed for scalability, utilizing standard organic transformations like amide condensation, hydrolysis, and reduction that are well-understood and easily controlled in multi-ton reactors.
The novelty of this approach also extends to the purification profile. Because the chirality is controlled by the stoichiometric chiral auxiliary rather than a dynamic kinetic resolution or chromatographic separation, the intermediate streams possess inherently high optical purity. This reduces the burden on downstream processing and minimizes the loss of valuable material during purification steps. For procurement managers, this translates to a more predictable cost structure and a significant reduction in the cost of goods sold (COGS). The robustness of the chemical conditions allows for wider operating windows, enhancing the overall reliability of the manufacturing process and reducing the risk of batch failures due to sensitive reaction parameters.
Mechanistic Insights into Oxazolone-Mediated Asymmetric Michael Addition
The core of this synthetic innovation lies in the utilization of Evans-type chiral auxiliaries to direct the stereochemical outcome of the carbon-carbon bond formation. The process begins with the condensation of 4-bromophenylacetic acid with a chiral oxazolidinone, activated by pivaloyl chloride in the presence of a tertiary amine base. This forms a chiral imide which serves as a rigid template for the subsequent alkylation. In the critical Michael addition step, the enolate of this imide reacts with acrylonitrile. The presence of a titanium catalyst, such as titanium tetrachloride or tetraisopropyl titanate, plays a pivotal role in organizing the transition state. The titanium center coordinates with the carbonyl oxygens of the oxazolidinone and the incoming acrylonitrile, locking the conformation and forcing the nucleophilic attack to occur from the less hindered face.
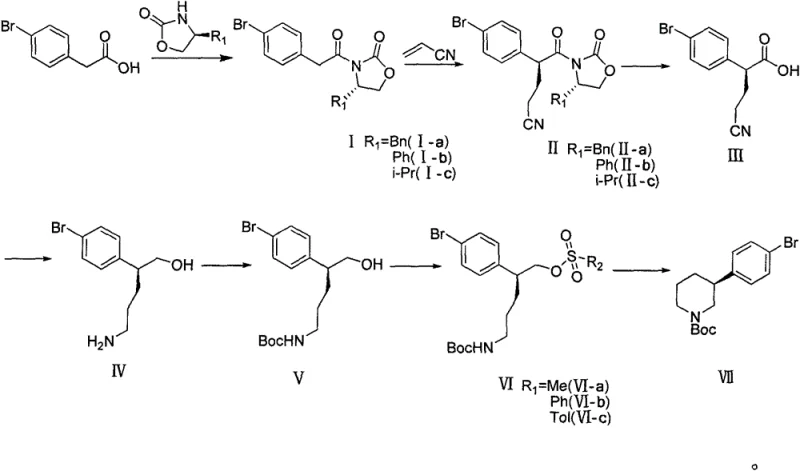
This coordination chemistry ensures that the newly formed stereocenter is established with exceptional fidelity, routinely achieving chiral purities exceeding 99% ee as demonstrated in the patent examples. Following the alkylation, the chiral auxiliary is cleaved via oxidative hydrolysis using hydrogen peroxide and lithium hydroxide. This step regenerates the chiral oxazolidinone for potential recycling and releases the free carboxylic acid intermediate. The subsequent reduction of the carboxylic acid to the primary alcohol, followed by protection of the amine and activation of the alcohol as a leaving group (mesylate or tosylate), sets the stage for the final intramolecular cyclization. This cascade of reactions is meticulously optimized to maintain the integrity of the chiral center throughout the sequence, preventing racemization which is a common pitfall in amine synthesis.
From an impurity control perspective, this mechanism offers distinct advantages. The byproducts generated are primarily the recycled chiral auxiliary and simple inorganic salts, which are easily removed via aqueous workups. Unlike transition metal-catalyzed cross-couplings, there is no risk of residual heavy metal contamination (e.g., Pd, Pt) which requires specialized scavenging resins and extensive testing to meet ICH Q3D guidelines. The deterministic nature of the stoichiometric chiral induction means that impurity profiles are consistent and predictable, facilitating smoother regulatory filings. For R&D teams, this predictability simplifies method validation and stability testing, accelerating the timeline from process development to commercial launch.
How to Synthesize Niraparib Intermediate Efficiently
The synthesis of the key chiral piperidine intermediate (Intermediate VII) is achieved through a streamlined seven-step sequence that prioritizes yield and operational simplicity. The process initiates with the activation of 4-bromophenylacetic acid and its coupling with a chiral oxazolidinone to form the foundational amide scaffold. This is followed by the stereoselective Michael addition with acrylonitrile, which constructs the carbon backbone with the required chirality. Subsequent hydrolysis removes the auxiliary, and reduction converts the acid functionality to an alcohol. After amine protection and activation of the alcohol, a final base-mediated cyclization closes the piperidine ring. Detailed standardized synthetic steps for each transformation, including specific reagent ratios and temperature controls, are provided in the technical guide below.
- Condense 4-bromophenylacetic acid with (S)-4-substituted-2-oxazolidinone using pivaloyl chloride and base to form the chiral amide intermediate.
- Perform a titanium-catalyzed Michael addition with acrylonitrile to establish the critical carbon-carbon bond with high stereoselectivity.
- Execute hydrolysis to remove the auxiliary, followed by reduction of the carboxylic acid and intramolecular cyclization to yield the final piperidine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain executives, the adoption of this synthetic route offers transformative benefits that extend beyond mere technical feasibility. The primary advantage is the drastic simplification of the supply chain architecture. By removing the dependency on specialized enzymes and precious metal catalysts, the manufacturing process becomes decoupled from volatile biological supply markets and fluctuating metal prices. The raw materials employed are bulk commodity chemicals produced by multiple vendors globally, ensuring a competitive bidding environment and safeguarding against supply disruptions. This diversification of the supply base is a critical risk mitigation strategy for maintaining continuous API production in a regulated environment.
- Cost Reduction in Manufacturing: The elimination of chiral chromatography and expensive biocatalysts directly impacts the bottom line. Chiral separation is often the most costly step in asymmetric synthesis due to low throughput and high solvent consumption. By achieving high enantiomeric excess through chemical induction, this route removes that bottleneck entirely. Furthermore, the avoidance of platinum and palladium catalysts reduces raw material costs and eliminates the need for expensive metal scavenging and recovery units. The overall yield profile of the route is high, minimizing waste disposal costs and maximizing the output per batch, leading to substantial cost savings in the final API price.
- Enhanced Supply Chain Reliability: The robustness of the chemical conditions ensures that the process is less susceptible to environmental variances compared to enzymatic routes. Enzymes often require strict cold chain logistics and have limited shelf lives, whereas the reagents used in this synthesis are stable solids or liquids with long storage durations. This stability simplifies inventory management and reduces the risk of raw material spoilage. Additionally, the scalability of the reactions means that production volumes can be ramped up quickly to meet market demand without the need for specialized bioreactor infrastructure, providing greater agility in responding to market fluctuations.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. It avoids the generation of heavy metal waste streams associated with Suzuki couplings and hydrogenations, simplifying wastewater treatment and regulatory compliance. The solvents used are standard organic solvents like dichloromethane, THF, and toluene, which have well-established recovery and recycling protocols in modern chemical plants. The high atom economy of the cyclization step and the recyclability of the chiral auxiliary contribute to a greener manufacturing footprint, aligning with the sustainability goals of major pharmaceutical companies and reducing the environmental compliance burden.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing manufacturing framework. Understanding these nuances is vital for making informed decisions about process adoption and supplier qualification.
Q: How does this chemical route compare to enzymatic methods for niraparib intermediates?
A: Unlike enzymatic routes which require expensive biocatalysts and complex operational controls, this chemical synthesis utilizes readily available reagents like 4-bromophenylacetic acid and standard titanium catalysts, significantly lowering production costs and simplifying scale-up.
Q: What level of chiral purity can be achieved with this oxazolone-based method?
A: The process demonstrates excellent stereocontrol, consistently achieving chiral purity greater than 99% ee in the Michael addition step, eliminating the need for costly chiral column separation required in earlier synthetic generations.
Q: Is this synthesis route suitable for large-scale industrial manufacturing?
A: Yes, the route is specifically designed for industrial viability, avoiding sensitive biological enzymes and precious metals like platinum, ensuring robust supply chain continuity and easier regulatory compliance for commercial API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Niraparib Intermediate Supplier
The technical potential of this oxazolone-mediated synthesis is immense, offering a clear path to a more resilient and cost-effective supply of Niraparib intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative route to life. Our facility is equipped with state-of-the-art reactors capable of handling the specific temperature and pressure requirements of the titanium-catalyzed steps, and our stringent purity specifications ensure that every batch meets the rigorous demands of global regulatory agencies. Our rigorous QC labs are fully validated to monitor chiral purity and trace impurities, guaranteeing the quality consistency essential for oncology drug manufacturing.
We invite you to discuss how this advanced synthesis can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from pilot batches and comprehensive route feasibility assessments to demonstrate how we can become your strategic partner in delivering high-quality Niraparib intermediates efficiently and reliably.
