Scalable Asymmetric Reduction of Ketones to High-Purity Chiral Secondary Alcohols
Scalable Asymmetric Reduction of Ketones to High-Purity Chiral Secondary Alcohols
The landscape of asymmetric synthesis is constantly evolving, driven by the dual demands of environmental sustainability and economic efficiency in the pharmaceutical sector. A pivotal advancement in this field is documented in Chinese Patent CN1376659A, which introduces a groundbreaking methodology for the preparation of chiral secondary alcohols with exceptional yield and stereoselectivity. This technology departs from conventional reliance on hazardous borane reagents, instead utilizing a novel composite reduction system comprising metal borohydrides, Lewis acids, and catalytic amounts of chiral ligands. For R&D directors and procurement specialists alike, this patent represents a significant opportunity to optimize the supply chain for critical API intermediates. By leveraging stable, low-cost starting materials, this process mitigates the risks associated with toxic gas handling while delivering optical purities that meet the rigorous standards of modern drug development. The implications for cost reduction in pharmaceutical intermediates manufacturing are profound, as the shift to solid-state reductants simplifies logistics and enhances operational safety profiles across the board.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric reduction of prochiral ketones has been dominated by methods such as the Itsuno-Corey reaction, which relies heavily on chiral oxazoline-catalyzed hydroboration. While chemically effective, these traditional pathways are plagued by significant logistical and safety drawbacks that hinder their scalability in a commercial setting. The primary reagent, borane (BH3), is highly toxic, pyrophoric, and typically supplied as a complex with tetrahydrofuran or dimethyl sulfide, requiring stringent storage conditions and specialized handling equipment. The volatility and reactivity of borane necessitate expensive containment systems and rigorous safety protocols, driving up the operational expenditure for manufacturing facilities. Furthermore, the cost of high-purity borane reagents is substantially higher than alternative hydride sources, creating a persistent burden on the cost of goods sold (COGS) for high-volume intermediates. From a supply chain perspective, the dependency on hazardous gas cylinders introduces fragility, as transportation regulations for dangerous goods can lead to delays and increased lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the methodology outlined in the patent data proposes a robust alternative that circumvents these inherent dangers by employing metal borohydrides such as sodium borohydride (NaBH4) or potassium borohydride (KBH4). These reagents are inexpensive, stable solids that are easy to store and handle under ambient conditions, effectively eliminating the need for pressurized gas infrastructure. The innovation lies in the activation of these mild reductants through the addition of Lewis acids like tin(II) chloride (SnCl2) or titanium tetrachloride (TiCl4), which generate a highly reactive species in situ capable of reducing ketones with high efficiency. When combined with naturally derived chiral ligands, this system achieves stereoselectivity comparable to or exceeding that of traditional borane methods, often reaching enantiomeric excess (ee) values of over 96%. This transition not only democratizes access to chiral synthesis for smaller laboratories but also provides a clear pathway for cost reduction in fine chemical manufacturing by replacing exotic reagents with commodity chemicals.
Mechanistic Insights into Metal Borohydride-Lewis Acid Catalyzed Reduction
The core of this technological breakthrough is the formation of a transient composite metal complex that serves as the active catalytic species. Upon mixing the metal borohydride M(BH4)n with the Lewis acid NXm in a solvent such as tetrahydrofuran (THF), a ligand exchange occurs that modifies the electronic properties of the boron center. This activation step is crucial, as unmodified borohydrides are typically too mild to reduce ketones asymmetrically with high selectivity. The Lewis acid coordinates with the hydride source, increasing its electrophilicity and facilitating the subsequent transfer of the hydride ion to the carbonyl carbon of the substrate. The presence of the chiral ligand creates a sterically defined environment around the metal center, ensuring that the hydride attack occurs preferentially from one face of the planar ketone. This precise spatial control is what drives the high enantioselectivity observed in the experimental data, allowing for the production of single-enantiomer alcohols essential for biological activity.
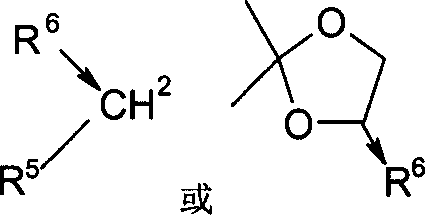
Crucial to the success of this mechanism is the selection of the chiral ligand, which acts as the stereochemical director. As illustrated in the structural data, the preferred ligands are often chiral beta-amino alcohols, which possess both nitrogen and oxygen donor atoms capable of coordinating to the metal center. This bidentate coordination stabilizes the transition state and rigidifies the catalyst structure, preventing non-selective background reactions. The patent data indicates that ligands with specific substituents, such as phenyl or methyl groups at the chiral center, yield optimal results, whereas simple chiral diols often fail to induce sufficient asymmetry. Furthermore, the choice of solvent plays a pivotal role in modulating the reaction kinetics and selectivity; THF is identified as the superior medium, likely due to its ability to solvate the metal cations effectively without interfering with the catalyst-substrate interaction. Impurity control is inherently built into this mechanism, as the high specificity of the chiral catalyst minimizes the formation of the undesired enantiomer, thereby simplifying downstream purification processes and reducing waste generation.
How to Synthesize Chiral Secondary Alcohols Efficiently
Implementing this synthesis route requires careful attention to the molar ratios of the components to ensure maximum conversion and selectivity. The general protocol involves pre-mixing the metal borohydride and Lewis acid in the chosen solvent at room temperature to allow for the formation of the active reducing species. Following this activation period, the chiral ligand is introduced, and the mixture is often heated to reflux to ensure complete complexation before the substrate is added. The prochiral ketone is then added slowly to the reaction mixture to manage the exotherm and maintain the integrity of the chiral environment. After the reaction is complete, typically indicated by TLC or HPLC analysis, the mixture is quenched with water, which decomposes any remaining borohydride and facilitates the separation of the organic product. The crude product can then be purified using standard techniques such as column chromatography or crystallization to achieve the desired pharmaceutical grade purity.
- Prepare the reducing agent by mixing metal borohydride (e.g., KBH4) and Lewis acid (e.g., SnCl2) in THF at room temperature.
- Add the chiral ligand (e.g., chiral beta-amino alcohol) and heat the mixture to reflux to activate the catalyst.
- Slowly add the prochiral ketone solution, maintain reflux, then quench with water and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal borohydride-based reduction system offers tangible strategic benefits that extend beyond mere chemical efficacy. The most immediate impact is seen in the realm of raw material sourcing and inventory management. By shifting from hazardous borane complexes to stable solid borohydrides, companies can drastically simplify their warehousing requirements, eliminating the need for specialized flammable gas cabinets and explosion-proof storage areas. This reduction in infrastructure overhead translates directly into lower operational costs and a reduced regulatory burden regarding hazardous material compliance. Additionally, the reagents utilized in this process, such as sodium borohydride and tin chloride, are commodity chemicals produced on a massive global scale, ensuring a reliable supply chain that is less susceptible to the market volatility often seen with specialty fine chemicals. This stability allows for more accurate long-term planning and budget forecasting, securing the continuity of production for critical drug intermediates.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the substitution of expensive reagents with cost-effective alternatives. Traditional asymmetric boronation requires stoichiometric or near-stoichiometric amounts of costly chiral auxiliaries and borane sources, whereas this new method operates with catalytic amounts of ligand and cheap inorganic salts. The elimination of borane gas cylinders removes a significant line item from the procurement budget, while the use of earth-abundant metals like tin or aluminum further depresses material costs. Moreover, the simplified workup procedure, which involves a straightforward aqueous quench rather than complex oxidative workups often required for boron species, reduces solvent consumption and waste disposal fees. These cumulative efficiencies result in substantial cost savings per kilogram of product, enhancing the overall margin profile for the manufactured intermediate without compromising on quality or purity standards.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the use of non-hazardous, shelf-stable reagents that do not require cold chain logistics or expedited shipping. Unlike borane-THF complexes which have limited shelf lives and degrade upon exposure to moisture, metal borohydrides can be stored for extended periods under ambient conditions without loss of potency. This durability reduces the risk of stockouts due to reagent degradation and allows manufacturers to maintain leaner inventory levels with confidence. Furthermore, the transport of solid powders is subject to fewer regulatory restrictions than compressed gases or pyrophoric liquids, streamlining the logistics network and reducing lead times for international shipments. This reliability ensures that production schedules remain uninterrupted, safeguarding against the delays that can ripple through the entire pharmaceutical development timeline and impact time-to-market for new therapies.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this process aligns perfectly with the principles of green chemistry and sustainable manufacturing. The avoidance of toxic borane gas minimizes the risk of accidental releases and exposure incidents, creating a safer working environment for plant operators. The waste stream generated is predominantly inorganic salts and aqueous waste, which are easier and cheaper to treat compared to the complex organoboron waste associated with traditional methods. This simplified waste profile facilitates easier regulatory approval for new manufacturing sites and reduces the environmental footprint of the production facility. As regulatory pressures on pharmaceutical manufacturers intensify regarding solvent usage and hazardous waste generation, adopting this cleaner technology positions companies favorably for future audits and compliance checks, ensuring long-term operational viability in a tightening regulatory landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric reduction technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the practical advantages for potential adopters. Understanding the nuances of ligand selection, reaction conditions, and workup procedures is essential for successfully translating this laboratory-scale innovation into a robust commercial process. We encourage technical teams to review these details closely to assess the feasibility of integrating this method into their existing synthetic routes for chiral alcohol production.
Q: What are the safety advantages of this reduction method over traditional borane methods?
A: This method replaces toxic and pyrophoric borane gas with stable, solid metal borohydrides like sodium or potassium borohydride, significantly reducing storage hazards and transportation costs.
Q: Which chiral ligands provide the highest enantioselectivity in this system?
A: Chiral beta-amino alcohol ligands derived from natural sources have demonstrated superior stereoselectivity, achieving up to 98% ee in model reactions compared to chiral diols.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the use of inexpensive, industrially available reagents and simple workup procedures involving water quenching makes this protocol highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Secondary Alcohols Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to innovative synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be reliably reproduced on an industrial scale. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of chiral intermediate meets the exacting standards required by global regulatory bodies. Our facility is equipped to handle the specific reagents and conditions outlined in this novel reduction system, allowing us to offer a seamless transition from process development to full-scale supply for your critical projects.
We invite you to engage with our technical procurement team to discuss how this advanced reduction technology can be tailored to your specific molecular targets. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits specific to your portfolio. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-quality chiral secondary alcohols efficiently. Let us collaborate to optimize your supply chain and accelerate your development timelines with our superior manufacturing solutions.
