Advanced Enzymatic Synthesis of Glutamine Dipeptide for Scalable Pharmaceutical Manufacturing
Advanced Enzymatic Synthesis of Glutamine Dipeptide for Scalable Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust, green, and cost-effective methodologies for producing critical clinical nutrition ingredients, specifically stable dipeptides like L-Alanyl-L-Glutamine (Ala-Gln). Patent CN110382705B introduces a groundbreaking biocatalytic approach that fundamentally shifts the production paradigm from traditional chemical synthesis to a highly efficient enzymatic process. This technology leverages a specialized alpha-amino acid ester acyltransferase derived from Elizabethkingia meningoseptica, engineered for exceptional stability and activity. By utilizing this novel biocatalyst, manufacturers can achieve high conversion rates under mild physiological conditions, effectively bypassing the harsh reagents and complex purification steps associated with legacy chemical routes. For R&D directors and procurement specialists, this patent represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates while adhering to stringent environmental standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical synthesis of glutamine dipeptide is fraught with significant technical and economic challenges that hinder large-scale commercial viability. Conventional routes typically involve multiple protection and deprotection steps to manage the reactive functional groups of amino acids, resulting in a lengthy and atom-inefficient process. These methods often require the use of toxic organic solvents and hazardous coupling agents, which not only increase the environmental footprint but also introduce difficult-to-remove impurities into the final product. Furthermore, the harsh reaction conditions, such as extreme pH levels or high temperatures, can lead to racemization and the formation of unwanted byproducts, necessitating expensive downstream purification processes like preparative chromatography. For supply chain managers, these complexities translate into longer lead times, higher production costs, and increased regulatory scrutiny regarding solvent residues and heavy metal contamination in the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the enzymatic method disclosed in the patent offers a streamlined, single-step biocatalytic transformation that directly couples L-alanine methyl ester hydrochloride with L-glutamine. This approach operates under mild aqueous conditions, typically maintaining a pH between 7.0 and 9.0 and temperatures ranging from 20°C to 40°C, which preserves the stereochemical integrity of the amino acid residues. The use of a highly specific alpha-amino acid ester acyltransferase ensures that the reaction proceeds with minimal side reactions, drastically reducing the impurity profile compared to chemical counterparts. By eliminating the need for protecting groups and toxic solvents, this novel route simplifies the workflow to a simple mixing and incubation process followed by straightforward isolation. This shift not only enhances the safety profile of the manufacturing facility but also significantly reduces the operational expenditure associated with waste treatment and solvent recovery, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Alpha-Amino Acid Ester Acyltransferase Catalysis
The core of this technological breakthrough lies in the unique properties of the alpha-amino acid ester acyltransferase (XPD series), which exhibits outstanding biological activity and substrate specificity. Unlike generic proteases that might hydrolyze peptide bonds, this specific acyltransferase facilitates the formation of the peptide bond between the carboxyl group of the alanine ester and the amino group of glutamine. The enzyme's active site is optimized to accommodate these specific substrates, ensuring a high turnover rate even at elevated substrate concentrations. The patent highlights that the enzyme can function efficiently with substrate mass fractions reaching up to 15%, which is substantially higher than typical enzymatic processes that suffer from substrate inhibition. This high tolerance allows for more concentrated reaction mixtures, thereby increasing the volumetric productivity of the bioreactor and reducing the water load for downstream processing. For process chemists, understanding this kinetic advantage is crucial for designing fed-batch strategies that maximize space-time yield.
A critical innovation in this patent is the genetic engineering of the expression host to prevent product degradation, a common pitfall in peptide biosynthesis. The inventors utilized CRISPR/Cas9 technology to knock out specific protease and peptidase genes, including pepA, pepB, pepD, and pepN, within the E. coli host cell. In wild-type strains, these endogenous enzymes would rapidly hydrolyze the newly synthesized glutamine dipeptide back into free amino acids, severely limiting yield. By creating a protease-deficient chassis, the patent ensures that the intracellular and extracellular environment is conducive to product accumulation rather than degradation. This genetic modification stabilizes the product in the reaction broth, allowing for longer reaction times and higher final titers without the risk of reverse hydrolysis. This level of metabolic control demonstrates a sophisticated understanding of host-pathogen interactions and enzyme stability, providing a robust platform for the commercial scale-up of complex pharmaceutical peptides.
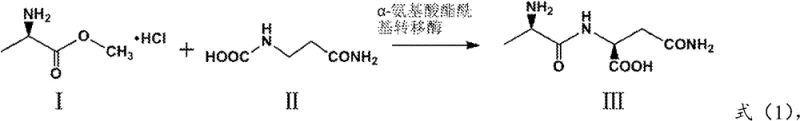
How to Synthesize L-Alanyl-L-Glutamine Efficiently
The implementation of this enzymatic route requires precise control over reaction parameters to fully leverage the catalytic efficiency of the XPD enzyme variants. The process begins with the preparation of an aqueous reaction system containing the substrates L-alanine methyl ester hydrochloride and L-glutamine, typically dissolved in a buffered solution such as borate or phosphate buffer to maintain pH stability. The engineered expression host cells, containing the recombinant plasmid pET28a-XPD, are then introduced into the reaction mixture, either as whole cells or as purified enzyme powder, depending on the specific downstream purification requirements. Maintaining the reaction temperature within the optimal range of 20°C to 40°C is essential to balance enzyme activity with thermal stability, while real-time pH monitoring ensures the reaction environment remains conducive to acyl transfer rather than hydrolysis. The detailed standardized synthesis steps, including specific primer sequences for gene modification and exact fermentation conditions, are outlined in the comprehensive guide below for technical replication.
- Prepare the reaction substrate by dissolving L-alanine methyl ester hydrochloride and L-glutamine in an aqueous buffer solution.
- Add the engineered expression host cell solution containing alpha-amino acid ester acyltransferase (XPD series) to the reaction mixture.
- Maintain the reaction at pH 7.0-9.0 and temperature 20-40°C, monitoring product formation via HPLC until conversion is complete.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic manufacturing process offers profound strategic advantages beyond mere technical feasibility. The elimination of toxic organic solvents and heavy metal catalysts fundamentally alters the cost structure of production, removing the need for expensive solvent recovery systems and rigorous heavy metal testing protocols. This simplification of the upstream process directly correlates to a reduction in capital expenditure for facility setup and a decrease in ongoing operational costs related to hazardous waste disposal. Furthermore, the use of renewable biological catalysts aligns with global sustainability goals, enhancing the corporate social responsibility profile of the supply chain. The ability to produce high-purity intermediates with fewer processing steps means that inventory turnover can be accelerated, reducing the working capital tied up in work-in-progress materials and enabling a more responsive supply chain capable of meeting fluctuating market demands for clinical nutrition products.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for expensive protecting groups and toxic coupling reagents that are standard in chemical peptide synthesis. By operating in an aqueous medium with whole-cell biocatalysts, the method significantly lowers the cost of goods sold (COGS) through reduced raw material consumption and simplified downstream processing. The high conversion rates achieved by the engineered XPD enzymes mean that less starting material is wasted, maximizing the yield per batch and optimizing the utilization of reactor volume. Additionally, the absence of harsh chemicals reduces the wear and tear on manufacturing equipment, extending asset life and lowering maintenance costs over the long term.
- Enhanced Supply Chain Reliability: Reliance on complex chemical supply chains for specialized reagents often introduces vulnerability to market fluctuations and geopolitical disruptions. In contrast, the biological components of this process, such as the expression host and substrates, are derived from widely available, commodity-grade raw materials. The robustness of the E. coli expression system ensures consistent enzyme production, mitigating the risk of batch-to-batch variability that can plague chemical synthesis. This stability allows for more accurate forecasting and planning, ensuring a continuous supply of high-quality glutamine dipeptide to meet the rigorous demands of pharmaceutical customers without unexpected interruptions or quality deviations.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is inherently safer and more environmentally compliant than scaling exothermic chemical reactions. The mild operating conditions reduce the risk of thermal runaway incidents, facilitating easier technology transfer from pilot scale to commercial production. Moreover, the aqueous nature of the waste stream simplifies effluent treatment, helping manufacturers meet increasingly stringent environmental regulations without investing in costly abatement technologies. This environmental compatibility not only reduces regulatory risk but also positions the manufacturer as a preferred partner for global pharmaceutical companies that prioritize green chemistry principles in their supplier selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented enzymatic technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical application of the XPD enzyme system. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing contexts. The answers reflect the consensus on how this biocatalytic method resolves historical bottlenecks in dipeptide production.
Q: Why is the enzymatic method superior to chemical synthesis for Ala-Gln?
A: Chemical synthesis often requires harsh conditions, toxic solvents, and complex protection/deprotection steps, leading to impurities. The enzymatic method described in CN110382705B operates under mild conditions (20-40°C, neutral pH) with high specificity, eliminating toxic byproducts and simplifying purification.
Q: How does the patent address product degradation during synthesis?
A: The patent utilizes a genetically modified E. coli host where protease and peptidase genes (such as pepA, pepB, pepD, pepN) are knocked out. This prevents the host enzymes from hydrolyzing the newly formed glutamine dipeptide, significantly improving yield and stability.
Q: What represents the key commercial advantage of this biocatalytic route?
A: The process allows for high substrate concentrations (up to 15% mass fraction) and achieves high conversion rates without expensive transition metal catalysts. This translates to reduced raw material waste, lower environmental compliance costs, and a more robust supply chain for clinical nutrition ingredients.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-Alanyl-L-Glutamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic synthesis route described in CN110382705B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities designed to handle sensitive biocatalytic processes with precision. We are committed to delivering products that meet stringent purity specifications, leveraging our expertise in protein engineering and downstream processing to guarantee the quality and consistency required for clinical applications.
We invite forward-thinking pharmaceutical partners to collaborate with us to harness the full commercial potential of this green synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our integrated manufacturing solutions can drive efficiency and reliability in your supply chain for L-Alanyl-L-Glutamine and other critical nutritional intermediates.
