Revolutionizing Chiral Amine Production with Engineered Amine Dehydrogenase Mutants
The pharmaceutical and fine chemical industries are currently witnessing a paradigm shift towards sustainable manufacturing, driven by the urgent need for greener synthesis routes and higher stereochemical precision. Patent CN110628739B introduces a groundbreaking advancement in this domain by disclosing a novel amine dehydrogenase mutant derived from Geobacillus kaustophilus phenylalanine dehydrogenase. This engineered biocatalyst addresses the critical limitations of traditional chemical synthesis, offering a robust solution for the asymmetric reductive amination of prochiral carbonyl compounds. The technology enables the production of high-purity chiral amines and amino alcohols with optical purity exceeding 99% ee, utilizing inexpensive ammonia water as the nitrogen source and generating only water as a by-product. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for complex pharmaceutical intermediates while adhering to stringent environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical methods for preparing chiral amines, such as kinetic resolution of racemates, are inherently inefficient due to a maximum theoretical yield of only 50%, which inevitably leads to significant material waste and increased raw material costs. Furthermore, asymmetric synthesis routes often rely on expensive transition metal catalysts that require rigorous removal steps to meet pharmaceutical purity standards, adding complexity and cost to the downstream processing. These chemical processes frequently necessitate harsh reaction conditions, including high temperatures and pressures, which pose safety risks and increase energy consumption. The use of stoichiometric chiral auxiliaries or resolving agents further exacerbates the environmental burden, generating substantial amounts of chemical waste that require costly disposal. Consequently, manufacturers face continuous pressure to find alternatives that can improve atom economy and reduce the overall carbon footprint of chiral amine production.
The Novel Approach
In stark contrast, the biocatalytic approach detailed in the patent utilizes a specifically engineered amine dehydrogenase mutant that operates under mild physiological conditions, typically between 20°C and 40°C, significantly reducing energy requirements. This enzymatic method achieves near-perfect stereoselectivity with product optical purity consistently greater than 99% ee, eliminating the need for complex chiral separation processes. By employing cheap ammonia water as the amine donor, the process drastically reduces raw material costs compared to expensive amine sources used in chemical synthesis. The reaction produces clean water as the sole by-product, aligning perfectly with green chemistry principles and simplifying waste treatment protocols. This novel biosynthetic pathway not only enhances the efficiency of chiral amine manufacturing but also provides a scalable and environmentally compliant solution for the production of high-value pharmaceutical intermediates.
Mechanistic Insights into GkAmDH-Catalyzed Asymmetric Reductive Amination
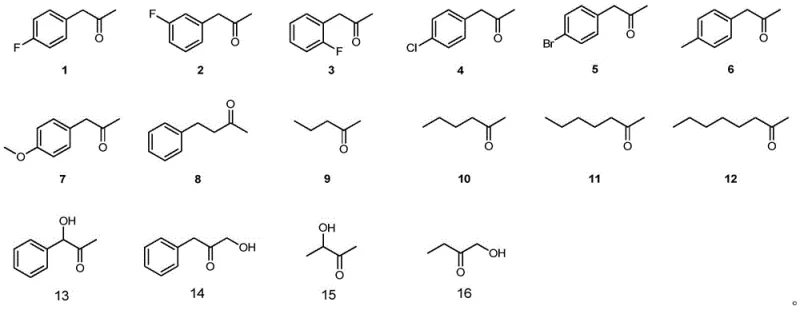
The core of this technological breakthrough lies in the molecular modification of the phenylalanine dehydrogenase from Geobacillus kaustophilus, specifically through site-directed mutagenesis at positions 78 and 276. The patent describes several high-activity mutants, such as GkAmDH_K78S/N276L, where lysine at position 78 is mutated to serine and asparagine at position 276 is mutated to leucine. These specific amino acid substitutions alter the enzyme's active site geometry and electronic environment, significantly enhancing its catalytic efficiency and substrate tolerance, particularly towards hydroxyketone substrates which are notoriously difficult for wild-type enzymes. The engineered enzyme facilitates the direct asymmetric reductive amination of prochiral ketones, utilizing the reduced form of nicotinamide adenine dinucleotide (NADH) as a hydride donor to establish the chiral center with high fidelity.
To ensure the economic viability of this biocatalytic process, the system incorporates an efficient cofactor regeneration mechanism. Since NADH is an expensive cofactor, the process couples the amine dehydrogenase reaction with a secondary enzymatic reaction, such as glucose oxidation catalyzed by glucose dehydrogenase or formate oxidation by formate dehydrogenase. This coupling allows for the in-situ regeneration of NADH from NAD+, enabling the use of catalytic rather than stoichiometric amounts of the cofactor. The substrate scope is remarkably broad, covering various aromatic and aliphatic ketones as well as hydroxyketones, as illustrated by the diverse structures capable of being converted into valuable chiral amines and amino alcohols. This versatility makes the technology applicable to a wide range of synthetic targets in the pharmaceutical and agrochemical sectors.
How to Synthesize Chiral Amines Efficiently
Implementing this biocatalytic route requires precise control over reaction parameters to maximize yield and stereoselectivity. The process begins with the preparation of a recombinant expression system, typically using E. coli BL21(DE3) transformed with the mutant gene, followed by fermentation and cell disruption to obtain the crude enzyme extract. The synthesis reaction is conducted in an ammonia/ammonium buffer system at a pH of approximately 9.0, with the addition of the ketone substrate and the cofactor regeneration system. Detailed standard operating procedures regarding enzyme loading, substrate concentration, and reaction time are critical for achieving the reported space-time yields of up to 130.9 g/L/d. The following guide outlines the standardized steps for executing this synthesis effectively.
- Prepare the reaction system using ammonia/ammonium buffer (pH 9.0) and add the specific GkAmDH mutant catalyst (e.g., GkAmDH_K78S/N276L) along with coenzyme NADH.
- Introduce the prochiral ketone substrate (e.g., 4-methoxypropiophenone) and a co-substrate like glucose or formate for in-situ NADH regeneration.
- Maintain the reaction at 20-40°C with stirring until conversion is complete, then extract the chiral amine product using dichloromethane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic technology translates into tangible strategic advantages beyond mere technical performance. The elimination of transition metal catalysts removes the need for expensive and time-consuming heavy metal scavenging steps, which significantly streamlines the purification process and reduces the consumption of specialized resins or activated carbon. The use of ammonia water as a nitrogen source offers a substantial cost reduction compared to traditional amine donors, while the mild reaction conditions lower energy costs associated with heating and cooling. Furthermore, the high conversion rates and selectivity minimize the formation of by-products, leading to higher overall yields and reduced waste disposal costs. These factors collectively contribute to a more resilient and cost-effective supply chain for chiral building blocks.
- Cost Reduction in Manufacturing: The biocatalytic process eliminates the need for precious metal catalysts and complex chiral ligands, which are major cost drivers in traditional chemical synthesis. By using inexpensive ammonia and regenerating the cofactor in situ, the raw material costs are drastically simplified and optimized. The high stereoselectivity ensures that nearly all starting material is converted into the desired enantiomer, avoiding the 50% yield loss inherent in kinetic resolution methods. This improvement in atom economy directly lowers the cost of goods sold (COGS) and enhances the profitability of manufacturing high-purity chiral amines.
- Enhanced Supply Chain Reliability: Reliance on enzymatic catalysts produced via fermentation reduces dependency on fluctuating markets for rare earth metals or complex chemical reagents. The recombinant enzyme can be produced consistently at scale, ensuring a stable supply of the biocatalyst for continuous manufacturing operations. Additionally, the mild reaction conditions reduce the risk of safety incidents and equipment corrosion, leading to fewer unplanned downtimes. This stability is crucial for maintaining consistent delivery schedules for critical pharmaceutical intermediates, thereby strengthening the reliability of the overall supply chain.
- Scalability and Environmental Compliance: The process generates water as the only by-product, significantly reducing the environmental burden and simplifying compliance with increasingly strict environmental regulations. The absence of toxic organic solvents and heavy metals facilitates easier waste treatment and lowers the costs associated with environmental protection measures. The technology has demonstrated high space-time yields, indicating strong potential for scale-up from laboratory to industrial production without significant loss of efficiency. This scalability ensures that the supply can meet growing market demands while maintaining a sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this amine dehydrogenase technology. These answers are derived directly from the patent data and technical specifications to provide clarity on the process capabilities and limitations. Understanding these details is essential for evaluating the feasibility of integrating this biocatalytic route into existing manufacturing workflows.
Q: What are the advantages of using amine dehydrogenase mutants over traditional chemical synthesis?
A: Unlike traditional chemical methods that often suffer from low theoretical yields (max 50% for kinetic resolution) and require harsh conditions or transition metals, the amine dehydrogenase mutants described in patent CN110628739B offer 100% theoretical yield, >99% optical purity, and operate under mild, environmentally friendly conditions using cheap ammonia.
Q: Can this biocatalytic process handle hydroxyketone substrates for amino alcohol synthesis?
A: Yes, a key innovation of this technology is the specific engineering of the enzyme (e.g., mutations at positions 78 and 276) to significantly enhance activity and stability towards hydroxyketone substrates, enabling the efficient synthesis of chiral amino alcohols which were previously difficult to produce with high stereoselectivity using wild-type enzymes.
Q: How is the coenzyme NADH managed to ensure cost-effectiveness in large-scale production?
A: The process utilizes an in-situ cofactor regeneration system. By coupling the reaction with glucose dehydrogenase or formate dehydrogenase, the oxidized NAD+ is continuously recycled back to NADH using inexpensive co-substrates like glucose or formate, eliminating the need for stoichiometric amounts of expensive cofactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amines Supplier
NINGBO INNO PHARMCHEM stands at the forefront of biocatalytic innovation, leveraging advanced enzymatic technologies like the amine dehydrogenase mutants described in patent CN110628739B to deliver superior chiral amine solutions. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of chiral amine or amino alcohol meets the highest quality standards required by the global pharmaceutical industry.
We invite you to collaborate with our technical procurement team to explore how this green synthesis route can optimize your specific supply chain. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the potential economic benefits of switching to our biocatalytic processes. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us help you achieve greater efficiency and sustainability in your chiral synthesis operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
