Industrial Scale-Up of Aryl Compounds via Safe Nickel-Catalyzed Cross-Coupling for Global Markets
The chemical industry is constantly seeking robust methodologies to construct biaryl scaffolds, which serve as critical backbones for active pharmaceutical ingredients and advanced functional materials. Patent CN1466487A presents a transformative approach to preparing aryl compounds through a nickel-catalyzed cross-coupling reaction between substituted aryl halides and Grignard reagents. This technology fundamentally shifts the operational paradigm from hazardous cryogenic conditions to manageable industrial temperatures, addressing long-standing safety and scalability concerns in fine chemical synthesis. By utilizing a novel supported nickel catalyst system combined with a controlled metering strategy for the Grignard reagent, this process achieves high selectivity and yield without the need for extreme cooling. The reaction scheme illustrates the core transformation where an aryl halide (Ar-X) couples with an organomagnesium species (R-Mg-Hal) to form the desired biaryl product (Ar-R) under nickel catalysis.
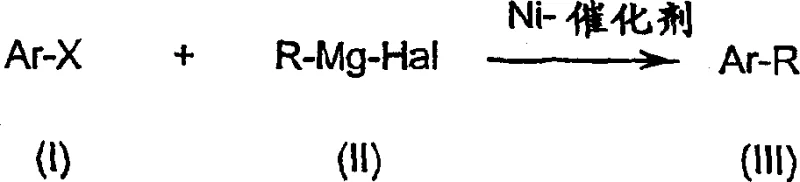
This innovation is particularly relevant for manufacturers aiming to become a reliable pharmaceutical intermediate supplier, as it streamlines the production of complex molecular architectures. The patent details not only the coupling methodology but also the preparation of the catalyst itself, emphasizing air-stable precursors that simplify supply chain logistics. For R&D teams evaluating process intensification, this technology offers a compelling alternative to palladium-based systems or harsh lithium-mediated couplings, providing a balance of cost-efficiency and performance that is essential for commercial viability in the competitive global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl compounds via cross-coupling with Grignard reagents has been plagued by significant operational hazards and inefficiencies, particularly when relying on heterogeneous Ni/C catalysts as described in prior art such as Inorg. Chim. Acta 296, 164 (1999). A primary drawback of these conventional procedures is the stringent requirement for cryogenic conditions, typically necessitating the addition of Grignard reagents at temperatures as low as -78°C. Maintaining such extreme低温 on an industrial scale is energetically prohibitive and requires specialized equipment that drastically increases capital expenditure. Furthermore, the batch-wise addition of the entire Grignard charge at low temperature creates a latent safety risk; if the reaction initiation is delayed, a sudden exotherm upon warming can lead to thermal runaway, posing severe threats to plant safety and personnel. Additionally, the catalyst precursors used in these older methods often require preparation under strict inert atmospheres and storage in the absence of air, complicating handling and increasing the risk of catalyst deactivation before use.
The Novel Approach
In stark contrast, the methodology disclosed in CN1466487A introduces a sophisticated process control strategy that eliminates the need for cryogenic cooling while enhancing reaction safety and selectivity. The core innovation lies in the reverse addition protocol: instead of adding the Grignard reagent to a cold mixture, the substituted aryl compound and the nickel catalyst are pre-charged and heated to the reaction temperature, typically ranging from 20°C to 120°C, preferably 35-100°C. The Grignard reagent is then metered in slowly over a period of hours, ensuring that its concentration in the reaction mixture remains consistently low. This kinetic control prevents the accumulation of unreacted organometallic species, thereby mitigating the risk of violent exotherms and suppressing side reactions such as homocoupling. Moreover, the use of air-stable catalyst precursors, which can be prepared without organic solvents or inert gas protection, represents a logistical breakthrough, allowing for easier transport and storage. This approach facilitates cost reduction in fine chemical manufacturing by removing the energy burden of deep freezing and simplifying the reactor setup.
Mechanistic Insights into Ni-Catalyzed Cross-Coupling
The efficacy of this process is rooted in the unique behavior of the supported nickel catalyst, which operates through a heterogeneous mechanism that combines the activity of homogeneous systems with the separability of heterogeneous ones. The catalyst, typically comprising nickel nanoparticles dispersed on high-surface-area supports like activated carbon, silica, or alumina, facilitates the oxidative addition of the aryl halide to the Ni(0) center. This step is critical and is significantly enhanced by the presence of phosphorus ligands, such as triphenylphosphine or triphenylphosphite, which stabilize the active nickel species and modulate its electronic properties. Following oxidative addition, the transmetallation step occurs with the Grignard reagent. By maintaining a low concentration of the Grignard reagent through metered addition, the process ensures that transmetallation occurs efficiently on the catalyst surface without overwhelming the system. This controlled environment favors the desired cross-coupling pathway over competing decomposition routes, leading to the formation of the Ar-R bond with high fidelity. The final reductive elimination step releases the product and regenerates the Ni(0) catalyst, completing the cycle.
From an impurity control perspective, the mechanistic design of this process offers substantial advantages for producing high-purity OLED material or pharmaceutical intermediates. In traditional bulk addition methods, high local concentrations of Grignard reagent can lead to Wurtz-type homocoupling of the aryl halide or nucleophilic attack on sensitive functional groups present on the aromatic ring. The metered addition strategy effectively suppresses these side reactions by keeping the Grignard reagent as the limiting reagent at the catalyst interface throughout the reaction duration. Furthermore, the heterogeneous nature of the catalyst minimizes nickel leaching into the product stream, a critical factor for meeting stringent regulatory limits on heavy metals in drug substances. The ability to filter off the solid catalyst post-reaction not only simplifies purification but also allows for catalyst recycling, contributing to a greener and more sustainable manufacturing footprint. This level of control over the reaction trajectory ensures that the final product meets rigorous quality specifications with minimal downstream processing.
How to Synthesize Aryl Compounds Efficiently
To implement this advanced coupling technology in a production setting, operators must adhere to a precise sequence of operations that leverages the air-stability of the catalyst precursors and the thermal safety of the metered addition protocol. The process begins with the activation or preparation of the supported nickel catalyst, followed by the assembly of the reaction mixture under mild inert conditions. The key to success lies in the thermal management during the Grignard dosing phase, where temperature control dictates the reaction rate and selectivity. Detailed standard operating procedures regarding specific stoichiometry, solvent choices like tetrahydrofuran, and workup protocols are essential for reproducibility. The following guide outlines the critical phases of this synthesis, serving as a foundational reference for process engineers aiming to commercial scale-up of complex polymer additives or agrochemical intermediates.
- Preparation of Air-Stable Nickel Catalyst: Impregnate support material (activated carbon, silica, or alumina) with aqueous nickel salt solution, dry at 150-400°C, and reduce to form Ni(0) species.
- Reaction Setup and Metered Addition: Precharge aryl halide and catalyst in solvent (e.g., THF), heat to 35-100°C, and slowly meter in the Grignard reagent to maintain low concentration.
- Workup and Purification: Quench reaction with alcohol or water, filter off the heterogeneous catalyst for reuse, and distill the filtrate under high vacuum to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1466487A translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of cryogenic requirements fundamentally alters the cost structure of the manufacturing process, removing the need for expensive refrigeration units and the associated high energy consumption required to maintain sub-zero temperatures. This shift allows for the utilization of standard glass-lined or stainless steel reactors equipped with conventional heating and cooling systems, thereby maximizing asset utilization and reducing capital intensity. Furthermore, the air-stability of the catalyst precursors significantly de-risks the supply chain; unlike sensitive organometallic catalysts that require cold chain logistics and inert atmosphere shipping, these materials can be transported and stored under ambient conditions. This robustness ensures supply continuity and reduces the administrative burden associated with handling hazardous or sensitive materials, making the overall procurement process more resilient and cost-effective.
- Cost Reduction in Manufacturing: The process achieves significant economic optimization by removing the energy-intensive cryogenic step, which is traditionally a major cost driver in Grignard reactions. By operating at near-ambient or reflux temperatures, the facility saves substantially on utility costs related to chilling brine or liquid nitrogen. Additionally, the ability to recover and reuse the heterogeneous nickel catalyst through simple filtration reduces the consumption of expensive metal resources and lowers waste disposal costs. The high selectivity achieved through metered addition minimizes the formation of by-products, which in turn reduces the load on purification units such as distillation columns or chromatography systems, leading to further operational savings and higher overall throughput.
- Enhanced Supply Chain Reliability: The use of air-stable catalyst precursors simplifies inventory management and logistics, as there is no longer a dependency on specialized inert packaging or temperature-controlled warehousing. This stability ensures that raw materials remain viable for longer periods, reducing the risk of spoilage and write-offs. Moreover, the process utilizes common and readily available starting materials such as aryl chlorides and bromides, along with standard Grignard reagents, which are widely sourced from established chemical suppliers. This broadens the supplier base and mitigates the risk of single-source bottlenecks, ensuring a steady flow of materials necessary for continuous production schedules and timely delivery to customers.
- Scalability and Environmental Compliance: The inherent safety of the metered addition protocol makes this process highly scalable from pilot plant to multi-ton production without the exponential increase in risk associated with batch exotherms. The controlled release of heat allows for safer operation in larger vessels, facilitating the commercial scale-up of complex fine chemicals. Environmentally, the process aligns with green chemistry principles by reducing solvent usage through potential recycling and minimizing heavy metal waste via catalyst recovery. The absence of cryogenic fluids also reduces the facility's carbon footprint, supporting corporate sustainability goals and ensuring compliance with increasingly stringent environmental regulations regarding energy use and chemical emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this nickel-catalyzed cross-coupling technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing clarity on safety, catalyst handling, and product quality. Understanding these aspects is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The responses highlight the practical advantages of the method, focusing on its robustness and suitability for industrial application.
Q: How does this nickel-catalyzed process improve safety compared to traditional methods?
A: Traditional methods often require cryogenic temperatures (-78°C) and bulk addition of Grignard reagents, leading to dangerous delayed exotherms. This patent eliminates cryogenics by using metered addition at reaction temperature (20-120°C), ensuring thermal control and operational safety.
Q: What are the storage requirements for the novel nickel catalyst precursors?
A: Unlike conventional sensitive catalysts requiring inert atmosphere storage, the oxide precursor materials described in this patent are insensitive to oxygen and can be stored in air, significantly simplifying logistics and handling.
Q: Can the supported nickel catalyst be recovered and reused?
A: Yes, the heterogeneous nature of the supported nickel catalyst allows for simple filtration after the reaction. The patent explicitly describes washing and drying the catalyst for reuse in subsequent batches, reducing heavy metal waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes that balance efficiency, safety, and quality. Our technical team has extensively analyzed the potential of the nickel-catalyzed cross-coupling process described in CN1466487A and is fully prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory concept to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of aryl compounds delivered meets the highest international standards required by the pharmaceutical and agrochemical industries.
We invite you to collaborate with us to explore how this advanced manufacturing technique can optimize your supply chain and reduce your overall production costs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating the economic benefits of switching to this safer, more efficient protocol. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to drive innovation and reliability in your chemical supply chain, ensuring your projects succeed with the highest level of technical support and commercial integrity.
