Revolutionizing Biaryl Synthesis: Green Aqueous Suzuki Coupling for Commercial Scale Production
The landscape of fine chemical manufacturing is undergoing a paradigm shift towards sustainability, driven by the urgent need to reduce environmental footprints while maintaining high efficiency in the production of complex molecular scaffolds. Patent CN102491862A presents a groundbreaking methodology for the preparation of biaryl compounds through a Suzuki cross-coupling reaction conducted entirely in pure water, marking a significant departure from traditional organic solvent-dependent processes. This innovation addresses the critical challenge of reacting water-insoluble substrates in an aqueous environment without the aid of organic co-solvents or complex surfactant systems, thereby offering a robust pathway for the synthesis of high-purity pharmaceutical intermediates. By leveraging a specialized palladium catalyst system combined with methoxy-terminated polyethylene glycol-modified imidazolium salts, this technology enables reactions to proceed at 100°C with exceptional yields, ranging from 92% to 98% across various substrate scopes. For R&D directors and process chemists, this represents a viable route to streamline synthesis workflows, while procurement managers will recognize the inherent cost advantages of replacing expensive organic solvents with water. As a reliable pharmaceutical intermediates supplier, understanding such technological leaps is essential for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional Suzuki-Miyaura cross-coupling reactions, while powerful for constructing Csp2-Csp2 bonds found in countless active pharmaceutical ingredients (APIs) and agrochemical intermediates, have historically been plagued by significant operational and environmental drawbacks. Conventional protocols typically rely on volatile organic solvents such as toluene, dioxane, or DMF, which necessitate rigorous recovery systems and generate substantial hazardous waste streams that increase disposal costs and regulatory burdens. Furthermore, the standard ligand systems often employ phosphines, which are notoriously sensitive to oxygen and moisture, requiring strict inert gas protection (nitrogen or argon) throughout the reaction and workup phases. This dependency on anhydrous and anaerobic conditions not only complicates the engineering requirements for large-scale reactors but also introduces potential safety hazards associated with handling pyrophoric reagents. Additionally, the separation of products from these high-boiling organic solvents often requires energy-intensive distillation processes, and the removal of trace phosphine oxides or metal residues can be challenging, potentially compromising the purity profile required for downstream drug substance manufacturing. These factors collectively contribute to higher production costs and longer lead times, creating bottlenecks in the supply chain for high-purity biaryl compounds.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102491862A introduces a transformative approach by utilizing pure water as the sole reaction medium, effectively eliminating the need for any organic co-solvents. The core of this innovation lies in the use of methoxy-terminated polyethylene glycol (PEG)-modified imidazolium salts, which serve as precursors for N-heterocyclic carbene (NHC) ligands that exhibit superior stability and solubilizing properties. Unlike traditional surfactants that can be difficult to separate from the final product, these PEG-modified ligands facilitate the reaction of hydrophobic aryl halides and boronic acids in water through a unique micellar-like environment without forming stable emulsions that hinder extraction. The synthesis of these critical ligand precursors involves a straightforward two-step process starting from commercially available PEG monomethyl ethers, as illustrated in the reaction scheme below, ensuring that the supply of these specialized reagents remains robust and scalable.
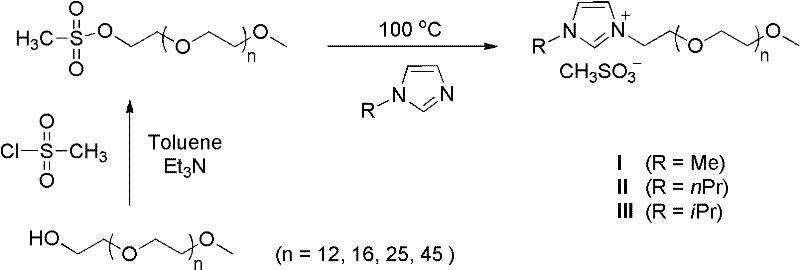
This novel catalytic system operates efficiently at 100°C without the necessity for inert gas protection, a feature that drastically simplifies reactor design and operational procedures. The elimination of oxygen-sensitive phosphine ligands in favor of air-stable NHC precursors means that reactions can be set up in open vessels or with minimal headspace flushing, reducing the consumption of nitrogen and the complexity of sealing systems. For manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing, this transition to an aqueous system translates directly into lower utility costs for solvent recovery and waste treatment, while simultaneously enhancing the safety profile of the production facility. The method demonstrates broad substrate tolerance, successfully coupling various aryl halides including those with electron-withdrawing and electron-donating groups, proving its versatility for diverse synthetic applications.
Mechanistic Insights into Pd/NHC-Catalyzed Aqueous Suzuki Coupling
The mechanistic superiority of this aqueous system stems from the unique electronic and steric properties of the in situ generated Palladium-N-heterocyclic carbene (Pd-NHC) complex. NHC ligands are known for their strong sigma-donating capabilities, which stabilize the palladium center in low oxidation states more effectively than traditional phosphines, thereby facilitating the oxidative addition step even with less reactive aryl chlorides or sterically hindered substrates. In the aqueous environment, the polyethylene glycol chain attached to the imidazolium ring plays a dual role: it acts as a phase-transfer mediator that brings the hydrophobic organic substrates into close proximity with the catalyst, and it stabilizes the catalytic species against aggregation or precipitation. This "on-water" effect accelerates the reaction kinetics, allowing for completion times as short as 5 minutes for highly reactive substrates, while maintaining high turnover numbers that minimize the required loading of the precious palladium catalyst. The robustness of the Pd-NHC bond ensures that the catalyst remains active throughout the reaction cycle, preventing premature deactivation which is a common failure mode in aqueous organometallic chemistry.
From an impurity control perspective, the aqueous nature of the reaction medium offers distinct advantages for downstream purification. Since the reaction occurs in water, inorganic byproducts such as salts formed from the base (triethylamine hydrochloride) remain dissolved in the aqueous phase upon quenching with saturated saline solution. This allows for a clean separation of the organic product via simple liquid-liquid extraction with ethyl acetate, avoiding the complex filtration steps often required to remove insoluble inorganic bases or catalyst residues in organic solvents. The absence of high-boiling organic solvents means that the crude product obtained after concentration is significantly cleaner, reducing the burden on the final column chromatography or crystallization steps. This streamlined workup process not only improves the overall yield by minimizing product loss during purification but also ensures that the final biaryl compounds meet stringent purity specifications required for clinical grade materials, addressing a primary concern for R&D directors focused on quality assurance.
How to Synthesize Biaryl Compounds Efficiently
The implementation of this green synthesis protocol is designed to be straightforward and adaptable to existing manufacturing infrastructure, requiring minimal modification to standard batch reactors. The process begins with the precise charging of reagents, where the molar ratios are optimized to balance reaction rate and cost efficiency, typically employing a slight excess of the boronic acid to drive the equilibrium forward. Detailed standard operating procedures regarding temperature ramping, stirring speeds, and quenching protocols are critical to ensuring reproducibility at scale, and operators should adhere strictly to the defined parameters to maximize yield and minimize side reactions. The following guide outlines the standardized synthesis steps derived from the patent data, providing a clear roadmap for process engineers to validate this technology in their pilot plants.
- Prepare the reaction mixture by adding palladium catalyst, methoxy-terminated polyethylene glycol-modified imidazolium salt, aryl halide, aryl boronic acid, base, and water into a round bottom flask.
- Heat the mixture to 100°C and stir for 5 to 480 minutes to allow the Suzuki cross-coupling reaction to proceed to completion without inert gas protection.
- Quench the reaction with saturated saline solution, extract the product with ethyl acetate, concentrate the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous Suzuki coupling technology presents a compelling value proposition centered on risk mitigation and operational efficiency. The shift from organic solvents to water fundamentally alters the cost structure of the manufacturing process, removing the volatility associated with petrochemical-derived solvent prices and reducing the logistical complexity of storing and handling flammable liquids. Furthermore, the simplified reaction conditions eliminate the need for specialized inert atmosphere equipment, allowing for the utilization of standard glass-lined or stainless steel reactors that are already prevalent in multi-purpose chemical facilities. This compatibility with existing assets accelerates the timeline for technology transfer and commercial scale-up of complex pharmaceutical intermediates, enabling faster time-to-market for new drug candidates. The robustness of the supply chain is further enhanced by the use of commodity chemicals such as polyethylene glycol and imidazole for ligand synthesis, ensuring that raw material availability is not a bottleneck for production continuity.
- Cost Reduction in Manufacturing: The elimination of expensive organic solvents and the reduction in energy consumption for solvent recovery result in substantial cost savings per kilogram of product. By removing the requirement for inert gas protection, facilities can save significantly on nitrogen consumption and the maintenance of leak-tight systems, while the high catalytic efficiency reduces the loading of costly palladium metals. These cumulative efficiencies translate into a more competitive pricing structure for the final biaryl intermediates without compromising on quality or yield.
- Enhanced Supply Chain Reliability: Utilizing water as a solvent mitigates the risks associated with the supply and disposal of hazardous organic chemicals, which are often subject to fluctuating regulatory restrictions and transportation limitations. The simplicity of the workup procedure, involving basic extraction and concentration, reduces the processing time and minimizes the potential for equipment downtime due to fouling or clogging commonly seen with viscous organic reaction mixtures. This reliability ensures consistent delivery schedules and strengthens the resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: The green nature of this process aligns perfectly with increasingly stringent environmental regulations, reducing the generation of hazardous waste and lowering the carbon footprint of the manufacturing operation. The absence of VOC emissions simplifies permitting processes and reduces the need for expensive abatement technologies, making it easier to scale production from pilot batches to multi-ton commercial volumes. This environmental compliance not only protects the company from regulatory fines but also enhances its reputation as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this aqueous Suzuki coupling technology, based on the specific data and embodiments provided in the patent literature. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating this method into their current production portfolios. The answers provided reflect the proven capabilities of the system as demonstrated in the experimental examples, offering clarity on performance expectations and operational requirements.
Q: How does this method solve the solubility issue of hydrophobic substrates in water?
A: The method utilizes methoxy-terminated polyethylene glycol-modified imidazolium salts which act as amphiphilic ligands, effectively solubilizing non-water-soluble substrates in the pure aqueous phase without requiring organic co-solvents.
Q: Is inert gas protection required for this Suzuki coupling process?
A: No, unlike traditional phosphine ligand systems which are oxygen-sensitive, the Pd/N-heterocyclic carbene system described in patent CN102491862A is insensitive to oxygen, eliminating the need for inert gas protection and simplifying operations.
Q: What are the environmental benefits of using pure water as a solvent?
A: Using pure water eliminates the need for volatile organic compounds (VOCs) and toxic organic solvents, significantly reducing waste treatment costs and aligning with green chemistry principles for sustainable manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biaryl Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green chemistry technologies like the aqueous Suzuki coupling method described in CN102491862A to redefine the standards of efficiency and sustainability in fine chemical production. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. We are committed to delivering high-purity biaryl compounds that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch. Our capability to handle complex catalytic systems and aqueous chemistries positions us as a strategic partner for clients seeking to optimize their supply chains for next-generation pharmaceutical intermediates.
We invite you to collaborate with us to leverage these advanced synthetic methodologies for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your target molecules, and let us provide you with specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply of high-quality intermediates produced through cutting-edge, environmentally responsible processes that drive value and innovation in your drug development pipeline.
