Scalable Synthesis of 16α-Hydroxyprednisolone: A Technical Breakthrough for API Manufacturing
Scalable Synthesis of 16α-Hydroxyprednisolone: A Technical Breakthrough for API Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective routes for synthesizing high-value corticosteroid intermediates. Patent CN111253457A introduces a significant advancement in the preparation of 16α-hydroxyprednisolone, a critical building block for modern glucocorticoids such as budesonide and ciclesonide. This novel methodology addresses long-standing challenges in steroid functionalization by replacing hazardous and inefficient reagents with a streamlined four-step sequence involving oxidation, bromohydroxylation, debromination, and alcoholysis. By utilizing 21-hydroxypregna-1,4,9(11),16-tetraene-3,20-dione-21-acetate as the starting material, the process achieves exceptional control over stereoselectivity and impurity profiles. For R&D directors and procurement specialists, this technology represents a viable pathway to secure a reliable supply of high-purity pharmaceutical intermediates while mitigating the operational risks associated with traditional heavy metal reductions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 16α-hydroxyprednisolone has been plagued by inefficiencies inherent in older synthetic strategies, such as the route disclosed in CN201310698900.3. Traditional methods often rely on prednisolone as a starting material, necessitating a cumbersome sequence of cyclization, ring-opening, esterification, elimination, and re-hydrolysis. A critical bottleneck in these conventional pathways occurs during the elimination of hydroxyl groups to prepare 16,17-ene compounds, a step prone to generating substantial side-reaction impurities that are difficult to remove. Furthermore, the reliance on expensive starting materials like prednisolone drastically inflates the cost of goods sold (COGS), rendering these processes economically unviable for large-scale industrial production. The complexity of the reaction steps also increases the cumulative yield loss, often resulting in overall conversions that fail to meet the rigorous demands of commercial API manufacturing.
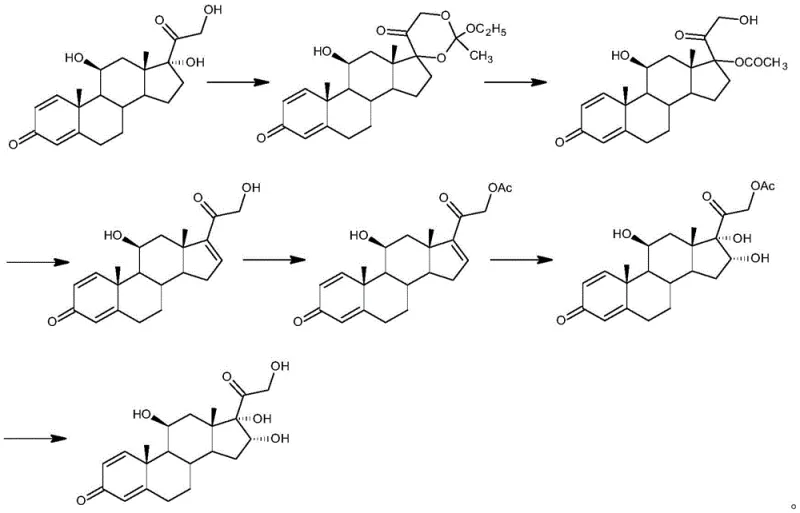
The Novel Approach
In stark contrast, the methodology outlined in CN111253457A offers a streamlined alternative that bypasses the economic and technical pitfalls of prior art. The new route initiates with the readily available and cost-effective 21-hydroxypregna-1,4,9(11),16-tetraene-3,20-dione-21-acetate. The process is elegantly divided into four distinct chemical transformations: a controlled oxidation to form an epoxide intermediate, followed by regioselective bromohydroxylation. The subsequent debromination step utilizes a superior reagent system to install the critical 16α-hydroxyl group, concluding with a mild alcoholysis to reveal the final product. This approach not only simplifies the operational workflow but also inherently minimizes the formation of by-products, ensuring a cleaner reaction profile. By reducing the number of unit operations and avoiding expensive precursors, this novel approach establishes a new benchmark for efficiency in steroid intermediate manufacturing.
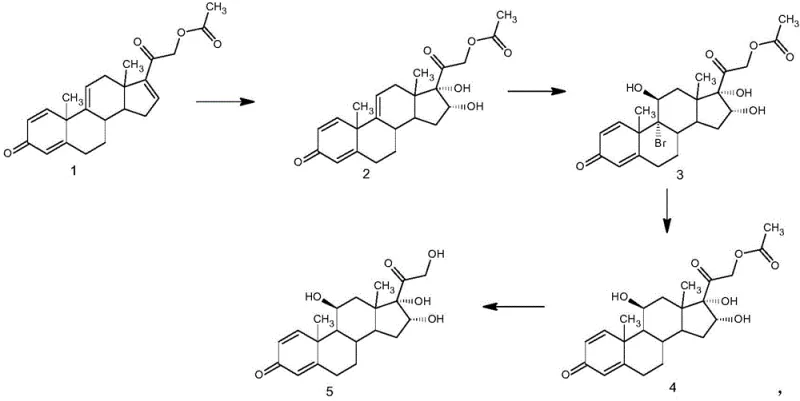
Mechanistic Insights into Oxidative Functionalization and Debromination
The success of this synthetic route hinges on precise control over reaction conditions, particularly during the oxidation and debromination phases. In the initial oxidation step, potassium permanganate is employed as the oxidant under strictly controlled acidic conditions. The patent specifies maintaining the reaction temperature between -20°C and -5°C, a critical parameter that prevents over-oxidation and the degradation of the sensitive steroid skeleton. The use of acidic catalysts such as formic acid or oxalic acid facilitates the formation of the epoxide intermediate while suppressing the generation of oxidative by-products. Following this, the bromohydroxylation step introduces functionality at the C16 and C17 positions using N-bromosuccinimide or dibromohydantoin. The choice of acidic catalyst in this step, such as fluoroboric acid or methanesulfonic acid, is crucial for directing the stereochemical outcome, ensuring the correct orientation of the incoming hydroxyl and bromine groups necessary for the final biological activity.
Perhaps the most significant mechanistic innovation lies in the debromination step, which traditionally relies on zinc powder. The patented method replaces zinc with a combination of thioglycolic acid and chromous chloride (CrCl2) in hydrochloric acid. This substitution is transformative because zinc powder is physically dense and prone to agglomeration, leading to poor mixing and incomplete reactions in large reactors. In contrast, the CrCl2/thioglycolic acid system offers superior solubility and reactivity, allowing the reaction to proceed efficiently at mild temperatures ranging from -5°C to 15°C. This enhanced reactivity not only shortens the reaction time but also drastically reduces the formation of elimination by-products. The mechanism likely involves a single-electron transfer facilitated by the chromium species, which selectively removes the bromine atom while preserving the newly formed 16α-hydroxyl configuration, thereby securing high stereochemical purity.
How to Synthesize 16α-Hydroxyprednisolone Efficiently
Implementing this synthesis requires adherence to specific operational parameters to maximize yield and purity. The process begins with the dissolution of the tetraene acetate starting material in a ketone solvent like acetone or butanone, followed by the careful addition of the oxidant at sub-zero temperatures. Subsequent steps involve precise stoichiometric control of the brominating agents and the chromium reductant. The final alcoholysis step utilizes alkali metal hydroxides in alcoholic solvents to cleave the acetate protecting group without affecting the sensitive ketone functionalities. For a detailed breakdown of the specific reagent quantities, solvent volumes, and workup procedures required to replicate this high-yielding process, please refer to the standardized synthesis guide below.
- Perform controlled oxidation of the tetraene acetate starting material using potassium permanganate at low temperatures (-20 to -5°C) to form the epoxide intermediate.
- Execute bromohydroxylation using N-bromosuccinimide or dibromohydantoin in the presence of an acidic catalyst to introduce the hydroxyl and bromine groups.
- Conduct debromination using thioglycolic acid and chromous chloride (CrCl2) to remove the bromine atom while retaining stereochemistry.
- Finalize the synthesis through alcoholysis with an alkali metal hydroxide solution to deprotect the acetate group and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented process offers substantial strategic advantages for supply chain managers and procurement officers seeking to optimize their API ingredient sourcing. The shift away from expensive starting materials like prednisolone to more abundant tetraene acetates fundamentally alters the cost structure of the intermediate. Furthermore, the elimination of problematic reagents like zinc powder reduces the burden on waste treatment facilities and simplifies the filtration and purification stages. These operational improvements translate directly into a more resilient supply chain, capable of meeting fluctuating market demands for asthma and allergy medications without the bottlenecks associated with complex, low-yield syntheses.
- Cost Reduction in Manufacturing: The replacement of zinc powder with the CrCl2/thioglycolic acid system in the debromination step is a primary driver of cost efficiency. As noted in the patent data, this specific modification can reduce processing costs by 15-20% in this critical step alone by eliminating the need for extensive mechanical stirring to handle heavy metal slurries and reducing reaction times. Additionally, the overall conversion rate exceeding 70% ensures that raw material costs are amortized over a larger volume of saleable product, significantly lowering the cost per kilogram of the final intermediate compared to legacy methods.
- Enhanced Supply Chain Reliability: The robustness of this four-step sequence enhances supply continuity by minimizing the risk of batch failures due to impurity buildup. Traditional methods often suffer from variable yields caused by the unpredictable behavior of zinc powder in large-scale reactors. By utilizing homogeneous or semi-homogeneous reagent systems that are easier to mix and control, manufacturers can achieve consistent batch-to-batch reproducibility. This reliability is crucial for maintaining the steady flow of intermediates required for the continuous production of downstream glucocorticoids like budesonide, ensuring that pharmaceutical customers face no interruptions in their own manufacturing schedules.
- Scalability and Environmental Compliance: The mild reaction conditions, particularly the ability to run the debromination at temperatures as high as 15°C rather than requiring extreme cooling or heating, reduce the energy load on production facilities. Moreover, the high purity of the crude product (>99.5% after simple recrystallization) minimizes the need for resource-intensive chromatographic purifications. This aligns with modern green chemistry principles by reducing solvent consumption and waste generation, making the process easier to scale from pilot plants to multi-ton commercial production while meeting increasingly stringent environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in patent CN111253457A, providing clarity on the feasibility and advantages of adopting this route for industrial applications.
Q: What are the critical advantages of the CrCl2 debromination method over traditional zinc powder reduction?
A: The use of thioglycolic acid and CrCl2 eliminates the physical handling issues associated with heavy zinc powder, such as clumping and uneven stirring. This results in more complete reactions, better temperature control (-5 to 15°C), and significantly reduced impurity formation compared to traditional methods.
Q: What is the expected purity and yield of 16α-hydroxyprednisolone using this patented route?
A: According to patent CN111253457A, the optimized process achieves an overall mass yield exceeding 70% and a final product purity greater than 99.5% as determined by HPLC, making it highly suitable for downstream API synthesis.
Q: Why is 16α-hydroxyprednisolone considered a high-value pharmaceutical intermediate?
A: It serves as the essential halogen-free precursor for synthesizing next-generation glucocorticoids like budesonide and ciclesonide. These drugs are critical for treating asthma and allergic rhinitis due to their high local anti-inflammatory potency and reduced systemic side effects.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16α-Hydroxyprednisolone Supplier
The technical potential of this synthesis route underscores the importance of partnering with a manufacturer capable of executing complex steroid chemistry with precision. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of 16α-hydroxyprednisolone meets the exacting standards required for the synthesis of life-saving respiratory medications.
We invite global pharmaceutical partners to leverage our technical expertise to secure a stable supply of this critical intermediate. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce overall production costs.
