Breakthrough Purification Technology for High-Purity 16α-Hydroxyprednisolone Manufacturing
The pharmaceutical industry continuously demands higher purity standards for corticosteroid intermediates, particularly for critical molecules like 16α-hydroxyprednisolone. A recent technological breakthrough detailed in patent CN112125943A introduces a sophisticated purification strategy that addresses long-standing challenges in removing structurally similar byproducts. This innovation is pivotal for manufacturers seeking to secure a reliable pharmaceutical intermediate supplier capable of delivering material that exceeds the stringent 99.5% purity threshold required for downstream API synthesis. By shifting away from traditional recrystallization limitations, this method leverages selective chemical modification to achieve unprecedented levels of purity.
The core of this advancement lies in a novel post-synthesis treatment protocol that effectively eliminates 'Impurity H', a persistent contaminant generated during the upstream synthesis from tetraene acetate. Unlike conventional approaches that struggle to separate this impurity due to its structural homology with the target compound, the new methodology employs a controlled hypochlorite oxidation step followed by a specific precipitation technique. This ensures that the final 16α-hydroxyprednisolone not only meets but significantly surpasses global quality specifications, offering a robust solution for cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 16α-hydroxyprednisolone starting from tetraene acetate has been plagued by the formation of a specific byproduct known as Impurity H. As illustrated in the reaction pathway below, this impurity arises naturally during the synthetic sequence and shares a nearly identical steroid backbone with the desired product. 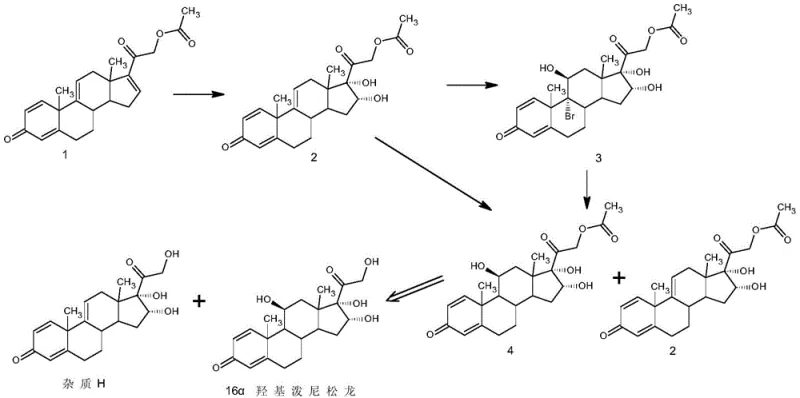
Traditional purification strategies, primarily relying on simple recrystallization from solvents like methanol, have proven inadequate for removing this specific contaminant. Data indicates that standard methods often result in final products containing approximately 0.90% of Impurity H, with overall purity stagnating around 98.2%. This level of contamination is unacceptable for high-grade pharmaceutical applications, necessitating complex and expensive chromatographic separations that are difficult to scale and economically inefficient for bulk production.
The Novel Approach
The patented method introduces a transformative purification sequence that fundamentally alters the impurity profile before the final hydrolysis step. Instead of attempting to crystallize the impurity away, the process chemically differentiates the crude mixture. By treating the crude 16α-hydroxyprednisolone acetate with an aqueous hypochlorite solution in the presence of an organic acid, the process selectively targets the impurity matrix. Following this reaction, a water precipitation step effectively isolates the purified acetate intermediate, leaving the modified impurities in the mother liquor. 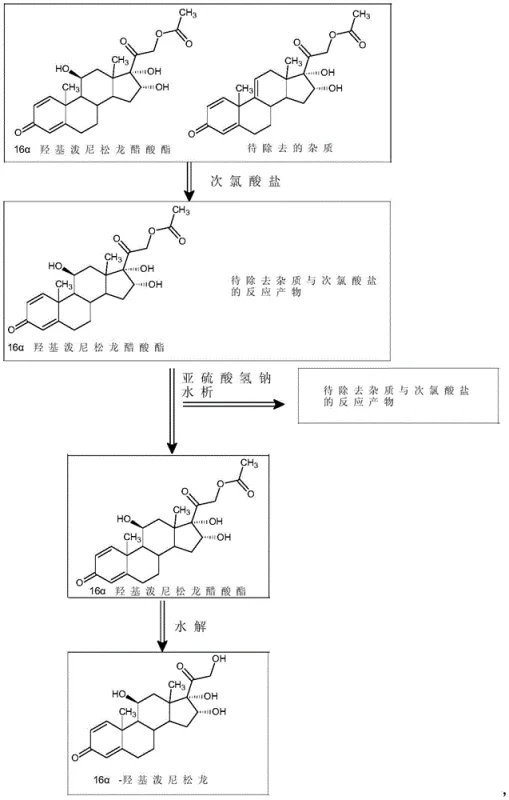
This strategic intervention allows for the removal of Impurity H to levels below 0.02%, pushing the final assay to greater than 99.5%. The subsequent hydrolysis of this highly purified acetate intermediate yields the final 16α-hydroxyprednisolone with exceptional quality. This approach bypasses the need for resource-intensive chromatography, offering a streamlined, scalable, and economically superior route for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Selective Hypochlorite Oxidation
The efficacy of this purification method relies on the differential reactivity of the steroid nucleus and its substituents under mild oxidative conditions. When the crude acetate is exposed to hypochlorite ions (ClO⁻) at controlled temperatures between 15°C and 40°C, specific functional groups on the impurity molecules undergo oxidation or chlorination that the main product resists, or vice versa, altering their polarity and solubility characteristics. The addition of organic acids such as formic or acetic acid helps modulate the pH and reactivity of the hypochlorite species, ensuring the reaction remains selective and does not degrade the valuable steroid skeleton.
Following the oxidative treatment, the addition of sodium bisulfite serves a dual purpose: it quenches excess oxidant to prevent over-reaction, and it facilitates the subsequent phase separation. The critical step of 'water analysis' or precipitation exploits the changed solubility profile of the purified acetate. While the target molecule precipitates efficiently upon the addition of water to the concentrated organic phase, the oxidized impurity derivatives remain soluble in the aqueous-organic mixture. This physical separation mechanism is far more robust than thermal crystallization, providing a consistent barrier against impurity carryover into the final hydrolysis stage.
How to Synthesize 16α-Hydroxyprednisolone Efficiently
The synthesis protocol outlined in the patent provides a clear, three-stage operational framework designed for industrial reproducibility. The process begins with the dissolution of the crude acetate in a binary solvent system, followed by the critical hypochlorite treatment and purification, and concludes with a low-temperature hydrolysis. Detailed standardized synthesis steps are provided in the guide below to ensure operators can replicate the high-purity results consistently.
- Dissolve crude 16α-hydroxyprednisolone acetate in a dichloromethane/alcohol mixture, add organic acid, and treat with aqueous hypochlorite at 15-40°C.
- Quench with sodium bisulfite, concentrate the solvent, and perform water precipitation to filter out modified impurities.
- Hydrolyze the purified acetate intermediate using sodium sulfite in organic solvent at -10 to 10°C to yield the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this purification technology translates directly into enhanced operational stability and reduced risk. By eliminating the reliance on difficult-to-remove impurities, manufacturers can guarantee a consistent supply of high-quality material without the batch-to-batch variability often associated with marginal purification processes. This reliability is crucial for maintaining uninterrupted API production schedules and meeting regulatory compliance standards without costly delays.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic purification steps significantly lowers the operational expenditure associated with producing high-purity steroids. By utilizing inexpensive and widely available reagents like sodium hypochlorite and sodium sulfite, the process avoids the high costs of specialty resins and solvents. Furthermore, the simplified workflow reduces labor hours and energy consumption, leading to substantial cost savings in the overall manufacturing budget.
- Enhanced Supply Chain Reliability: The robustness of this method against impurity fluctuations ensures a steady output of qualified material. Since the purification is chemical rather than purely physical, it is less sensitive to minor variations in the crude feedstock quality. This resilience minimizes the risk of batch rejection and ensures that reducing lead time for high-purity pharmaceutical intermediates becomes a achievable reality for downstream partners.
- Scalability and Environmental Compliance: The reaction conditions are mild, operating near ambient temperatures for the purification step and requiring only moderate cooling for hydrolysis. This reduces the load on industrial chilling systems and simplifies reactor requirements. Additionally, the use of common inorganic salts and the avoidance of heavy metal catalysts streamline waste treatment protocols, facilitating easier environmental compliance and safer large-scale operations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this advanced purification route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on how this method outperforms legacy techniques.
Q: Why is Impurity H difficult to remove in conventional 16α-hydroxyprednisolone synthesis?
A: Impurity H possesses a chemical structure highly similar to the target molecule, making standard recrystallization ineffective. Conventional methods often leave impurity levels around 0.90%, failing to meet the strict <0.2% market requirement.
Q: How does the hypochlorite treatment improve purity?
A: The novel method utilizes a selective reaction where aqueous hypochlorite modifies the impurity profile or the matrix in a way that alters solubility. Subsequent water precipitation effectively separates these modified species, driving purity above 99.5%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process operates under mild conditions (15-40°C for purification, -10 to 10°C for hydrolysis) and uses commercially available reagents like sodium hypochlorite and sodium sulfite, ensuring excellent scalability and safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16α-Hydroxyprednisolone Supplier
At NINGBO INNO PHARMCHEM, we understand that the quality of your final API depends heavily on the purity of your starting materials. Our technical team has extensively analyzed advanced purification routes like the one described in CN112125943A to ensure our production capabilities align with the highest global standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. Our rigorous QC labs are equipped to detect and quantify trace impurities, guaranteeing that every batch of 16α-hydroxyprednisolone we supply meets the >99.5% purity benchmark.
We invite you to collaborate with us to optimize your supply chain for corticosteroid intermediates. By leveraging our technical expertise and advanced manufacturing capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to quality can drive efficiency in your drug development pipeline.
