Breakthrough (R)-ω-Transaminase Mutants Enable Efficient Commercial Scale-up of Complex Pharmaceutical Intermediates
Breakthrough (R)-ω-Transaminase Mutants Enable Efficient Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking robust biocatalytic solutions to overcome the limitations of traditional chemical synthesis, particularly for chiral amines used in diabetes therapeutics. Patent CN111411094B, filed in 2021, introduces a groundbreaking (R)-ω-transaminase mutant that addresses critical bottlenecks in the production of sitagliptin intermediates. This technology leverages advanced protein engineering to create a biocatalyst with significantly enhanced enzymatic activity, substrate tolerance, and stereoselectivity compared to existing commercial enzymes. By utilizing gene mining techniques on Aspergillus terreus derived sequences, the inventors have developed a six-point mutant capable of driving reactions at high substrate concentrations up to 900 mM. This development represents a pivotal shift away from the monopolized technologies previously held by major multinational corporations, offering a viable pathway for generic manufacturers to access high-efficiency biocatalytic routes. The ability to achieve conversion rates as high as 95.4% underlines the potential for this technology to redefine cost structures and supply chain reliability in the manufacturing of DPP-4 inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the biocatalytic preparation of sitagliptin has been dominated by specific proprietary enzyme variants that, while effective, often operate under restrictive conditions that limit industrial scalability. Prior art, such as the process described in Chinese patent CN108586346A, utilizes transaminases that require relatively low substrate concentrations, typically around 367 mM, to maintain acceptable conversion rates. This limitation necessitates larger reactor volumes to produce the same amount of product, thereby increasing capital expenditure and operational costs related to solvent handling and downstream processing. Furthermore, the reliance on a single source of enzyme technology creates supply chain vulnerabilities, where licensing fees and restricted access can hinder the competitiveness of generic drug manufacturers. Conventional wild-type transaminases often lack the necessary stability and activity to handle the steric bulk of complex ketone substrates efficiently, leading to incomplete reactions and difficult purification challenges that compromise the overall yield and purity of the final active pharmaceutical ingredient.
The Novel Approach
The innovative strategy outlined in CN111411094B overcomes these hurdles through a meticulously engineered (R)-ω-transaminase mutant designed specifically for high-load biocatalysis. Unlike previous iterations, this novel enzyme variant exhibits remarkable tolerance to substrate concentrations ranging from 600 mM to 900 mM, effectively more than doubling the throughput capacity per batch compared to older methods. The core of this advancement lies in the specific biocatalytic conversion of the precursor ketone analog, 1-(3-oxopyrrolidin-1-yl)-4-(2,4,5-trifluorophenyl)-1,3-butanedione, into the valuable sitagliptin intermediate. 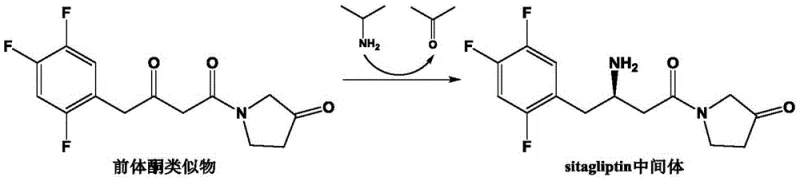 As illustrated in the reaction scheme, the process utilizes isopropylamine as an amino donor in the presence of pyridoxal phosphate (PLP), driving the equilibrium towards the desired chiral amine product with exceptional efficiency. This approach not only simplifies the synthetic route by eliminating the need for multiple protection and deprotection steps often seen in purely chemical syntheses but also ensures a greener manufacturing profile by operating under mild aqueous conditions.
As illustrated in the reaction scheme, the process utilizes isopropylamine as an amino donor in the presence of pyridoxal phosphate (PLP), driving the equilibrium towards the desired chiral amine product with exceptional efficiency. This approach not only simplifies the synthetic route by eliminating the need for multiple protection and deprotection steps often seen in purely chemical syntheses but also ensures a greener manufacturing profile by operating under mild aqueous conditions.
Mechanistic Insights into Multi-Site Directed Mutagenesis
The superior performance of this biocatalyst is rooted in a sophisticated multi-site directed mutagenesis strategy that optimizes the enzyme's active site architecture. The final optimized variant, designated as AtTA6, incorporates six specific amino acid substitutions: L182M, R79A, Q51S, V149D, L235E, and G216A. Each mutation plays a distinct role in enhancing the enzyme's interaction with the bulky trifluorophenyl substrate. For instance, the mutation at position 182 from Leucine to Methionine likely alters the hydrophobicity and flexibility of the substrate binding pocket, allowing for better accommodation of the large aromatic group. Similarly, the introduction of charged residues like Aspartic Acid at position 149 and Glutamic Acid at position 235 may facilitate improved electrostatic interactions or stabilize the transition state during the amino transfer mechanism. This rational design approach, guided by high-throughput screening of hundreds of recombinant strains, ensures that the enzyme maintains its structural integrity while maximizing catalytic turnover numbers even at elevated temperatures up to 50°C.
Impurity control is another critical aspect where this mechanistic understanding translates to commercial value. The engineered enzyme demonstrates an enantiomeric excess (e.e.) value exceeding 99% for the (R)-configuration, which is paramount for meeting stringent regulatory requirements for chiral drugs. High stereoselectivity minimizes the formation of the unwanted (S)-enantiomer, which can be difficult to separate and may pose toxicity risks. By ensuring that the biocatalytic step produces a highly pure intermediate directly, the downstream purification burden is significantly reduced. This reduces the need for extensive chromatographic separations or recrystallization steps, which are often the most costly and time-consuming parts of API manufacturing. The robustness of the mutant also implies a lower likelihood of side reactions or enzyme degradation products contaminating the final mixture, further streamlining the path to a high-purity pharmaceutical intermediate suitable for global markets.
How to Synthesize Sitagliptin Intermediate Efficiently
Implementing this biocatalytic route requires a systematic approach to fermentation and reaction engineering to fully leverage the mutant's capabilities. The process begins with the cultivation of recombinant E. coli strains harboring the optimized gene sequence, followed by induction to express the transaminase enzyme. The subsequent biotransformation step is conducted in a buffered system with controlled pH and temperature to maximize enzyme stability and reaction kinetics. Detailed standard operating procedures regarding the specific ratios of co-factors, amino donors, and substrate loading are essential for reproducibility at scale. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Ferment the recombinant E. coli BL21(DE3)/pET28b/AtTA6 strain in LB medium with kanamycin induction to produce the wet cell biomass containing the active enzyme.
- Perform the biocatalytic reaction using the wet cells, precursor ketone analog substrate (600-900 mM), isopropylamine as amino donor, and PLP cofactor at 50°C and pH 9.0.
- Isolate the product (R)-1-[3-amino-4-(2,4,5-trifluorophenyl)butyryl]pyrrole-3-ketone via centrifugation and subsequent chemical processing including hydrolysis and condensation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel transaminase technology offers compelling strategic advantages that extend beyond simple technical metrics. The ability to run reactions at significantly higher substrate concentrations directly correlates to reduced solvent usage and smaller reactor footprints, which are key drivers in lowering the cost of goods sold (COGS). By shifting from low-concentration processes to high-density biocatalysis, manufacturers can achieve substantial cost savings in raw material procurement and waste disposal without compromising on yield or quality. This efficiency gain is particularly relevant in the competitive landscape of generic diabetes medications, where margin pressure is constant and operational excellence is a primary differentiator for maintaining profitability.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in solvent volume required for high-concentration reactions lead to a drastically simplified cost structure. Since the process relies on a renewable biocatalyst rather than precious metals, there is no need for costly metal scavenging steps or validation for residual metal limits, which further reduces analytical and processing expenses. The high conversion rate of over 95% ensures that raw material utilization is maximized, minimizing the financial loss associated with unreacted starting materials and reducing the load on recovery systems.
- Enhanced Supply Chain Reliability: Diversifying the enzyme source away from monopolized providers mitigates the risk of supply disruptions and excessive licensing fees. The use of a genetically engineered strain that can be produced via standard fermentation techniques ensures a consistent and scalable supply of the biocatalyst. This autonomy allows pharmaceutical companies to secure their supply chains against external market fluctuations and geopolitical tensions that might affect the availability of specialized reagents, ensuring continuous production of critical diabetes therapies.
- Scalability and Environmental Compliance: The robust nature of the mutant enzyme at elevated temperatures and high substrate loads facilitates easier scale-up from laboratory to commercial production scales. The aqueous nature of the biocatalytic process aligns well with green chemistry principles, reducing the generation of hazardous organic waste compared to traditional asymmetric hydrogenation or chemical resolution methods. This environmental compliance not only lowers waste treatment costs but also enhances the corporate sustainability profile, which is increasingly important for meeting the ESG (Environmental, Social, and Governance) criteria of modern pharmaceutical buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this transaminase technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of what partners can expect when integrating this route into their manufacturing portfolio. Understanding these specifics helps in making informed decisions about process adoption and resource allocation.
Q: What is the maximum substrate concentration achievable with this new transaminase mutant?
A: According to patent CN111411094B, the optimized six-point mutant (AtTA6) can effectively operate at substrate concentrations ranging from 600 mM to 900 mM, with optimal conversion rates observed at 800 mM.
Q: How does the stereoselectivity of this mutant compare to wild-type enzymes?
A: The engineered mutant demonstrates exceptional stereoselectivity, consistently achieving an enantiomeric excess (e.e.) value of greater than 99% for the (R)-configuration, which is critical for pharmaceutical purity standards.
Q: What specific amino acid mutations contribute to the enhanced activity?
A: The high-activity variant contains six specific point mutations: L182M, R79A, Q51S, V149D, L235E, and G216A, which collectively improve substrate binding pocket accommodation and catalytic turnover.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalytic technologies like the (R)-ω-transaminase mutant described in CN111411094B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are successfully translated into robust industrial processes. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of sitagliptin intermediate meets the highest global regulatory standards. We are committed to leveraging our technical expertise to help clients navigate the complexities of enzyme immobilization, cofactor regeneration, and downstream processing to achieve optimal economic outcomes.
We invite forward-thinking pharmaceutical companies to collaborate with us to explore the full commercial potential of this high-efficiency synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments that demonstrate how our integrated manufacturing capabilities can accelerate your time-to-market while reducing overall production costs for this critical diabetes therapeutic intermediate.
