Advanced Manufacturing of 2,4,5-Trifluorophenylacetic Acid for High-Volume API Production
The pharmaceutical industry's relentless pursuit of efficient, safe, and scalable synthetic routes for critical intermediates has led to significant advancements in process chemistry, particularly for blockbuster drugs like Sitagliptin. Patent CN101244994B, published in early 2013, introduces a novel and robust methodology for the preparation of 2,4,5-trifluorophenylacetic acid, a pivotal building block in the synthesis of DPP-IV inhibitors. This technology represents a paradigm shift from traditional, hazardous methods to a streamlined process utilizing Friedel-Crafts acylation followed by a Willgerodt-Kindler reaction. For R&D directors and procurement specialists evaluating supply chain resilience, this patent offers a compelling solution that balances high chemical purity with operational safety. The disclosed method achieves a final product content exceeding 99.9%, demonstrating exceptional selectivity and efficiency that addresses the rigorous quality standards required for modern active pharmaceutical ingredient (API) manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4,5-trifluorophenylacetic acid has been plagued by significant technical and safety hurdles that hinder large-scale industrial adoption. Early approaches, such as those described in US20040068141, relied on the nucleophilic substitution of 2,4,5-trifluorobromobenzene with diethyl malonate under harsh alkaline conditions, a process often characterized by difficult purification steps and moderate yields. Another pathway, detailed in US20040077901, utilized a Grignard reaction followed by oxidation with expensive ruthenium catalysts and sodium periodate, imposing strict anhydrous requirements that complicate reactor operations and increase capital expenditure. Perhaps most critically, prior art like CN1749232 employed a chloromethylation and cyanation sequence involving sodium cyanide, a highly toxic reagent that poses severe safety risks to personnel and creates complex wastewater treatment challenges for manufacturing facilities.
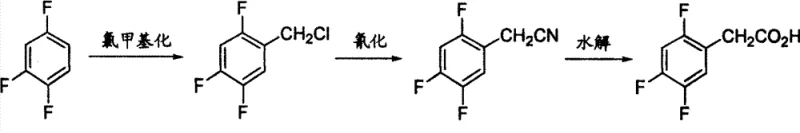
The Novel Approach
In stark contrast to these legacy methods, the innovative process outlined in CN101244994B leverages the versatility of the Willgerodt-Kindler reaction to construct the acetic acid side chain directly from a ketone precursor. By starting with readily available 1,2,4-trifluorobenzene, the process first installs an acetyl group via Friedel-Crafts acylation, followed by a sulfur-mediated conversion to a thioamide, which is subsequently hydrolyzed to the target acid. This route completely avoids the use of toxic cyanides and expensive transition metal oxidants, relying instead on abundant and cost-effective reagents like sulfur and dimethylamine salts. The mild reaction conditions and straightforward workup procedures not only enhance operator safety but also simplify the engineering controls required for commercial scale-up, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their production portfolios.
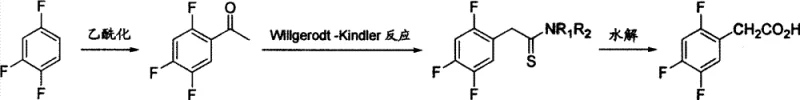
Mechanistic Insights into Friedel-Crafts and Willgerodt-Kindler Cascade
The core of this synthetic strategy lies in the precise execution of two distinct chemical transformations that work in tandem to build molecular complexity with high fidelity. The initial step involves a classic Friedel-Crafts acylation where 1,2,4-trifluorobenzene reacts with acetyl chloride in the presence of a Lewis acid catalyst, typically aluminum trichloride. The electron-withdrawing nature of the fluorine atoms directs the electrophilic attack to the desired position, yielding 2,4,5-trifluoroacetophenone with high regioselectivity. Following isolation, the ketone undergoes the Willgerodt-Kindler reaction, a powerful transformation that migrates the carbonyl group to the terminal position of the side chain while simultaneously introducing nitrogen and sulfur functionality. This proceeds through the formation of a polysulfide intermediate which rearranges to form the thioamide, effectively extending the carbon chain by one unit while preserving the aromatic substitution pattern essential for biological activity.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based oxidation or nucleophilic substitution pathways. The stepwise nature of the synthesis allows for the removal of by-products at the thioamide stage, preventing the carryover of halogenated impurities or heavy metal residues into the final API. The hydrolysis of the thioamide is conducted under controlled alkaline or acidic conditions, ensuring complete conversion to the carboxylic acid without degrading the sensitive trifluoro-substituted aromatic ring. Furthermore, the final recrystallization step, often performed using ethanol-water mixtures, serves as a potent purification tool that removes trace organic impurities, ensuring the final material meets the stringent purity specifications demanded by regulatory bodies for diabetes medication production.
How to Synthesize 2,4,5-Trifluorophenylacetic Acid Efficiently
Implementing this synthesis requires careful attention to stoichiometry and temperature control to maximize yield and minimize side reactions. The process begins with the acylation step, where maintaining the temperature below 40°C during reagent addition prevents poly-acylation, followed by heating to drive the reaction to completion. The subsequent Willgerodt-Kindler step utilizes polar aprotic solvents like DMF to solubilize the sulfur and amine components, requiring sustained heating to facilitate the rearrangement. Finally, the hydrolysis step demands precise pH adjustment to precipitate the product efficiently. For a detailed breakdown of the specific molar ratios, solvent volumes, and thermal profiles required for GMP-compliant manufacturing, please refer to the standardized protocol below.
- Perform Friedel-Crafts acetylation of 1,2,4-trifluorobenzene using acetyl chloride and aluminum trichloride to obtain 2,4,5-trifluoroacetophenone.
- Execute the Willgerodt-Kindler reaction by heating the ketone with sulfur and dimethylamine hydrochloride in DMF to form the thioamide intermediate.
- Hydrolyze the thioamide intermediate under alkaline conditions followed by acidification and recrystallization to yield the final acid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits that extend beyond simple chemical yield. By eliminating the dependency on scarce and costly transition metal catalysts like ruthenium, manufacturers can significantly reduce raw material volatility and exposure to geopolitical supply risks associated with precious metals. Furthermore, the removal of highly toxic cyanide reagents simplifies the regulatory compliance landscape, reducing the overhead costs related to hazardous waste disposal and specialized safety training. This streamlined approach facilitates a more robust and continuous supply of high-purity intermediates, ensuring that downstream API production schedules remain uninterrupted even during periods of market fluctuation.
- Cost Reduction in Manufacturing: The replacement of expensive oxidants and catalysts with commodity chemicals like sulfur and acetyl chloride drives down the direct cost of goods sold. Additionally, the simplified workup procedures reduce solvent consumption and energy usage, leading to substantial operational savings without compromising product quality.
- Enhanced Supply Chain Reliability: Utilizing 1,2,4-trifluorobenzene as a starting material leverages a well-established global supply chain for fluorinated aromatics, mitigating the risk of raw material shortages. The robustness of the reaction conditions also means that production is less susceptible to minor variations in utility availability, ensuring consistent output.
- Scalability and Environmental Compliance: The absence of toxic cyanide and heavy metals makes this process inherently greener and easier to scale from pilot plant to multi-ton production. This alignment with green chemistry principles reduces the environmental footprint of the manufacturing site and eases the burden on wastewater treatment facilities.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding safety, purity, and scalability, drawing directly from the experimental data and beneficial effects reported in the patent literature. These insights are intended to provide clarity on how this method compares to legacy technologies and why it represents a superior choice for modern pharmaceutical manufacturing.
Q: Why is the Willgerodt-Kindler route preferred over cyanide-based methods for this intermediate?
A: The Willgerodt-Kindler route eliminates the use of highly toxic sodium cyanide, significantly reducing environmental hazards and safety risks associated with industrial handling and waste disposal.
Q: What is the expected purity of 2,4,5-trifluorophenylacetic acid using this method?
A: According to patent data, the final product content can exceed 99.9% after recrystallization, meeting stringent specifications for API synthesis.
Q: Does this process require expensive transition metal catalysts?
A: No, unlike alternative Grignard-based routes that require Ruthenium catalysts, this method utilizes common Lewis acids and sulfur, drastically lowering raw material costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,5-Trifluorophenylacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of life-saving medications depends on the reliability and quality of the supply chain. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the dynamic demands of the global pharmaceutical market. We are committed to delivering 2,4,5-trifluorophenylacetic acid with stringent purity specifications, supported by our rigorous QC labs that employ state-of-the-art analytical techniques to verify every batch. By leveraging advanced synthetic methodologies like the Willgerodt-Kindler reaction, we provide our partners with a secure source of critical intermediates that align with both economic and environmental goals.
We invite you to collaborate with us to optimize your supply chain for Sitagliptin and related therapeutic agents. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
