Revolutionizing Pharmaceutical Intermediate Production with Engineered Monoamine Oxidase Technology
Revolutionizing Pharmaceutical Intermediate Production with Engineered Monoamine Oxidase Technology
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for synthesizing complex active pharmaceutical ingredients (APIs) and their precursors. Patent CN115109760A introduces a groundbreaking advancement in this domain by disclosing a novel monoamine oxidase and its specific application in the preparation of critical pharmaceutical intermediates, such as those required for Boceprevir and Nirmatrelvir. This technology addresses long-standing challenges in biocatalysis, specifically targeting the oxidative transformation of bicyclic amines with unprecedented efficiency. By leveraging genetically engineered enzymes with optimized amino acid sequences (SEQ ID NO: 2 or SEQ ID NO: 4), the disclosed method achieves superior reaction kinetics and conversion rates compared to traditional wild-type enzymes. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes, reduce reaction times, and enhance the overall purity profile of high-value antiviral intermediates. The integration of this biocatalytic step into existing manufacturing workflows offers a compelling value proposition for reducing both environmental impact and production costs.
![Chemical structure of the key pharmaceutical intermediate (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester](/insights/img/monoamine-oxidase-pharma-intermediate-supplier-20260307145631-03.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
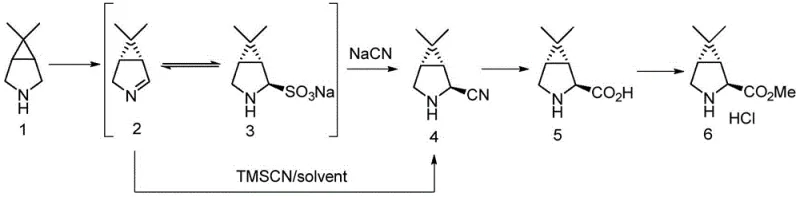
Prior art methods for synthesizing key intermediates like (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic acid methyl ester often relied on wild-type monoamine oxidases derived from sources such as Aspergillus niger or Aspergillus oryzae. These conventional biocatalysts suffered from inherent limitations, primarily characterized by low enzymatic activity and suboptimal reaction efficiency. In many documented cases, the conversion of the amine substrate to the requisite imine intermediate was incomplete, with wild-type enzymes achieving conversion rates as low as 20% to 40%. This inefficiency necessitated larger reactor volumes, extended reaction times, and complex downstream purification processes to remove unreacted starting materials and byproducts. Furthermore, the instability of wild-type enzymes under industrial conditions often led to batch-to-batch variability, posing significant risks for supply chain consistency and cost predictability in large-scale API manufacturing.
The Novel Approach
The innovative approach detailed in the patent overcomes these historical bottlenecks through the deployment of specifically engineered monoamine oxidase variants. By utilizing recombinant DNA technology to express enzymes with sequences defined in SEQ ID NO: 2 or SEQ ID NO: 4, the process achieves a dramatic improvement in catalytic performance. Experimental data indicates that these novel enzymes can drive the oxidation reaction to near-completion, with conversion rates reaching up to 99% under optimized conditions. This leap in efficiency allows for a streamlined synthetic sequence where the enzymatic oxidation is seamlessly coupled with subsequent chemical transformations, such as bisulfite addition and cyanation. The result is a robust, high-yielding process that minimizes waste and maximizes throughput. For procurement managers, this translates to a more reliable supply of high-purity intermediates, while the reduced reaction complexity offers substantial opportunities for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Monoamine Oxidase-Catalyzed Oxidation
The core of this technological breakthrough lies in the precise mechanistic action of the engineered monoamine oxidase on the bicyclic amine substrate. The enzyme facilitates an oxidative deamination reaction, converting the saturated amine (Compound 1) into the corresponding imine (Compound 2) with high stereoselectivity. This transformation is dependent on the presence of molecular oxygen and essential cofactors such as FAD (Flavin Adenine Dinucleotide). A critical aspect of the mechanism involves the management of hydrogen peroxide, a byproduct of the oxidation that can otherwise inactivate the enzyme. The process incorporates catalase to decompose hydrogen peroxide into water and oxygen, thereby maintaining enzyme stability and prolonging the catalytic cycle. This synergistic enzyme system ensures that the reaction proceeds smoothly without the accumulation of deleterious oxidants that could compromise product quality or enzyme longevity.
![Enzymatic oxidation mechanism converting 6,6-dimethyl-3-azabicyclo[3.1.0]hexane to the imine intermediate](/insights/img/monoamine-oxidase-pharma-intermediate-supplier-20260307145631-06.png)
Furthermore, the stereochemical integrity of the product is rigorously maintained throughout the catalytic cycle. The engineered active site of the monoamine oxidase is tailored to recognize the specific spatial configuration of the 6,6-dimethyl-3-azabicyclo[3.1.0]hexane substrate, ensuring that only the desired (1R,5S) enantiomer of the imine is produced. This high level of chiral induction is paramount for the synthesis of antiviral drugs like Boceprevir, where the biological activity is strictly dependent on the absolute configuration of the molecule. By eliminating the formation of unwanted stereoisomers at the earliest stage of the synthesis, the process significantly simplifies downstream purification. This mechanistic precision reduces the burden on analytical laboratories and ensures that the final API meets the stringent regulatory standards required for clinical use, thereby mitigating the risk of costly batch rejections.
How to Synthesize (1R,2S,5S)-6,6-dimethyl-3-azabicyclo[3.1.0]hexane-2-carboxylic Acid Methyl Ester Efficiently
The practical implementation of this technology involves a multi-step sequence that begins with the fermentation of recombinant host cells to produce the biocatalyst, followed by the enzymatic reaction and subsequent chemical derivatization. The process is designed to be operationally simple, utilizing standard fermentation equipment and common chemical reagents. The initial enzymatic step is conducted under mild aqueous conditions, typically at a controlled pH of roughly 7.3 and temperatures around 23°C, which favors enzyme stability. Following the biocatalytic oxidation, the reaction mixture undergoes a series of chemical transformations including nucleophilic addition of bisulfite, displacement with cyanide, acidic hydrolysis, and final esterification. This integrated chemo-enzymatic strategy combines the selectivity of biology with the versatility of organic chemistry to deliver the target intermediate in high yield and purity.
- Oxidize 6,6-dimethyl-3-azabicyclo[3.1.0]hexane using engineered monoamine oxidase (SEQ ID NO: 2 or 4) in the presence of oxygen and catalase to form the imine intermediate.
- React the resulting imine with sodium bisulfite to generate the sulfonate adduct, followed by treatment with cyanide (NaCN or TMSCN) to yield the nitrile compound.
- Hydrolyze the nitrile to the corresponding carboxylic acid using hydrochloric acid, followed by esterification with methanol and thionyl chloride to obtain the final methyl ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this engineered enzyme technology offers distinct strategic advantages over traditional synthetic routes. The primary benefit lies in the drastic simplification of the manufacturing process, which directly correlates to improved operational efficiency and reduced overhead. By achieving near-quantitative conversion in the enzymatic step, the need for extensive recycling of unreacted starting materials is eliminated, thereby reducing solvent consumption and energy usage associated with distillation and separation units. This efficiency gain is particularly valuable in the context of volatile raw material markets, as it maximizes the atom economy of the process and ensures that every kilogram of expensive substrate is converted into valuable product.
- Cost Reduction in Manufacturing: The implementation of high-activity monoamine oxidase variants leads to significant cost savings by shortening reaction cycles and increasing reactor throughput. Unlike wild-type enzymes that require prolonged incubation times to achieve modest conversions, the engineered catalysts drive the reaction to completion rapidly, allowing for faster batch turnover. Additionally, the high selectivity of the enzyme minimizes the formation of difficult-to-remove impurities, which reduces the complexity and cost of downstream purification processes. This streamlined workflow eliminates the need for expensive chromatographic separations often required to resolve stereoisomers, resulting in a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The robustness of the recombinant enzyme production system ensures a consistent and reliable supply of the biocatalyst, mitigating the risks associated with sourcing natural enzymes from variable biological sources. The use of standardized E. coli expression hosts allows for scalable fermentation processes that can be easily ramped up to meet surging demand for antiviral intermediates. This reliability is crucial for maintaining uninterrupted production schedules for critical medications. Furthermore, the stability of the enzyme under process conditions reduces the likelihood of batch failures due to catalyst deactivation, providing greater predictability for delivery timelines and inventory planning.
- Scalability and Environmental Compliance: The aqueous nature of the biocatalytic step aligns perfectly with green chemistry principles, significantly reducing the environmental footprint of the manufacturing process. By operating at mild temperatures and neutral pH, the process consumes less energy compared to high-temperature chemical oxidations. The elimination of heavy metal catalysts and the reduction in organic solvent usage contribute to lower waste generation and simpler effluent treatment. This environmental compatibility not only facilitates regulatory compliance but also enhances the sustainability profile of the supply chain, a factor that is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this monoamine oxidase technology. These insights are derived directly from the experimental data and embodiments described in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this biocatalytic route into their existing manufacturing infrastructure.
Q: What distinguishes the engineered monoamine oxidase in CN115109760A from wild-type enzymes?
A: The engineered monoamine oxidase (SEQ ID NO: 2 or 4) demonstrates significantly higher enzymatic activity and conversion rates compared to wild-type enzymes from Aspergillus niger or oryzae, which typically show only 20-40% conversion. The novel variants achieve conversion rates exceeding 98% within reduced reaction times.
Q: How does this biocatalytic route improve impurity control for API intermediates?
A: The high stereoselectivity of the engineered enzyme ensures the formation of the desired (1R,5S) imine intermediate with minimal formation of unwanted stereoisomers. Subsequent chemical steps preserve this chirality, resulting in a final product with high optical purity, crucial for antiviral drug synthesis.
Q: Is this process scalable for commercial manufacturing of Boceprevir intermediates?
A: Yes, the patent outlines a robust fermentation and downstream processing workflow compatible with industrial scale-up. The use of recombinant E. coli expression systems and stable enzyme formulations supports large-volume production required for global supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boceprevir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced biocatalysis in the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the engineered monoamine oxidase process can be seamlessly transitioned from the laboratory to the plant. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to maintain stringent purity specifications for high-value antiviral intermediates. We are committed to delivering products that meet the highest quality standards, supporting our partners in bringing life-saving medications to market faster and more efficiently.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in enzymatic synthesis can drive value and reliability for your organization.
