Advanced Biocatalytic Synthesis of Chiral Azabicyclo Compounds for Hepatitis C Therapeutics
The pharmaceutical landscape for Hepatitis C Virus (HCV) treatment has been significantly shaped by protease inhibitors such as Boceprevir and Telaprevir, which rely heavily on complex chiral azabicyclic scaffolds for their biological efficacy. As depicted in the structural overview, these molecules contain critical stereocenters that dictate their pharmacological activity, making the synthesis of their key intermediates, specifically Fragment A and Fragment B, a paramount challenge for process chemists. 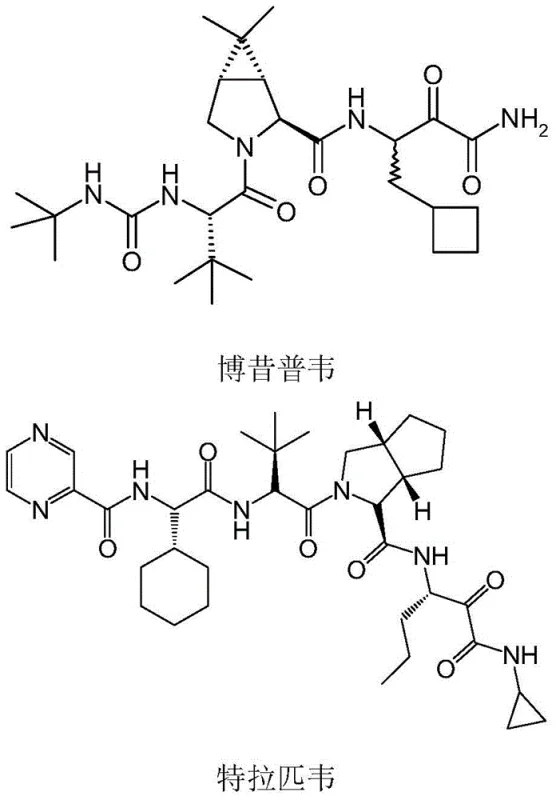 Recent advancements in biocatalysis have offered a transformative alternative to traditional synthetic methods, as detailed in patent CN105624128B, which discloses a novel immobilized monoamine oxidase (MAO) system. This technology addresses the longstanding issues of enzyme instability and recovery, providing a robust platform for the asymmetric synthesis of (1S,2S,5R)-aza-bicyclic compounds. By leveraging a specialized epoxy resin carrier derivatized with iminodiacetic acid and nickel, the invention achieves firm enzyme binding with minimal activity loss, thereby enabling a greener and more economically viable production route for these high-value pharmaceutical intermediates.
Recent advancements in biocatalysis have offered a transformative alternative to traditional synthetic methods, as detailed in patent CN105624128B, which discloses a novel immobilized monoamine oxidase (MAO) system. This technology addresses the longstanding issues of enzyme instability and recovery, providing a robust platform for the asymmetric synthesis of (1S,2S,5R)-aza-bicyclic compounds. By leveraging a specialized epoxy resin carrier derivatized with iminodiacetic acid and nickel, the invention achieves firm enzyme binding with minimal activity loss, thereby enabling a greener and more economically viable production route for these high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key intermediates like Fragment A has relied on multi-step chemical routes that are fraught with significant operational and environmental hazards. For instance, prior art routes such as those disclosed in WO2007075790 utilize hazardous reagents including silver nitrate, potassium cyanide, and hydrochloric acid for cyano addition, which not only inflate raw material costs but also create severe difficulties in post-processing and waste treatment. Other approaches, such as those found in the Journal of Medicinal Chemistry, employ lithium aluminum hydride and palladium carbon for reduction steps, necessitating harsh reaction conditions and cumbersome workup procedures that are ill-suited for large-scale manufacturing. Furthermore, routes requiring secondary reduction reactions with alane or borohydrides often demand strict temperature control and extended reaction times, leading to low safety profiles and poor atom economy. These chemical methodologies frequently involve the use of toxic heavy metals and volatile organic solvents, resulting in complex impurity profiles that require extensive purification, such as silica gel column chromatography, which is notoriously difficult to scale up for industrial production.
The Novel Approach
In stark contrast, the biocatalytic strategy outlined in the present invention utilizes a highly engineered immobilized monoamine oxidase to facilitate a direct oxidative addition reaction under mild aqueous conditions. This novel approach eliminates the need for protecting groups and harsh chemical reducing agents, streamlining the synthetic sequence into a more efficient cascade process. The use of an immobilized enzyme system allows for the reaction to proceed at ambient temperatures (20-35°C) and neutral pH levels (6.0-8.0), drastically reducing energy consumption and equipment corrosion risks associated with acidic or basic chemical catalysis. Moreover, the immobilization matrix ensures that the biocatalyst can be easily separated from the product stream via simple filtration, enabling its reuse across multiple production batches without significant loss of catalytic potency. This shift from stoichiometric chemical reagents to a recyclable biocatalyst represents a paradigm shift in process chemistry, offering a pathway to high-purity chiral intermediates with a substantially reduced environmental footprint and simplified regulatory compliance regarding heavy metal residues.
Mechanistic Insights into Immobilized MAO-Catalyzed Oxidation-Addition
The core of this technological breakthrough lies in the precise engineering of the biocatalyst and the mechanistic pathway it facilitates. The process begins with the preparation of a stable support matrix, where commercial epoxy resin is sequentially derivatized with iminodiacetic acid (IDA) and nickel sulfate to create a chelating carrier that firmly anchors the recombinant monoamine oxidase. This specific immobilization chemistry prevents enzyme leaching while maintaining the conformational flexibility required for substrate binding. In the catalytic cycle, the prochiral azabicyclic substrate, represented generally by Formula I, undergoes enantioselective oxidation by the immobilized MAO in the presence of an oxidant and catalase to generate a reactive chiral imine intermediate. 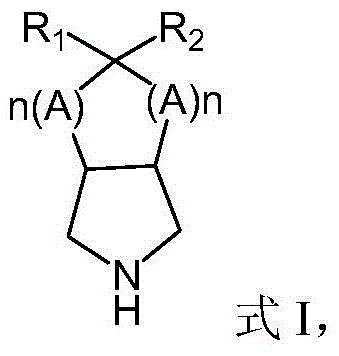 This transient imine species is then immediately trapped by a nucleophile (MR), such as bisulfite or cyanide, in a subsequent addition step to yield the optically pure (1S,2S,5R)-aza-bicyclic product, shown as Formula II.
This transient imine species is then immediately trapped by a nucleophile (MR), such as bisulfite or cyanide, in a subsequent addition step to yield the optically pure (1S,2S,5R)-aza-bicyclic product, shown as Formula II. 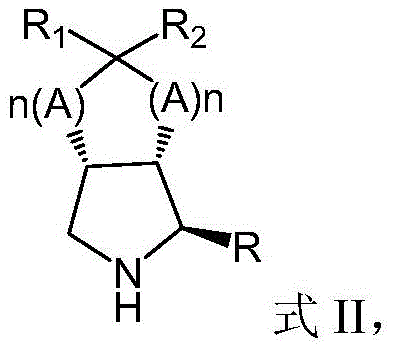 The presence of catalase is crucial in this system to decompose hydrogen peroxide generated during the oxidation step, preventing oxidative damage to the enzyme and ensuring sustained catalytic turnover. The stereochemical outcome is dictated by the chiral environment of the enzyme's active site, which discriminates between the pro-R and pro-S hydrogen atoms of the substrate, ensuring the formation of the desired stereoisomer with high enantiomeric excess without the need for downstream resolution steps.
The presence of catalase is crucial in this system to decompose hydrogen peroxide generated during the oxidation step, preventing oxidative damage to the enzyme and ensuring sustained catalytic turnover. The stereochemical outcome is dictated by the chiral environment of the enzyme's active site, which discriminates between the pro-R and pro-S hydrogen atoms of the substrate, ensuring the formation of the desired stereoisomer with high enantiomeric excess without the need for downstream resolution steps.
Impurity control in this enzymatic process is inherently superior to chemical methods due to the high specificity of the enzyme. Unlike chemical oxidants which may attack various functional groups indiscriminately, the monoamine oxidase targets only the specific amine moiety, minimizing the formation of side products such as over-oxidized species or ring-opened byproducts. The mild reaction conditions further suppress thermal degradation pathways that often plague high-temperature chemical syntheses. Additionally, the immobilization matrix itself acts as a physical barrier that can exclude larger molecular impurities or aggregates from accessing the active site, thereby enhancing the overall purity of the crude reaction mixture. This high level of selectivity simplifies the downstream purification process, often allowing for direct crystallization or simple extraction, which significantly improves the overall yield and reduces the solvent intensity of the manufacturing process. The stability of the immobilized enzyme against pH fluctuations and organic co-solvents also ensures consistent performance even when substrate solubility requires the addition of minor amounts of water-miscible organic solvents.
How to Synthesize Chiral Azabicyclo Compounds Efficiently
The practical implementation of this biocatalytic route involves a streamlined workflow designed for scalability and reproducibility in a GMP environment. The process initiates with the activation of the epoxy resin carrier followed by the careful loading of the recombinant enzyme under controlled pH and temperature conditions to maximize binding capacity. Once the biocatalyst is prepared, the oxidation-addition reaction is conducted in a buffered aqueous system where the substrate concentration and oxidant feed rate are optimized to maintain steady-state kinetics and prevent substrate inhibition. Detailed standard operating procedures for the preparation of the immobilized carrier, the expression of the recombinant enzyme, and the execution of the catalytic reaction are critical for ensuring batch-to-batch consistency.
- Preparation of the immobilized carrier by derivatizing epoxy resin with iminodiacetic acid (IDA) and nickel sulfate.
- Immobilization of recombinant monoamine oxidase onto the carrier in phosphate buffer at room temperature.
- Catalytic oxidation-addition reaction of prochiral azabicyclic compounds with MR in aqueous solution using the immobilized enzyme.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this immobilized enzyme technology presents a compelling value proposition centered on cost stability and operational resilience. The transition from hazardous chemical reagents to a biocatalytic system fundamentally alters the cost structure of manufacturing these complex intermediates by removing the dependency on volatile commodity chemicals like lithium aluminum hydride and precious metal catalysts. The elimination of expensive heavy metal scavengers and the reduction in solvent consumption for purification steps contribute to a significantly lower cost of goods sold (COGS). Furthermore, the ability to reuse the immobilized enzyme for multiple batches drastically reduces the recurring cost of the biocatalyst itself, transforming what was once a single-use expense into a durable capital asset. This reusability also simplifies inventory management, as there is less need to store large quantities of unstable or hazardous reagents, thereby lowering warehousing costs and insurance premiums associated with dangerous goods.
- Cost Reduction in Manufacturing: The enzymatic process eliminates the need for costly stoichiometric reagents such as silver salts and cyanides, which are not only expensive but also incur high disposal costs due to their toxicity. By operating in an aqueous medium at ambient temperatures, the process significantly reduces energy consumption related to heating, cooling, and solvent recovery, leading to substantial utility savings. The simplified workup procedure, which avoids complex chromatographic separations, further reduces the consumption of silica gel and organic solvents, directly impacting the variable costs of production. Additionally, the high stereoselectivity of the enzyme minimizes the loss of valuable chiral material to unwanted isomers, maximizing the effective yield of the desired API intermediate and reducing the overall material input required per kilogram of product.
- Enhanced Supply Chain Reliability: Relying on a biocatalytic route diversifies the supply chain away from petrochemical-derived reagents that are subject to market volatility and geopolitical disruptions. The recombinant enzyme can be produced via fermentation using renewable feedstocks, ensuring a sustainable and secure supply of the critical catalyst. The robustness of the immobilized enzyme allows for longer shelf-life and easier transportation compared to sensitive chemical catalysts that may require inert atmosphere handling or cryogenic storage. This stability ensures that production schedules are not disrupted by catalyst degradation or supply shortages, providing a more predictable lead time for the delivery of high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: The aqueous nature of the reaction system makes it inherently safer and easier to scale up from pilot plant to commercial production volumes without the exponential increase in risk associated with exothermic chemical reactions. The process generates significantly less hazardous waste, particularly heavy metal sludge and halogenated solvent waste, facilitating easier compliance with increasingly stringent environmental regulations. The reduced E-factor (mass of waste per mass of product) aligns with green chemistry principles, enhancing the corporate sustainability profile of the manufacturer. This environmental advantage not only mitigates regulatory risk but also appeals to end-users who are prioritizing suppliers with strong environmental, social, and governance (ESG) credentials in their sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this immobilized enzyme technology in pharmaceutical manufacturing. These answers are derived directly from the experimental data and specifications provided in the patent literature to ensure accuracy and reliability for process development teams.
Q: What are the advantages of using immobilized monoamine oxidase over free enzymes?
A: The immobilized enzyme described in patent CN105624128B offers firm binding with minimal loss of enzymatic activity, simple separation from the reaction mixture, and the ability to be reused for multiple batches, significantly reducing operational costs compared to free enzymes which are difficult to recover.
Q: How does this biocatalytic route compare to traditional chemical synthesis for Fragment A?
A: Traditional chemical routes often require harsh reagents like lithium aluminum hydride, silver nitrate, or potassium cyanide, and extreme conditions such as -78°C. The enzymatic route operates under mild aqueous conditions (20-35°C, pH 6.0-8.0), eliminating the need for toxic heavy metals and complex protection-deprotection steps.
Q: What is the stability and reusability of the immobilized catalyst?
A: The patented immobilization method ensures high stability, allowing the catalyst to be reused for at least 5 batches while maintaining high conversion rates (>90%) and significant residual enzyme activity, demonstrating excellent potential for continuous industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Azabicyclo Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes for complex pharmaceutical intermediates like the chiral azabicyclo compounds used in HCV therapeutics. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify stereochemical integrity and impurity profiles. Our state-of-the-art facilities are equipped to handle both chemical and biocatalytic processes, allowing us to offer flexible manufacturing solutions tailored to the specific needs of your drug development pipeline.
We invite you to collaborate with us to leverage this cutting-edge biocatalytic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this enzymatic route for your specific application. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in immobilized enzyme technology can drive efficiency and quality in your supply chain.
