Revolutionizing Darunavir Intermediate Synthesis via High-Concentration Alcohol Tolerant Biocatalysis
The pharmaceutical industry constantly seeks robust synthetic routes for complex antiretroviral agents, and Patent CN111235123A presents a groundbreaking advancement in the biocatalytic production of Darunavir intermediates. This intellectual property discloses a novel carbonyl reductase derived from Lactobacillus strains via site-directed mutagenesis, specifically engineered to withstand high concentrations of alcoholic solutions. Traditional biocatalytic processes often struggle with substrate solubility, necessitating the addition of auxiliary cosolvents that complicate downstream purification and introduce impurities. However, this innovative enzyme maintains exceptional catalytic activity even in reaction systems containing up to 60% isopropanol by mass volume. This tolerance allows for a dramatic increase in substrate concentration, potentially reaching 200g/L or higher, which directly translates to improved space-time yields and reduced reactor volumes. For R&D directors and process chemists, this represents a pivotal shift towards more efficient, environmentally benign manufacturing protocols that align with modern green chemistry principles while ensuring the stringent stereochemical control required for HIV protease inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
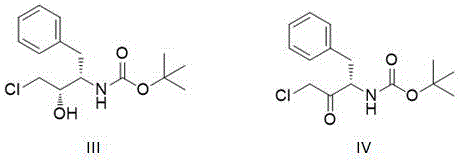
Historically, the synthesis of (2S,3S)-N-tert-butyloxycarbonyl-3-amino-1-chloro-2-hydroxy-4-phenylbutane, a critical intermediate for Darunavir, has relied heavily on chemical reduction methods or early-generation biocatalysts with significant operational drawbacks. Conventional chemical routes often utilize aluminum hydride reagents such as lithium tri-tert-butoxyaluminum hydride, which, while effective, generate substantial amounts of hazardous waste and require rigorous safety protocols due to their pyrophoric nature. Furthermore, earlier biocatalytic attempts faced severe limitations regarding substrate solubility; since both the ketone substrate and the alcohol product are poorly soluble in aqueous buffers, prior art methods were forced to incorporate surfactants like Tween 60 or organic cosolvents like toluene to maintain system fluidity. These additives not only increased the complexity of the reaction mixture but also created significant hurdles during the isolation and purification stages, often capping the achievable optical purity at around 99% and necessitating energy-intensive separation steps to remove residual surfactants and solvents from the final API intermediate.
The Novel Approach
The technology described in CN111235123A fundamentally overcomes these solubility and purity bottlenecks by leveraging a mutated carbonyl reductase that thrives in high-alcohol environments. By engineering the enzyme to tolerate isopropanol concentrations that would typically denature wild-type proteins, the process eliminates the absolute need for external cosolvents or surfactants. Isopropanol serves a dual function in this system: it acts as an excellent solvent to dissolve high loads of the hydrophobic substrate, and simultaneously functions as the hydrogen donor for cofactor regeneration. This dual utility simplifies the reaction matrix significantly, creating a cleaner system that facilitates easier product recovery. The ability to operate at substrate concentrations of 200g/L without system viscosity issues means that manufacturers can achieve higher throughput in existing infrastructure, effectively decoupling production capacity from reactor size limitations while delivering a product with superior diastereomeric excess values exceeding 99.9%.
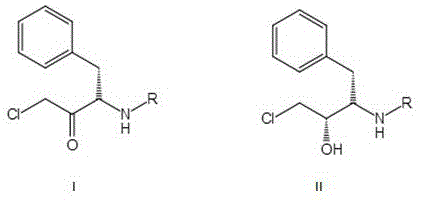
Mechanistically, the transformation involves the stereoselective reduction of the prochiral ketone moiety in the substrate to a chiral secondary alcohol with (S) configuration. As illustrated in the structural diagrams, the substrate contains a chloroacetyl group and a protected amine, requiring precise enzymatic control to avoid racemization or side reactions at the adjacent chiral center. The novel carbonyl reductase achieves this by binding the substrate in a specific orientation within its active site, ensuring that hydride transfer from the cofactor occurs exclusively from one face of the carbonyl plane. This high degree of specificity is crucial for Darunavir synthesis, where the wrong stereoisomer could act as an impurity or even an antagonist. The enzyme's active site architecture, modified through specific amino acid substitutions at positions 8, 173, 190, and 226, creates a microenvironment that stabilizes the transition state even in the presence of high organic solvent content, preventing the conformational collapse that typically leads to enzyme inactivation.
Mechanistic Insights into Carbonyl Reductase Catalytic Cycle
The core of this biocatalytic efficiency lies in the coupled redox system that drives the reaction forward while regenerating the expensive nicotinamide cofactors. In a standard reduction, the enzyme consumes NADH or NADPH to reduce the ketone substrate, oxidizing the cofactor to NAD+ or NADP+. Without a regeneration system, this would be economically unviable due to the high cost of stoichiometric cofactors. The patented process solves this by utilizing isopropanol as a sacrificial hydrogen donor. The carbonyl reductase, or a coupled dehydrogenase system, oxidizes the isopropanol to acetone, thereby reducing NAD+ back to NADH. This creates a closed loop where a catalytic amount of cofactor can drive the conversion of large quantities of substrate. The unique tolerance of this specific mutant enzyme to the accumulating acetone and high isopropanol concentrations ensures that the equilibrium is driven towards product formation without inhibiting the enzyme's turnover number, a common failure point in less robust biocatalytic systems.
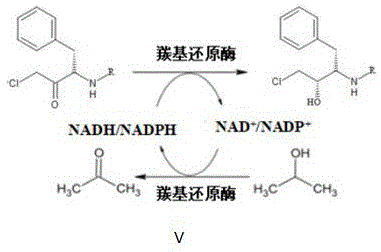
Impurity control in this system is inherently superior due to the absence of extraneous chemical additives. In traditional methods using glucose as a hydrogen donor, the accumulation of gluconic acid can lower the pH of the reaction mixture, requiring constant base addition which increases the ionic strength and complicates crystallization. Conversely, the isopropanol-driven system generates acetone, a volatile byproduct that can be easily removed via evaporation or distillation during the workup phase. Furthermore, the high stereoselectivity of the enzyme minimizes the formation of the (2R,3S) diastereomer, which is a critical quality attribute for the final drug substance. The combination of high substrate loading and clean reaction profiles means that the crude product obtained after simple concentration and crystallization already meets high purity specifications, reducing the burden on analytical QC labs and shortening the overall batch cycle time for commercial manufacturing campaigns.
How to Synthesize (2S,3S)-2-hydroxy-4-phenylbutane Derivatives Efficiently
To implement this advanced biocatalytic route, manufacturers must first establish the expression system for the recombinant carbonyl reductase, typically using E. coli BL21(DE3) hosts transformed with the optimized gene sequence. The process begins with the preparation of a reaction buffer, preferably potassium or sodium phosphate at pH 8.0, supplemented with magnesium ions to enhance enzyme stability. The substrate is then introduced into the system alongside a high concentration of isopropanol, ranging from 20% to 50% v/v, which serves to solubilize the reactant and drive the cofactor cycle. Once the temperature is equilibrated to between 30°C and 45°C, the crude enzyme solution is added to initiate the reduction. Detailed standardized operating procedures regarding induction times, cell lysis methods, and specific feeding strategies are critical for reproducibility and are outlined in the technical documentation below.
- Prepare a reaction system containing the substrate (3S)-4-phenyl-2-butanone derivative, high-concentration isopropanol (20-60% m/V), and buffer solution.
- Add the recombinant carbonyl reductase (CXP I) along with cofactor NAD+/NADP+ and magnesium ions to initiate the reduction.
- Maintain reaction at 30-45°C and pH 7.0-9.0 for 24 hours to achieve >99% conversion and >99.9% de value.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this high-alcohol tolerant biocatalysis offers profound strategic benefits centered around cost structure optimization and supply security. The most immediate impact is the drastic simplification of the raw material portfolio; by removing the requirement for specialized surfactants, toxic cosolvents like toluene, and stoichiometric chemical reducing agents, the bill of materials becomes leaner and less susceptible to volatility in specialty chemical markets. Additionally, the ability to run reactions at significantly higher substrate concentrations means that the same production volume can be achieved in smaller reactors or, conversely, that existing assets can produce substantially more output per batch. This intensification of the process directly lowers the fixed cost allocation per kilogram of product, enhancing margin potential without compromising on the rigorous quality standards demanded by global regulatory agencies for HIV therapeutics.
- Cost Reduction in Manufacturing: The elimination of auxiliary solvents and surfactants removes entire unit operations from the downstream processing train, such as extensive washing steps required to remove Tween 60 or extraction steps to remove toluene. This reduction in processing complexity translates to lower utility consumption, reduced solvent waste disposal costs, and decreased labor hours per batch. Furthermore, the use of isopropanol as both solvent and reagent consolidates material costs, and the volatile nature of the acetone byproduct simplifies purification, avoiding the need for expensive chromatographic separations often required to remove non-volatile impurities generated in glucose-based systems.
- Enhanced Supply Chain Reliability: Relying on a robust enzymatic process reduces the risk of batch failures associated with sensitive chemical reductions that require strict anhydrous conditions or cryogenic temperatures. The mild operating conditions of this biocatalytic method (ambient pressure, moderate temperature) make the supply chain more resilient to infrastructure disruptions. Moreover, the high yield and conversion rates ensure that raw material utilization is maximized, reducing the frequency of raw material orders and buffering the production schedule against upstream supply fluctuations of the starting ketone substrate.
- Scalability and Environmental Compliance: Scaling biocatalytic processes is generally more linear and predictable than scaling exothermic chemical reductions, which pose significant safety risks at large scales. This technology aligns perfectly with increasing global pressure to reduce the environmental footprint of pharmaceutical manufacturing. By replacing heavy metal reagents and chlorinated solvents with a bio-based catalyst and green solvent system, manufacturers can significantly improve their E-factor (environmental factor) metrics. This not only aids in meeting internal sustainability goals but also streamlines regulatory filings in jurisdictions with strict environmental compliance requirements, facilitating faster market access for generic or branded versions of the final drug product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonyl reductase technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific manufacturing needs. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: What is the primary advantage of this carbonyl reductase over conventional enzymes?
A: Unlike conventional carbonyl reductases that lose activity in high alcohol concentrations, this mutant enzyme maintains high catalytic efficiency in up to 60% isopropanol, allowing for higher substrate loading without auxiliary cosolvents.
Q: How does this process improve product purity for Darunavir intermediates?
A: By eliminating the need for surfactants like Tween 60 or organic cosolvents like toluene, the downstream purification is simplified, resulting in a final product with chiral purity exceeding 99.95% de.
Q: Can this biocatalytic method replace chemical reduction methods?
A: Yes, it offers a greener alternative to aluminum salt reductions, avoiding harsh conditions and heavy metal waste while achieving comparable or superior yields and stereoselectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Darunavir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent examples can be reliably reproduced in our GMP-compliant facilities. We understand that for critical antiviral intermediates, consistency is paramount; therefore, our stringent purity specifications and rigorous QC labs are designed to detect and control even trace levels of diastereomeric impurities, guaranteeing that every batch meets the exacting standards required for downstream API synthesis.
We invite you to engage with our technical procurement team to discuss how this advanced biocatalytic route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We are prepared to provide specific COA data from pilot runs and comprehensive route feasibility assessments to demonstrate our capability to deliver high-purity Darunavir intermediates with the reliability and efficiency your organization demands.
