Advanced Catalytic Synthesis of Tetraazamacrocycle Intermediates for Commercial Scale-Up
The pharmaceutical and diagnostic imaging industries rely heavily on the availability of high-purity macrocyclic chelating agents, particularly for the formulation of stable Gadolinium-based MRI contrast media. Patent CN1101817C discloses a groundbreaking preparation method for tetraazamacrocycles, specifically focusing on the synthesis of 1,4,7,10-tetraazacyclododecane, commonly known as Cyclen. This compound serves as the critical "building block" for forming stable complexes with paramagnetic metal ions, solving toxicity issues associated with free metal ions in medical diagnostics. The patent introduces a novel catalytic strategy that overcomes the economic and technical limitations of traditional alkylation methods, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
Historically, the synthesis of these intricate nitrogen-containing rings has been plagued by the high cost of reagents and the difficulty in controlling selectivity during the ring-closing steps. Conventional methods often relied on 1,2-dibromoethane as the alkylating agent due to its high reactivity. However, this reagent is not only significantly more expensive than its chlorinated counterparts but also poses challenges regarding side reactions, such as the unwanted quaternization of nitrogen atoms, which complicates purification and lowers overall yield. Alternatively, using 1,2-dichloroethane alone often results in sluggish reaction kinetics, requiring harsh conditions that degrade product quality. The innovation described in CN1101817C elegantly resolves this dichotomy by introducing a halide exchange catalyst system.
![Chemical structure of decahydro-2a,4a,6a,8a-tetraazacyclopenta[fg]acenaphthene intermediate (VI)](/insights/img/cyclen-intermediate-synthesis-catalytic-alkylation-supplier-20260307135159-03.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for tetraazamacrocycles typically face a trade-off between reactivity and cost efficiency. When manufacturers utilize 1,2-dibromoethane, the reaction proceeds rapidly even at moderate temperatures, but this high reactivity is a double-edged sword. It frequently leads to poly-alkylation and the formation of quaternary ammonium salts, which are difficult to separate from the desired macrocyclic product. This necessitates extensive downstream purification processes, increasing both processing time and waste generation. On the other hand, attempting to use the more economically attractive 1,2-dichloroethane without modification often fails to drive the cyclization to completion within a reasonable timeframe. The carbon-chlorine bond is stronger and less susceptible to nucleophilic attack by the amine groups, leading to incomplete conversions and the accumulation of linear oligomers rather than the desired cyclic structure.
The Novel Approach
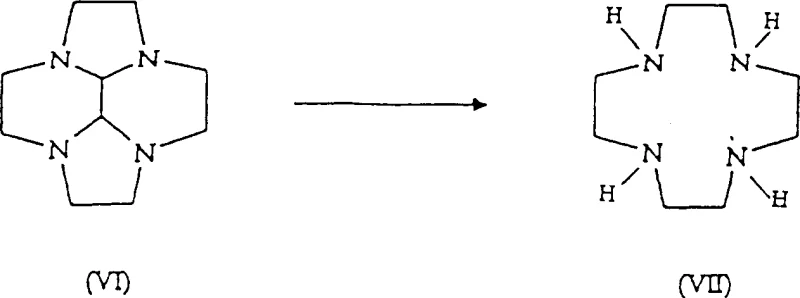
The methodology outlined in the patent introduces a sophisticated catalytic system that activates the inexpensive 1,2-dichloroethane in situ. By adding a catalytic amount of an alkali metal halide, such as sodium bromide (NaBr) or sodium iodide (NaI), to the reaction mixture containing 1,2-dichloroethane, the process generates a more reactive alkylating species dynamically. This approach leverages the Finkelstein reaction principle within the macrocyclization context. The result is a reaction environment where the alkylating agent is sufficiently active to promote ring closure at moderate temperatures (50-80°C) but is present in a controlled concentration that minimizes side reactions. This balance allows for the efficient production of the key intermediate, decahydro-2a,4a,6a,8a-tetraazacyclopenta[fg]acenaphthene (Compound VI), which is subsequently converted into the final Cyclen product.
Mechanistic Insights into Halide Exchange Catalyzed Macrocyclization
The core of this technological advancement lies in the nuanced understanding of nucleophilic substitution dynamics in dipolar aprotic solvents. In the presence of a catalyst like NaBr, a halide exchange occurs where chloride atoms in the 1,2-dichloroethane are partially replaced by bromide ions. This generates 1-bromo-2-chloroethane in situ, a mixed halide species that possesses higher reactivity than the starting dichloro compound due to the better leaving group ability of the bromide ion. However, because this species is generated catalytically and consumed rapidly by the amine nucleophile, its bulk concentration remains low. This kinetic control is crucial; it prevents the runaway alkylation that characterizes the use of pure 1,2-dibromoethane. The solvent choice, typically DMAC or DMSO, further enhances the nucleophilicity of the amine nitrogen atoms by solvating the cationic counter-ions, thereby facilitating the attack on the alkyl halide.
Furthermore, the process demonstrates exceptional impurity control mechanisms inherent to the catalytic cycle. By avoiding the use of excess highly reactive dibromo-species, the formation of polymeric by-products and quaternary salts is significantly suppressed. The reaction conditions, specifically the temperature range of 50-80°C and the use of inorganic bases like sodium carbonate, create an environment favorable for intramolecular cyclization over intermolecular polymerization. The base serves to neutralize the acid formed during the alkylation, driving the equilibrium forward without introducing organic impurities that are hard to remove. This results in a crude product with high GC purity (often exceeding 98%), reducing the burden on subsequent crystallization or distillation steps and ensuring a cleaner impurity profile for the final API intermediate.
How to Synthesize Decahydro-2a,4a,6a,8a-tetraazacyclopenta[fg]acenaphthene Efficiently
The synthesis of this critical intermediate involves a multi-step sequence beginning with the condensation of triethylenetetramine and glyoxal, followed by the pivotal catalytic alkylation step. The process is designed to be operationally simple while maximizing yield and minimizing raw material costs. The following guide outlines the standardized procedure derived from the patent examples, highlighting the critical parameters for successful implementation in a pilot or commercial plant setting.
- Condense triethylenetetramine with glyoxal derivatives in water with calcium hydroxide to form the octahydro-tetraazanaphthylene intermediate.
- Perform catalytic alkylation using 1,2-dichloroethane and a sodium halide catalyst (NaBr or NaI) in a dipolar aprotic solvent like DMAC.
- Oxidize and hydrolyze the resulting tetracyclic compound under acidic or alkaline conditions to yield the final 1,4,7,10-tetraazacyclododecane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic alkylation technology represents a strategic opportunity to optimize the cost structure of high-value diagnostic intermediates. The primary driver of value is the substitution of a specialty reagent with a commodity chemical. 1,2-dibromoethane is a relatively expensive reagent with a more complex supply chain compared to 1,2-dichloroethane, which is produced on a massive industrial scale for other applications. By shifting the dependency to the chlorinated analog and using a cheap salt catalyst, the direct material costs are drastically reduced. The patent explicitly notes that 1,2-dibromoethane can cost approximately seven times more than 1,2-dichloroethane, and the catalytic system allows for a theoretical cost saving of roughly 72% per mole of product based on raw material arithmetic.
- Cost Reduction in Manufacturing: The economic impact of this process extends beyond simple raw material substitution. The improved selectivity of the catalytic system means that less material is lost to side products, effectively increasing the yield per batch. Higher yields translate directly to lower cost of goods sold (COGS) and reduced waste disposal costs. Furthermore, the ability to run the reaction at moderate temperatures (50-80°C) rather than requiring extreme heating or cooling reduces energy consumption. The elimination of complex purification steps required to remove quaternary ammonium impurities further streamlines the manufacturing process, lowering labor and utility overheads significantly.
- Enhanced Supply Chain Reliability: Relying on 1,2-dichloroethane mitigates supply risk. As a high-volume commodity chemical, its availability is generally stable and less prone to the market fluctuations that affect specialized brominated reagents. This stability ensures consistent production schedules and reduces the likelihood of stockouts that could disrupt the supply of critical MRI contrast agent precursors. Additionally, the catalyst (NaBr or NaI) is a standard inorganic salt with a robust global supply chain, ensuring that the key enabling technology components are always accessible to the manufacturing site without long lead times.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process offers distinct advantages for large-scale operations. The reduction in hazardous waste generation, due to fewer side reactions and higher atom economy, simplifies compliance with increasingly stringent environmental regulations. The use of water-soluble inorganic bases and the potential for solvent recovery in dipolar aprotic systems aligns with green chemistry principles. The process is inherently scalable, as demonstrated by the patent examples which move from gram to multi-hundred-gram scales without loss of efficiency, making it suitable for transfer to 100 MT annual commercial production facilities with minimal re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tetraazamacrocycle synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN1101817C, providing clarity on the operational flexibility and performance expectations of the catalytic method.
Q: Why is the NaBr/1,2-dichloroethane system superior to using 1,2-dibromoethane directly?
A: The combination allows for in situ generation of a reactive alkylating species that balances reactivity and selectivity, avoiding the excessive side reactions and quaternization often seen with pure 1,2-dibromoethane, while utilizing significantly cheaper raw materials.
Q: What solvents are recommended for this macrocyclization process?
A: Dipolar aprotic solvents such as dimethylacetamide (DMAC), dimethylformamide (DMF), dimethyl sulfoxide (DMSO), or N-methyl-pyrrolidone are preferred to facilitate the nucleophilic substitution and stabilize the transition states.
Q: How does this process impact the cost of goods for MRI contrast agent precursors?
A: By replacing expensive 1,2-dibromoethane with commodity-grade 1,2-dichloroethane and a catalytic amount of salt, the raw material costs are drastically reduced, with patent data estimating potential savings of over 70% per mole of product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4,7,10-Tetraazacyclododecane Intermediate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising economics of the catalytic alkylation process described in CN1101817C can be fully realized in your supply chain. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of tetraazamacrocycle intermediate meets the exacting standards required for pharmaceutical and diagnostic applications.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your sourcing strategy. By leveraging our CDMO expertise, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective supply of high-purity chelating agent precursors for your next-generation medical imaging products.
